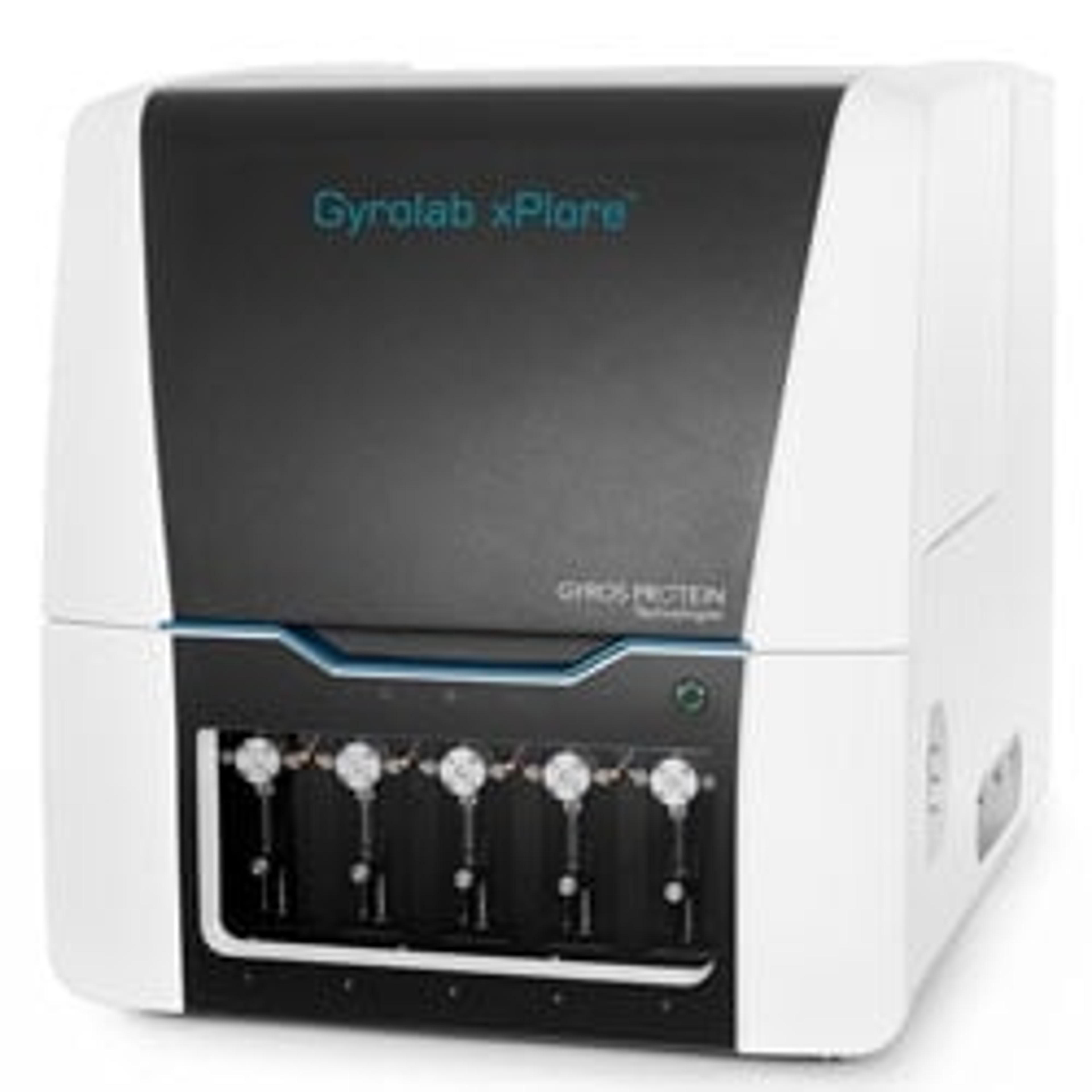
Gyrolab xPlore™ immunoassay system
Gyrolab xPlore is a single-CD, cost-effective microfluidic immunoassay platform for unattended parallel processing of 96 or 112 data points in one hour.
