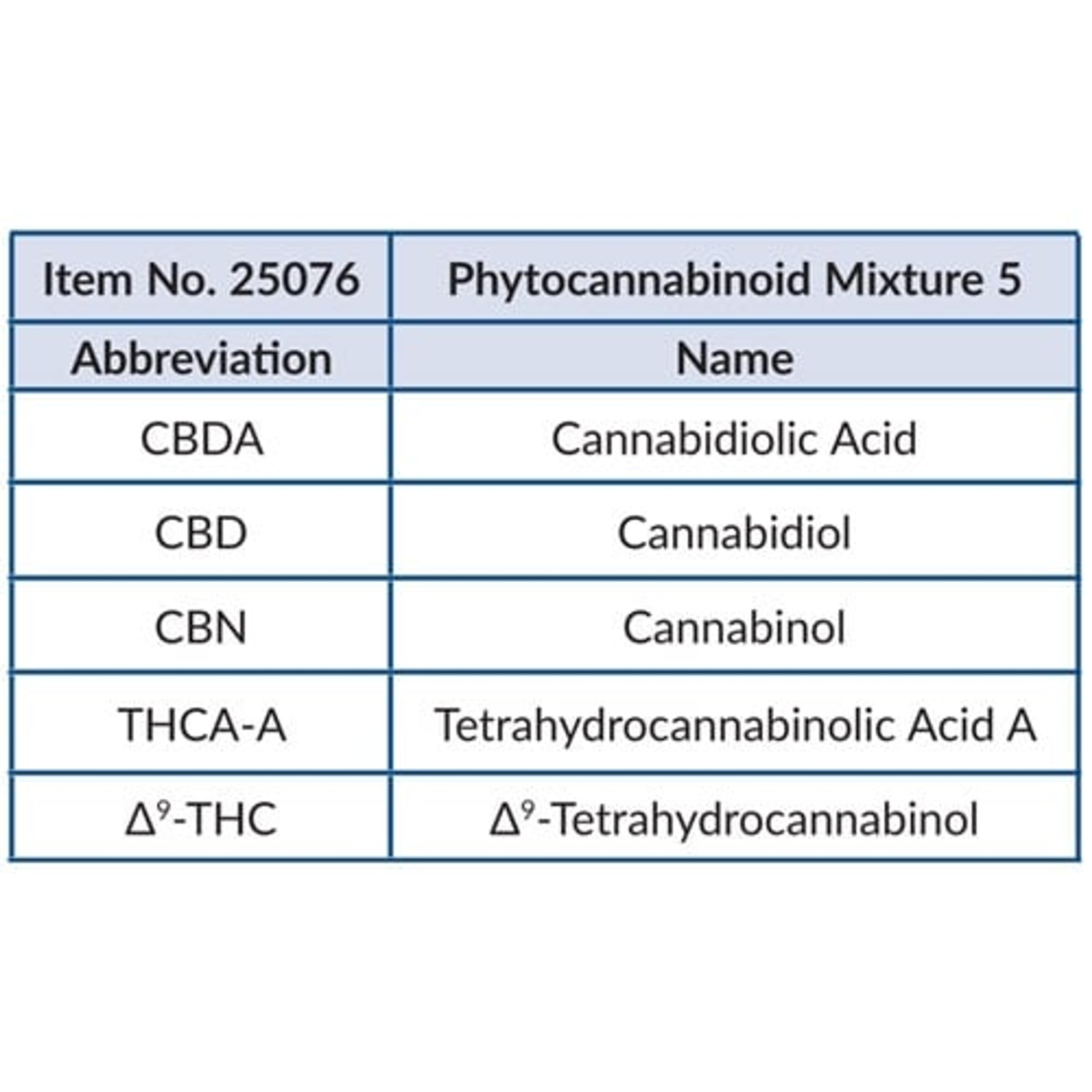
Phytocannabinoid Mixture 5 (CRM)
Cayman Chemical Co.Phytocannabinoid Mixture 5 (CRM) (Item No. 25076) is a certified reference material mixture provided as a DEA exempt preparation. Included are Δ9-THC, THCA-A, cannabidiol, cannabidiolic acid, and cannabinol. The mixture is supplied in an amber ampule in which the headspace has been purged with argon.