ACD/AutoChrom
Method development software for the chromatography expert. ACD/AutoChrom helps experts develop chromatographic separations using QbD principles. It uses a workflow-based structure to streamline and track a project.
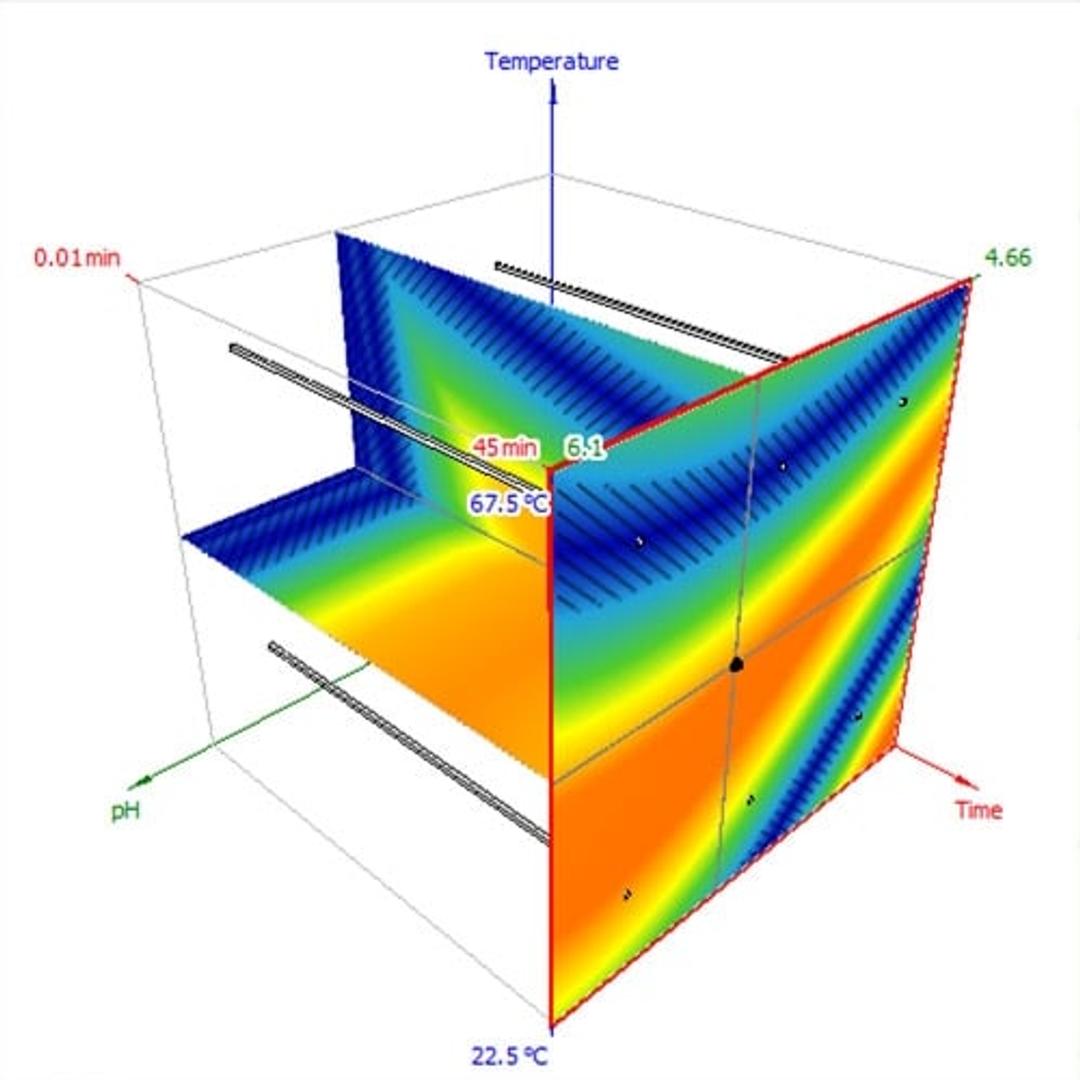
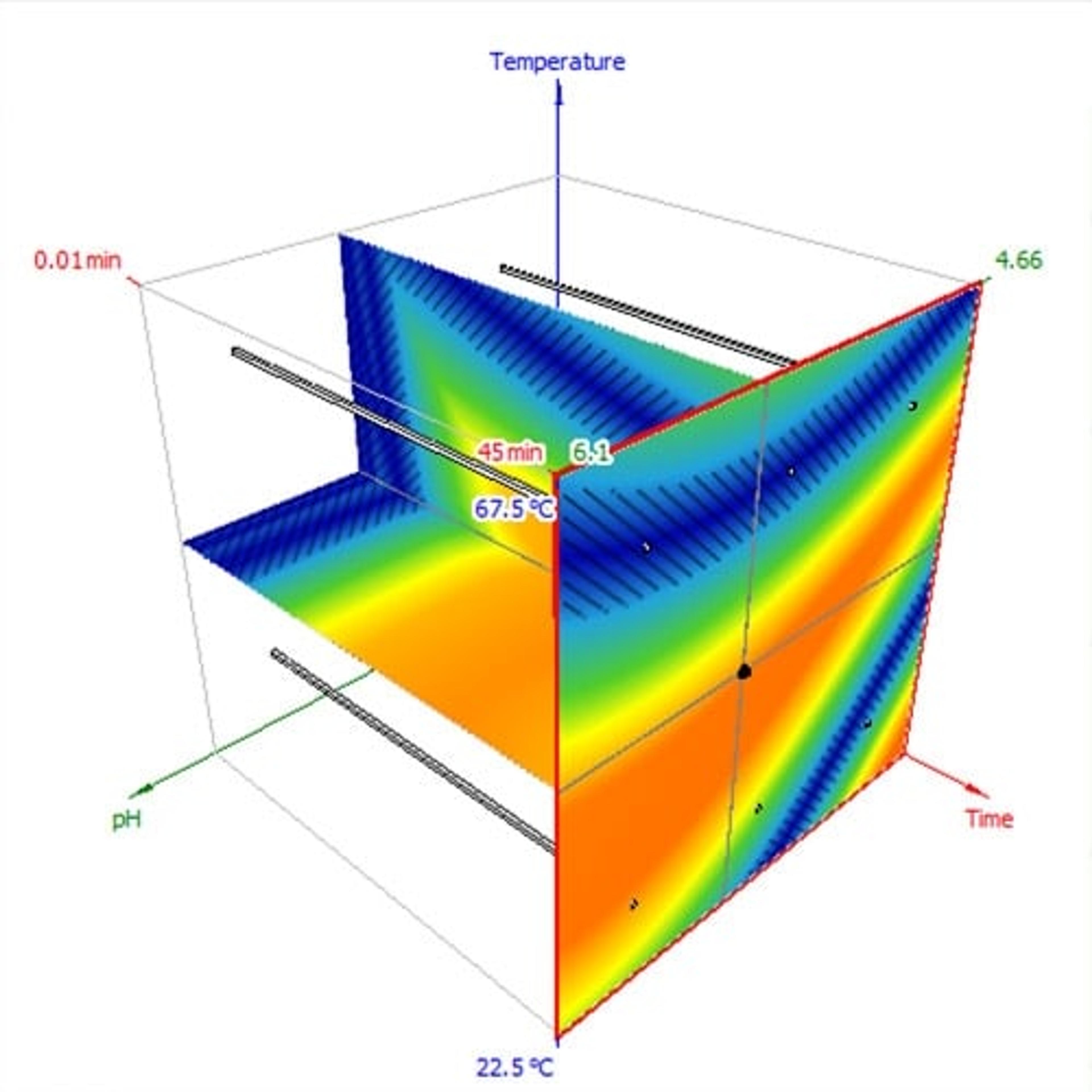
Avoid the uncertainty of undirected, one-trial-at-a-time method development. By making a plan from the beginning, you reduce the chances of wandering the design space aimlessly and missing optima. Software can help by connecting your development strategy to actual experiments, then directly running those experiments and turning the data into a model for optimization. The project-management features will also help you document your choices for data integrity and justifiable decisions.