Automation of assay allowed by the system has increased efficiency
CHO HCP quantification kit
This system allows for this assay to be run in an automated format. This has increased efficiency by minimising user intervention and required sample volume without sacrificing data quality. It’s been a great asset to our lab!
Review Date: 7 Feb 2018 | Gyros Protein Technologies
Key Features:
- Generates up to 112 high-quality immunoassay data points per CD within an hour
- Samples are processed in parallel
- Software 21 CFR part 11 compliant
- Backed by industry-leading technical support
- Nanoliter-scale, rapid analysis, and wide dynamic range
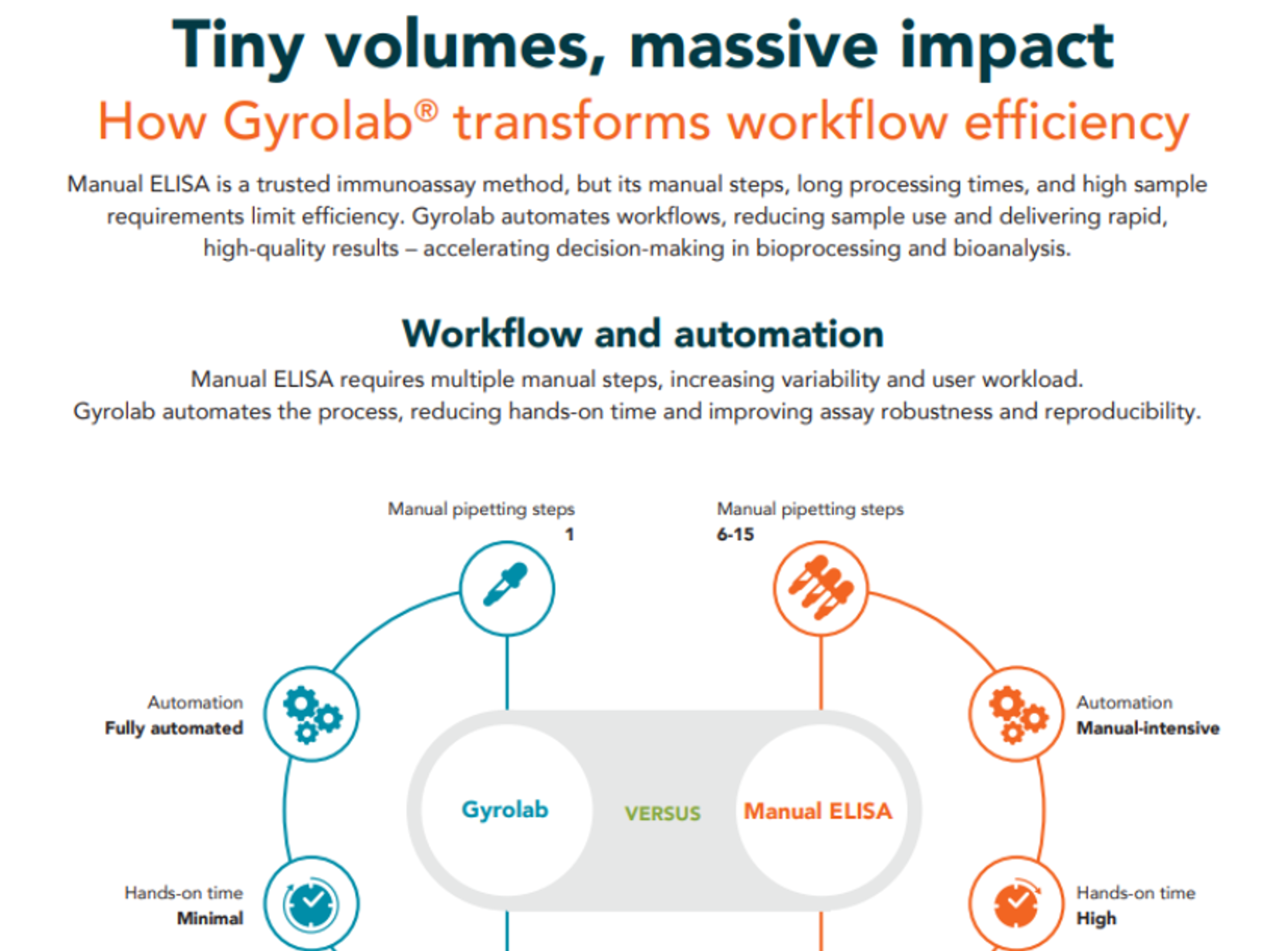
How Gyrolab transforms workflow efficiency
Discover how the Gyrolab® transforms immunoassay workflows by automating ELISA, cutting sample and reagent usage, and delivering results up to four times faster, boosting productivity and data quality in bioprocessing and bioanalysis.
