Improving Joint Health with Putative Therapeutics at Matis, Iceland’s Food & Biotechnology Research Institute
Use of cell imaging technology enables cutting-edge research into the assessment of bioactive compounds in cell lines
26 Apr 2017

Dr. Eva Kuttner Dr. Eva Kuttner is a Project Leader in the Analysis and Structure division at Matis.
Matis is a leading food and biotechnology research institute in Iceland. Matis’ vision is to increase the value of food processing and food production, and to ensure the safety and quality of food and food products. SelectScience® spoke to Dr. Eva Kuttner, to learn more about the technology the company is using in its research.
SS: Could you briefly introduce yourself and your place of work?
EK: My name is Dr. Eva Kuttner, I’m a Project Leader in the Analysis and Structure division at Matis (mat=food + is=Iceland). Matis is a government owned, non-profit, independent research company in Iceland. The main focus of Matis is research and development, aligned to the food and biotechnology industries, as well as providing Iceland's leading food analytical testing service for public and private authorities.
I personally am German, and I hold a Diploma (equal to an MSc) in Botany, from the Universität Braunschweig in Germany. I received my PhD in Integrative Biology (2007-2012) from the University of Guelph (Ontario, Canada).
SS: Could you describe your job role and work at Matis?
EK: My job title is Regional Manager, which means I am responsible for the 2 locations of Matis in the North of Iceland, Sauðárkrókur and Akureyri. My main work is that of a project leader (bioactives) in the Analysis and Structure Division and I am specialized in screening for bioactive secondary metabolites using in vitro chemical and cell-based assays.
Our laboratory focus is on identifying and describing compounds regarding antioxidant activity for healthcare (anti-diabetes, anti-inflammatory) and skin care. We now have a fully equipped cell laboratory (currently working with two cell lines), and we routinely run chemical and cell-based assays to identify and describe the bioactivity properties of extracted and fractionated compounds.
Testing putative therapeutics
SS: Please tell us about your marine compounds project that you are working on, and the main challenges involved?
EK: We are currently looking into how marine compounds influence the mineralization in a murine chondrocyte cell line (ATDC5). The aim of the project is to test putative therapeutics, to improve joint health. Specifically, we are interested in seeing how they affect differentiation of these cells.
SS: Which methods do you use to investigate this project and how does the BioTek Cytation 5 play a part in this?
EK: We are exposing the cells to different concentrations of fractionated marine compounds in a 96-well microplate format. After 24 days of incubation we stain the cells both with calcein and Alizarin Red to quantify calcium deposition. This is when the Cytation 5 from BioTek comes in: we have a workflow set up that images (magnification 4x) each well in phase contrast and fluorescence (we are using a GFP filter cube for this assay), and also carries out a read of the fluorescence signal. We then perform the Alizarin Red staining, take more pictures and extract the bound color after several washing steps. A further reading step quantifies the Alizarin Red using an absorbance read.
The BioTek Cytation 5 Cell Imaging Multi-Mode Reader combines automated digital widefield microscopy with conventional multi-mode microplate detection
The figure below shows an example read of the calcein stain: first picture, phase contrast, second picture, fluorescence channel (GFP filter cube) and the third picture, overlay of both:
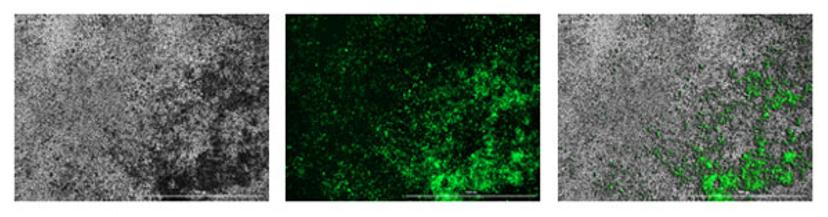
Image courtesy of Dr. Eva Kuttner
SS: What are your main research findings so far?
EK: We have identified several possible inhibitors of mineralization, but we are still in the process of improving the assay protocol to make the quantification of the mineralization more reproducible. The Alizarin Red stain has proven to represent more accurate calcium deposition into the cells, when testing the assay protocol using standard inhibitors like levamisole, so we are using the calcein stain to visualize mineralization, and Alizarin Red to quantify.
SS: What is the future of your research?
EK: We are looking into setting up assays investigating blood pressure. Our project partner developed a product line based on fish protein hydrolysate using trimmings from cod (Gadus morhua). The proteins are extracted and broken down to smaller peptides that have been shown to inhibit a key enzyme involved in increasing blood pressure (ACE). For this research we will develop and perform cell-based assays.

