65% of eligible lung cancer patients do not receive the most appropriate targeted therapies, Diaceutics report finds
Despite advances in biomarker testing, real-world data shows progress is not translating into treatment decisions, with the largest patient loss now occurring at the point of treatment decision
14 May 2026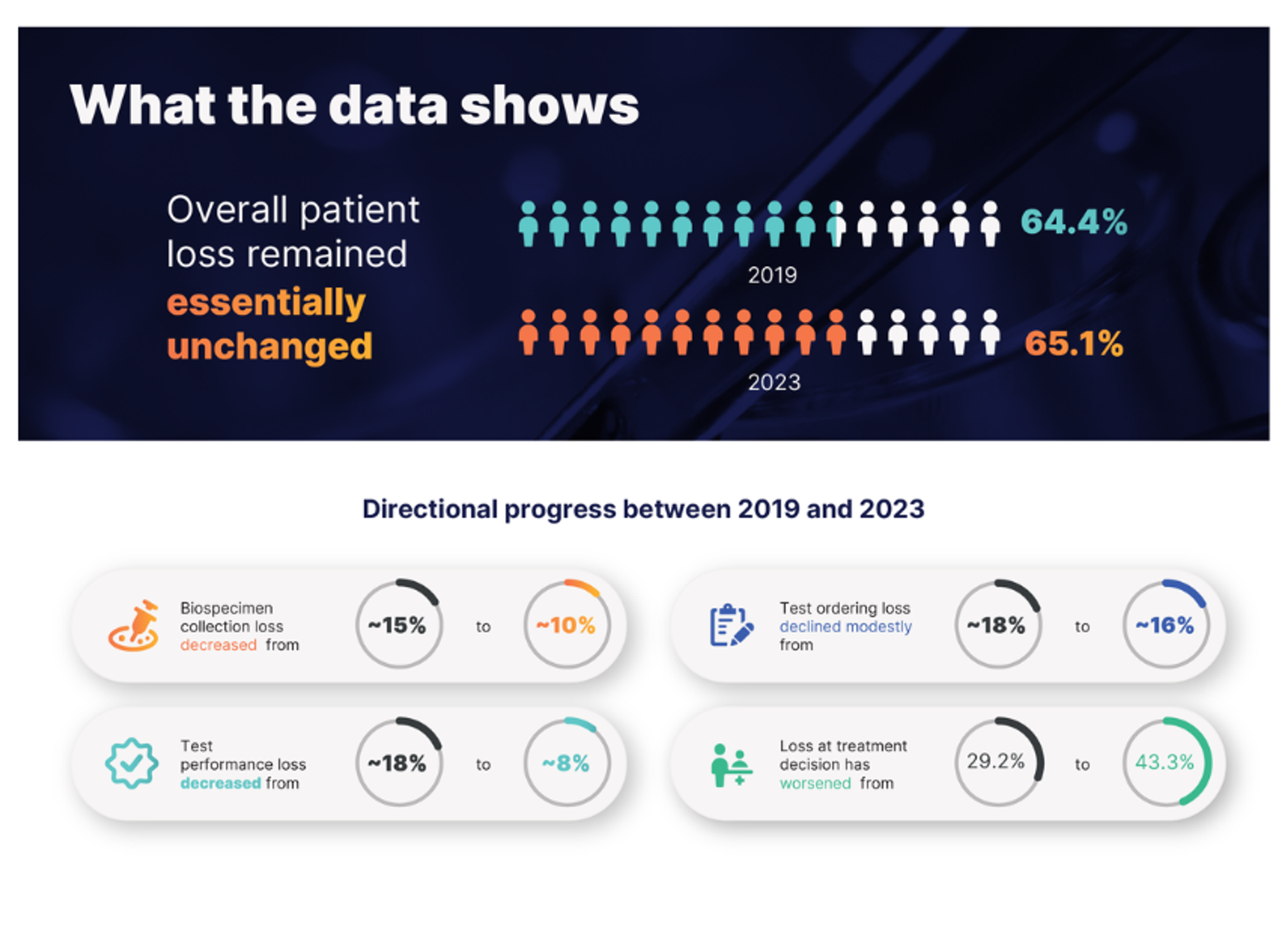
Industry news
Diaceutics, the intelligence and engagement company focused on unlocking the full potential of diagnostic-driven therapies, has released US research showing that nearly two-thirds of eligible patients with advanced non-small cell lung cancer (aNSCLC) are still not receiving the most appropriate treatment, even as biomarker testing improves1.