Increasing productivity in cell and gene therapy bioprocessing with fast and reliable immunoassays
In this white paper, Gyros Protein Technologies discusses the crucial role of immunoassay platforms in ensuring the timely release of safe and effective cell and gene therapeutics. Choosing the right platform involves considering various factors, including robustness, quick data generation from limited samples, support for rapid method development, and flexibility for novel assays. Automation and walk-away systems are essential for reducing manual labor and minimizing errors, allowing personnel to focus on more critical tasks. Additionally, ready validation and software that enables 21 CFR Part 11 compliance are necessary to meet regulatory guidelines.
Accelerating COVID-19 R&D work-flows with microfluidic immunoassays
In this white paper, Gyros Protein Technologies illustrates the contribution of automated flow-through microfluidic immunoassays using Gyrolab system in generating reliable data for applications in public health and the development of vaccines and antibody-based treatments.
Microsampling for bioanalysis
The drive to generate preclinical and clinical data from smaller volumes of precious sample has led to considerable developments in sampling methods, In this white paper Gyros Protein Technologies describe the most common methods used for microsampling and their respective advantages and disadvantages, how microsampling is being used in preclinical and clinical studies as well as in diagnostics. Download the white paper below to find out a whole host of biopharmaceutical implications via microsampling.
Your guide to miniaturized immunoassays for biotherapeutics
Building on advances in microfluidics, next-generation immunoassays offer unparalleled capability in automation and miniaturization, generating robust and reproducible results for faster decision-making.
In this eBook, discover the advantages of automated, miniaturized immunoassays across the drug development pathway, from pre-clinical to bioprocessing. Learn about challenges and trends in immunoassay technology and how automated, miniaturized immunoassays can help you maximize productivity across a range of biopharmaceutical applications, including:
- Research and clinical development
- Insulin quantification
- Anti-drug antibody studies
- In-solution affinity determination
- Vaccine development and production
One-hour, microfluidic SARS-CoV-2 antibody immunoassay
In this white paper, Gyros Protein Technologies demonstrates how high-quality reagents from Sino Biological can be used in a high-performance, one-hour microfluidic, nanoliterscale immunoassay using Gyrolab® platform for the qualitative detection of total antibodies generated against the receptor binding domain (RBD) of the spike protein of SARS-CoV-2 in human serum samples.
Download it below to explore details of the miniaturized, microfluidic SARS-CoV-2 antibody immunoassay development and learn more about the future directions for Gyrolab assay development to detect SARS-CoV-2 antibodies.
TechTalk: Scaling bioanalysis through automation with expert CRO insights
Friday, March 27 at 16:00 GMT | 17:00 CET | 12:00 EDT | 09:00 PDT
As bioanalysis demands continue to rise, driven by expanding study portfolios, accelerated timelines, and fluctuating project volumes, CROs and pharmaceutical organizations are under increasing pressure to scale efficiently while maintaining regulatory‑ready data quality. Automation is becoming essential for meeting these expectations, enabling teams to increase throughput without adding operational complexity or additional headcount.
