Live Cell Analysis in Chronic Inflammation Research at the University of Oxford
Professor David R. Greaves, University of Oxford, UK, discusses the technology that is enabling him to research tissue repair and chronic inflammation in real time
1 Mar 2017

David R. Greaves, University of Oxford, UK
Professor David R. Greaves is using the latest live cell imaging technology to carry out ground-breaking research into inflammation biology. Sonia Nicholas, Associate Editor for SelectScience®, spoke to Professor Greaves to find out more.
SN: Please could you confirm your name, full job title and place of work.
DRG: I’m Professor David R. Greaves, I am a University Lecturer in Cellular Pathology at the Sir William Dunn School of Pathology, University of Oxford, UK.
SN: Could you tell us about your job, what you do and what your responsibilities are?
DRG: I run a research laboratory working on macrophage biology and inflammation. I am very interested in macrophage chemotaxis as well as other aspects of macrophage cell biology such as phagocytosis and cytokine secretion.
In addition to doing biomedical research I run the BM Principles of Pathology course for second year medical students at the University of Oxford, I run a final year lecture course in Inflammation Biology, I run the British Heart Foundation 4-year Cardiovascular Sciences PhD program and I am a Tutorial Fellow in Medicine at Hertford College where I give tutorials in Biochemistry, Cell Biology, Endocrinology, Medical Genetics and Pathology.
Inflammation and disease
SN: Can you tell us more about your research into inflammation?
DRG: Inflammation is the normal physiological response to tissue injury and infection. Most of the time our inflammatory responses are of an appropriate magnitude, they are quickly resolved and any damage to our tissues is successfully repaired. Inflammation is important because it drives the development of many important human diseases including rheumatoid arthritis, cardiovascular disease, inflammatory bowel disease and many others. Recent research suggests chronic inflammation may be an important driver of major mood disorders including depression.
My research is aimed at identifying endogenous pathways that are involved in regulating the magnitude and duration of inflammatory responses. Recently, we have been looking at the role of two independent cell signaling pathways in regulating the inflammatory response. One is centered on endocannabinoids – a class of lipids that signals via a G protein coupled receptor (GPCR) called CB2 and the other pathway is centered on an unusual cytokine called Chemerin (TIG-2) whose effects are mediated by three different GPCRs – ChemR23, CCRL2 and GPR1.
Macrophages in healthy and inflamed tissues play an essential role in the initiation and resolution of inflammation. One important aspect of macrophage biology in the context of inflammation resolution is phagocytosis of cellular debris and phagocytosis of neutrophils that have undergone apoptosis. Macrophage phagocytosis of apoptotic cells (efferocytosis) has a profound effect on inflammation resolution. Macrophage efferocytosis changes the profile of macrophage cytokine secretion towards a more anti-inflammatory / pro-resolution phenotype, which in turn will enhance inflammation resolution. Failure to clear apoptotic neutrophils from a site of inflammation can lead to failure of resolution and a substantially worse outcome caused by secondary necrosis.
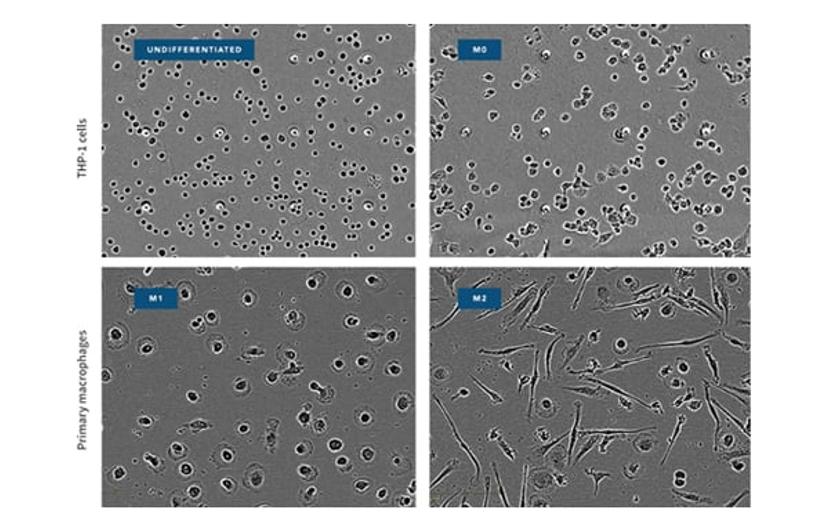
A powerful research tool
SN: How does the IncuCyte technology help you to achieve your research goals?