The updated Clinical Practice Gaps report, based on real-world data from more than 35,800 newly diagnosed aNSCLC patients between 2019 and 2023, reveals that the largest loss of patients now occurs at the treatment decision stage, underscoring a critical disconnect between precision diagnostics and therapy selection.
Updated Clinical Practice Gaps report builds on 2022 Journal of Clinical Oncology study
The new Clinical Practice Gaps report extends Diaceutics’ 2022 study, published in the Journal of Clinical Oncology, by applying the same seven-step precision medicine framework to a larger, more recent US aNSCLC cohort. The analysis compares outcomes from 2019 to 2023 for more than 35,800 newly diagnosed patients, providing an updated view of how precision oncology is being implemented in routine clinical practice.
Despite major advances in biomarker testing technologies and broader adoption of guideline-recommended assays, overall patient loss across the precision medicine pathway remains virtually unchanged: 65.1% in 2023 compared with 64.4% in 2019.
This means that almost two out of every three eligible aNSCLC patients in the US still do not receive the most appropriate treatment, as defined using National Comprehensive Cancer Network (NCCN) guidelines.
Largest gap shifts to treatment decision stage
While earlier stages of the precision medicine pathway show signs of improvement, the Diaceutics analysis identifies a significant shift in where patients are being lost. The largest gap now occurs at the point of treatment decision, rather than at the testing stage.
Patient loss at the treatment decision step has increased from 29.2% to 43.3% between 2019 and 2023, a rise of 12 percentage points. At the same time, patient losses earlier in the pathway have declined, reflecting positive progress in diagnostic access, test ordering, and biomarker result generation.
However, these diagnostic gains are not translating into better overall treatment options for patients. Instead, suboptimal or premature treatment decisions are preventing many individuals with aNSCLC from receiving therapies aligned with their biomarker profiles and NCCN guideline recommendations.
From testing problem to translation problem in precision oncology
Susanne Munksted, Chief Precision Medicine Officer at Diaceutics, emphasized that the core challenge in precision oncology has evolved.
“The headline hasn’t changed, around two-thirds of patients still miss out on the right treatment. But where we’re losing them has fundamentally shifted. Testing has improved thanks to a number of innovations and system-wide investments, the problem has just moved downstream. This is no longer primarily a testing problem; it is now a translation problem, and it demands a different response from the wider ecosystem, including industry.”
The findings point to a growing disconnect between expanding diagnostic capabilities and real-world treatment decision-making. As the number of actionable biomarkers and targeted therapies for non-small cell lung cancer increases, oncologists face rising complexity and time pressure when making first-line treatment choices.
Real-world data reveal how oncologists use biomarker information
Diaceutics’ research highlights specific behaviors that contribute to treatment decision failure in the US aNSCLC setting:
- 86% of oncologists form treatment plans before full biomarker results are available.
- Only 43% consistently wait for complete biomarker results before initiating first-line therapy.
- More than half of oncologists do not consistently act on the recommendations provided in biomarker reports.
These patterns mean that, even when high-quality biomarker testing is performed and relevant results are generated, many patients do not benefit from the most appropriate precision therapies. The gap between testing and treatment is increasingly driven by how, when, and whether clinicians integrate diagnostic insights into their decisions.
Call for new operating models in precision medicine
The updated Clinical Practice Gaps report calls for a fundamental shift in how precision medicine is operationalized across the oncology ecosystem. Diaceutics argues that stakeholders must rebalance their focus toward the treatment decision stage, ensuring that biomarker data are consistently ordered, interpreted, and applied in real time.