This SelectScience® TechTalk brings together valuable CRO insight from an expert working at the forefront of modern bioanalysis: Jason McGraw, Application Scientist, Charles River Laboratories. He will discuss how automated Gyrolab® immunoassay workflows are being implemented to boost productivity, reduce hands‑on time, and deliver consistent pharmacokinetics (PK) data across diverse study types.
In just 20 minutes, attendees will gain practical guidance on how automation is reshaping bioanalytical operations, supporting flexible scaling and helping teams meet rising regulatory expectations in today’s demanding scientific environment.
Key learning objectives:
- Gain insight into real-world implementation of Gyrolab immunoassay systems in a CRO environment.
- Understand how automated Gyrolab workflows support high-throughput bioanalysis without increasing operational complexity.
- Learn how CROs manage variable study volumes and aggressive timelines using standardized, automated assays.
- Explore strategies to reduce hands-on time and analyst variability while maintaining data quality and reproducibility.
- Identify opportunities to improve resource efficiency and turnaround times in PK bioanalysis.
Who should attend?
- Bioanalytical and DMPK leadership, including heads of bioanalysis/PK/DMPK, CRO scientific and operational directors, and laboratory leads in pharma and CROs
- Hands‑on bioanalytical innovators, including bioanalytical scientists, assay development and method transfer teams, laboratory automation leaders, and project managers overseeing bioanalytical timelines
Certificate of attendance
If you attend the live TechTalk, you will automatically receive a certificate of attendance, including a learning outcomes summary, for continuing education purposes.
If you view the on-demand TechTalk, you can request a certificate of attendance by emailing editor@selectscience.net.
TechTalk details
- Cost: Free to attend
- Location: Online
- Duration: 20 minutes
Registration is required to secure your place. If you register but can’t attend live, you will receive a link to the on‑demand recording once it becomes available
Miniaturized ligand binding assays enable scientists to do more with less
Gyros Protein Technologies is driven by a clear mission: to accelerate drug development by miniaturizing ligand binding assays so scientists can do more with less. This focus on productivity and efficiency underpins the development of tools that help researchers overcome challenges such as limited sample volumes and tight timelines. The GyroLab xPlore™ exemplifies this approach, delivering an end‑to‑end workflow in an hour. In this SelectScience interview, hear from Senior Field Application Scientist Eric Feely, as he discusses how the team continues to advance automation technologies and collaborate with automation groups to further streamline and accelerate laboratory workflows.
This video was filmed at SLAS 2026.
Anti-AAV antibody testing emerges as key to gene therapy
Advance gene therapy precision by reliably testing pre-existing AAV immunogenicity
Taking the difficulties out of assay development
Find out how Custom Assay Services from Gyros Protein Technologies accelerate biotherapeutic development
Trending in biopharma: From developing vaccines for pandemics, to overcoming omic workflow bottlenecks
Watch exclusive interviews with industry experts on the key to vaccine development success, the role of biomaterials in creating drug models, and more
5 essential resources to streamline your drug discovery and development processes
Explore the latest application eBooks with top tips on biopharmaceutical characterization and high-content screening
Accurate ligand high-affinity determination
Johan Engström, Senior Scientist at Gyros Protein Technologies, discusses the challenges of measuring high-affinity interactions of ligands in cancer immunotherapy
Gene therapy: Reliable capsid titer quantification for AAV-based therapeutics
Guest editorial: Technology to meet the challenge of efficiency in the regulated AAV vector space
New ultra-sensitive, automated immunoassays to streamline biopharma clinical assay development
EMD Serono senior scientist Carina Carter shares her experience with some of the latest immunoassay technology optimizing pharmacokinetics and immunogenicity assay development
Boosting vaccine discovery & development in a post-pandemic era with fast, high-performance immunoassays
Guest editorial: Technology to meet the needs of faster vaccine development