DRG: We have now been using the IncuCyte Live-Cell Analysis System to study several different aspects of macrophage cell biology in a wide range of different applications. We have found this real time live cell imaging system to be easy to use and the associated image analysis software makes this a very powerful research tool.
- We have used the IncuCyte system to study real time chemotaxis of murine primary macrophages and shape change in response to the potent chemoattractant Complement C5a (Taylor L and Brodermann M et al. PLoS One. 2016 ; 11(8):e0160685. doi: 10.1371/journal.pone.0160685). In our published study we were able to directly compare macrophage real time chemotaxis using the IncuCyte system with the ACEA xCELLigence system. Reassuringly we obtained very similar results for inhibition of macrophage chemotaxis using recombinant Netrin-1 protein in both systems.
- By combining pHRodo labeled bioparticles and the IncuCyte cell imaging system we were able to study the kinetics of macrophage phagocytosis using both primary murine macrophages from a range of different tissues (Kapellos T et al. Biochem Pharmacol. 2016; 116:107-19. doi: 10.1016/j.bcp.2016.07.011.) and human monocyte derived macrophages (our unpublished data). In future studies we want to extend our studies to new phagocytic cells such as microglia and Kupffer cells and new phagocytic meals including apoptotic cells and beta amyloid found in Alzheimer’s disease plaques.
- We have used the IncuCyte system to follow in vitro differentiation of bone marrow progenitor cells derived from our hCD68-Gfp transgenic mice into macrophages or dendritic cells (McNeill, Iqbal et al. Arterioscler Thromb Vasc Biol. 2017; 37: 258-263. doi: 10.1161/ATVBAHA.116.308367). These experiments took advantage of the IncuCyte system’s ability to continuously monitor cell growth and differentiation without having to take the cell cultures out of the tissue culture incubator. The results we obtained agreed well with single time point flow cytometry analyses.

IncuCyte Live-Cell Analysis System
SN: How did you monitor cell behavior before you installed the IncuCyte? What does this technology enable you to do that you couldn’t do before?
DRG: All the macrophage biology experiments that we have published using the IncuCyte platform could have been performed using other imaging modalities but I think that the big advantage of the IncuCyte live cell imaging platform lies in the ease of use and ease of analysis compared to other cell imaging methods (confocal microscopy, flow cytometry and Imagestream). What we particularly like about the IncuCyte system is the ability to develop protocols to study both generalized and cell type specific behavior. For instance we can follow proliferation or apoptosis of macrophages, and we can study macrophage specific cell behavior such as apoptosis or chemotaxis. Data analysis is greatly facilitated by user friendly software.
SN: What is next for your research?
DRG: I want to start using the IncuCyte system to do ‘scratch wound migration assays’ where we look for macrophage secreted factors that play a role in wound repair processes. Hopefully we can scale up this cell-based assay to look for novel chemicals, peptides and proteins that will enhance tissue repair in the context of inflammation resolution.
The long-term goal of research in my laboratory is to turn high quality basic science into new treatments that enhance wound repair and help resolve chronic inflammation. Our ability to study both murine and human macrophages on the IncuCyte platform will be important in future translational research programs.
SN: Do you have any advice for other researchers who are considering using IncuCyte technology?
DRG: Take your time in setting up the assays before you pile in to testing lots of different mediators, drugs etc. Every cell type is different so ‘one size fits all’ protocols are unlikely to work first time!