Key priorities highlighted in the report include:
- Improving consistency in biomarker test ordering in line with NCCN guidelines.
- Delivering real-time, actionable decision support to physicians at the point of care.
- Developing operating models that are diagnostic-enabled, data-driven, and continuously adaptive.
“We need a more systemic approach to making precision medicine the path of least resistance,” Munksted added. “To close the gap between testing and treatment, the industry must develop and adopt operating models that are diagnostic-enabled, data-driven, and continuously adaptive, to optimize the patient journey from tested, to identified, to appropriately treated.”
Diaceutics will continue to expand its real-world analysis of the precision medicine pathway in advanced non-small cell lung cancer and other indications, aiming to better understand and address the drivers of treatment decision failure.
NCCN guidelines as benchmark for appropriate treatment
In this analysis, Diaceutics relied on National Comprehensive Cancer Network (NCCN) guidelines to determine whether patients with advanced non-small cell lung cancer were receiving the most appropriate treatment. By using NCCN recommendations as a benchmark, the study provides a standardized view of how well current clinical practice aligns with evidence-based precision oncology.
References
1. The full report is available here: The Clinical Practice Gaps in Precision Medicine. Lead the Change: Close the Practice Gaps
Want the latest science news straight to your inbox? Become a SelectScience member for free today>>
Frequently asked questions
How does Diaceutics’ updated Clinical Practice Gaps report reveal ongoing treatment gaps for advanced non-small cell lung cancer (aNSCLC) patients in the US?
Diaceutics’ updated Clinical Practice Gaps report, based on real-world data from more than 35,800 newly diagnosed advanced non-small cell lung cancer (aNSCLC) patients in the US between 2019 and 2023, shows that nearly two-thirds of eligible patients still do not receive the most appropriate treatment as defined by National Comprehensive Cancer Network (NCCN) guidelines.
Overall patient loss across the precision medicine pathway remains virtually unchanged, at 65.1% in 2023 compared with 64.4% in 2019. This means that despite improvements in biomarker testing technologies and broader adoption of guideline-recommended assays, around two out of every three aNSCLC patients are still missing out on optimal, guideline-aligned precision therapies.
What does the Diaceutics research reveal about the shift from a testing problem to a translation problem in precision oncology for non-small cell lung cancer?
The Diaceutics research indicates that the main barrier in precision oncology for advanced non-small cell lung cancer has shifted from access to biomarker testing to the translation of test results into treatment decisions.
While earlier stages of the precision medicine pathway, such as diagnostic access, test ordering, and biomarker result generation, have improved, the largest loss of patients now occurs at the treatment decision stage. Patient loss at this step increased from 29.2% to 43.3% between 2019 and 2023.
The study shows that 86% of oncologists form treatment plans before full biomarker results are available, only 43% consistently wait for complete biomarker results before starting first-line therapy, and more than half do not consistently act on biomarker report recommendations. As a result, high-quality biomarker testing is not consistently translated into appropriate, NCCN-guideline-aligned treatment choices.
Why does Diaceutics call for new operating models in precision medicine for aNSCLC, and what priorities are highlighted to improve biomarker-driven treatment decisions?
Diaceutics calls for new operating models in precision medicine for advanced non-small cell lung cancer because current clinical practice is not fully leveraging biomarker data to guide treatment decisions, leading to persistent gaps between testing and therapy selection.
The updated Clinical Practice Gaps report emphasizes that stakeholders across the oncology ecosystem must rebalance their focus toward the treatment decision stage, ensuring that biomarker data are consistently ordered, interpreted, and applied in real time.
Key priorities include improving consistency in biomarker test ordering in line with NCCN guidelines, delivering real-time, actionable decision support to physicians at the point of care, and developing operating models that are diagnostic-enabled, data-driven, and continuously adaptive.
According to Diaceutics and Chief Precision Medicine Officer Susanne Munksted, a more systemic, diagnostic-enabled approach is needed to make precision medicine the path of least resistance and to optimize the journey from tested, to identified, to appropriately treated aNSCLC patients.