AutoChrom allows you to:
- Begin optimization with reasonable starting conditions and columns
- Define a strategy and implement it with help from software wizards and assistants
- Automatically run experiments for screening and optimization
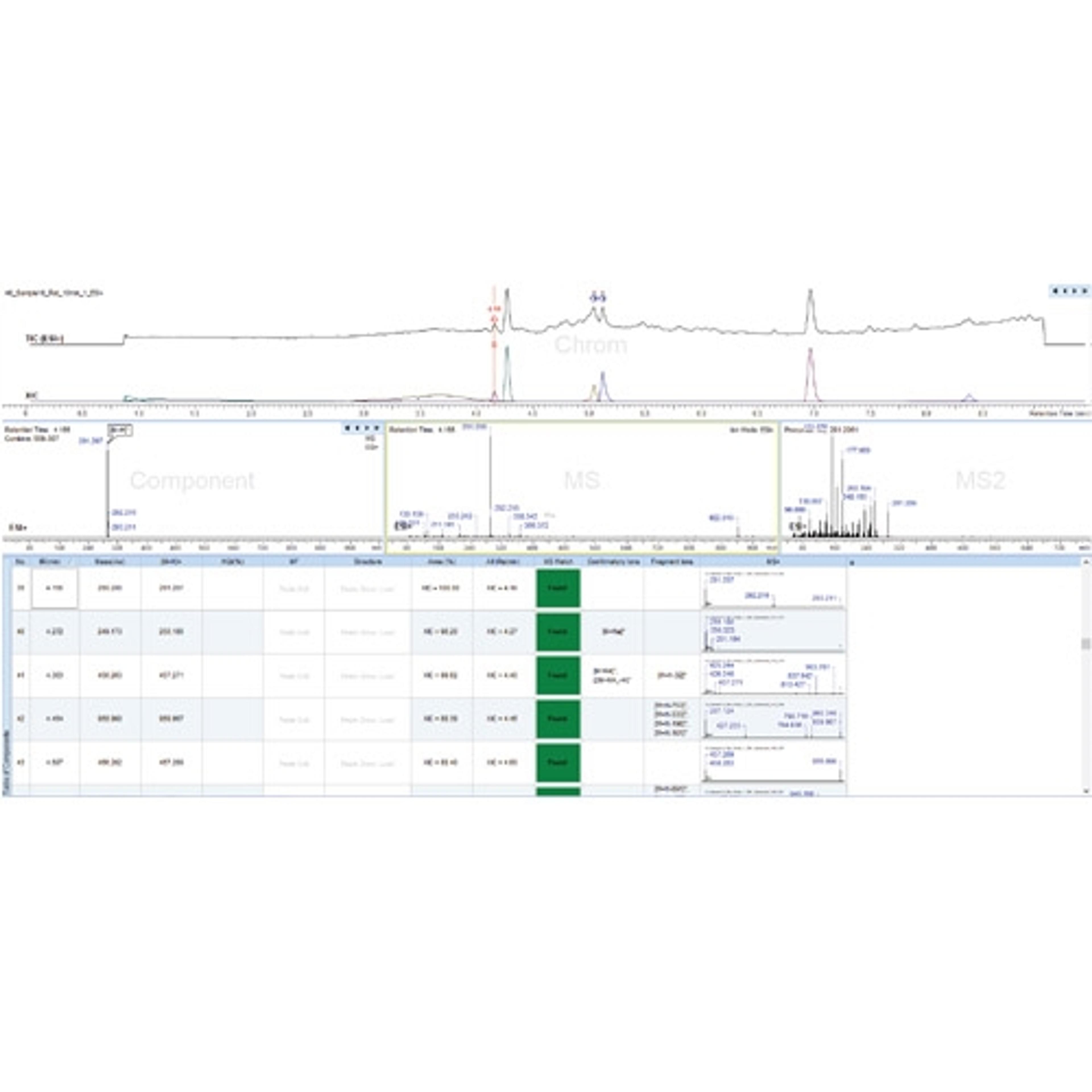
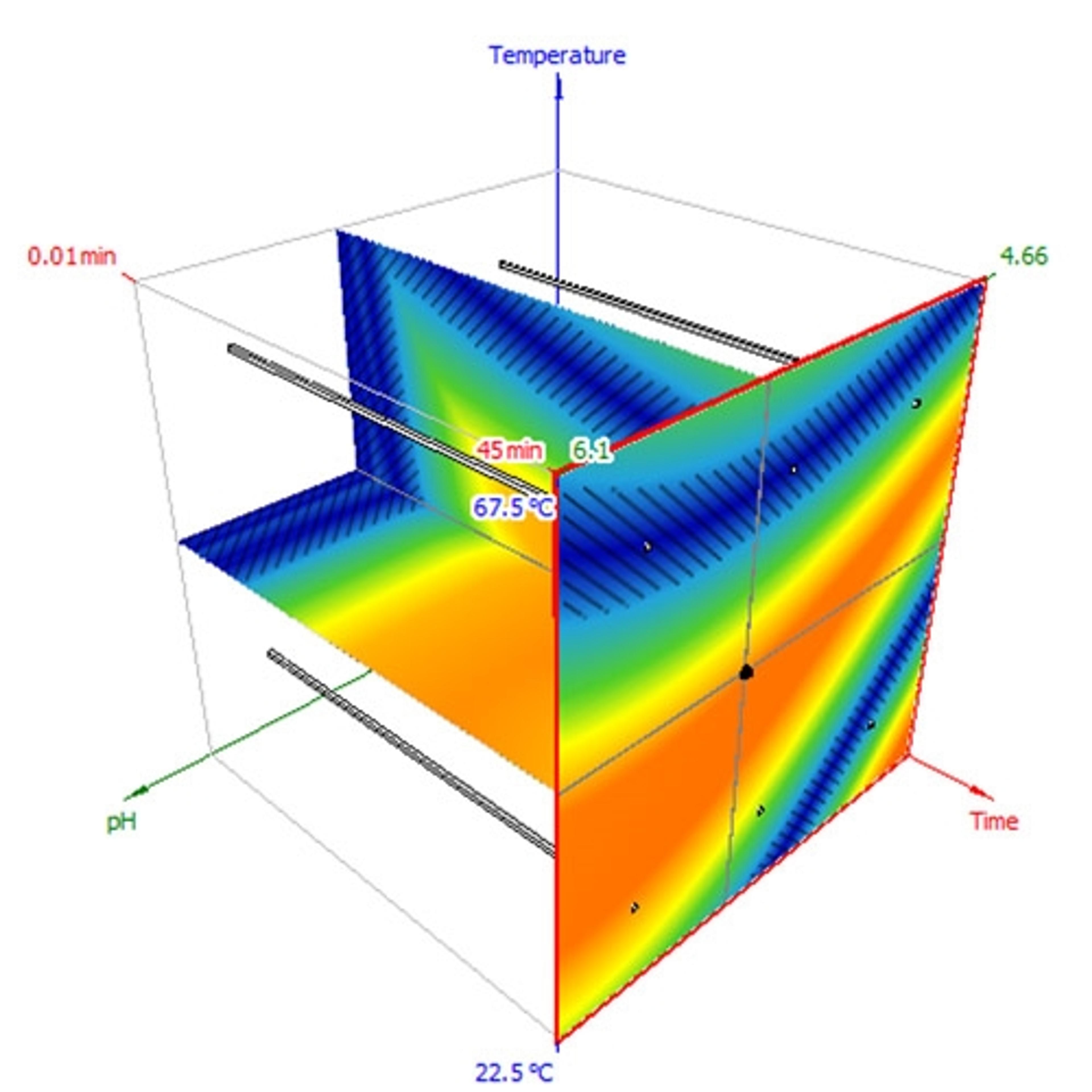
- Model your separation in 1D, 2D, or 3D
- Document the reasons for your decisions and database your projects for future learning
Brochures
AutoChrom: Automate method development & achieve QbD
In this flyer, ACD/Labs demonstrates AutoChrom®, the comprehensive software package for chromatographic method development (screening, optimization, robustness) and project management.
A Systematic Approach to the Development of Impurity Separation Methods for Drug Development
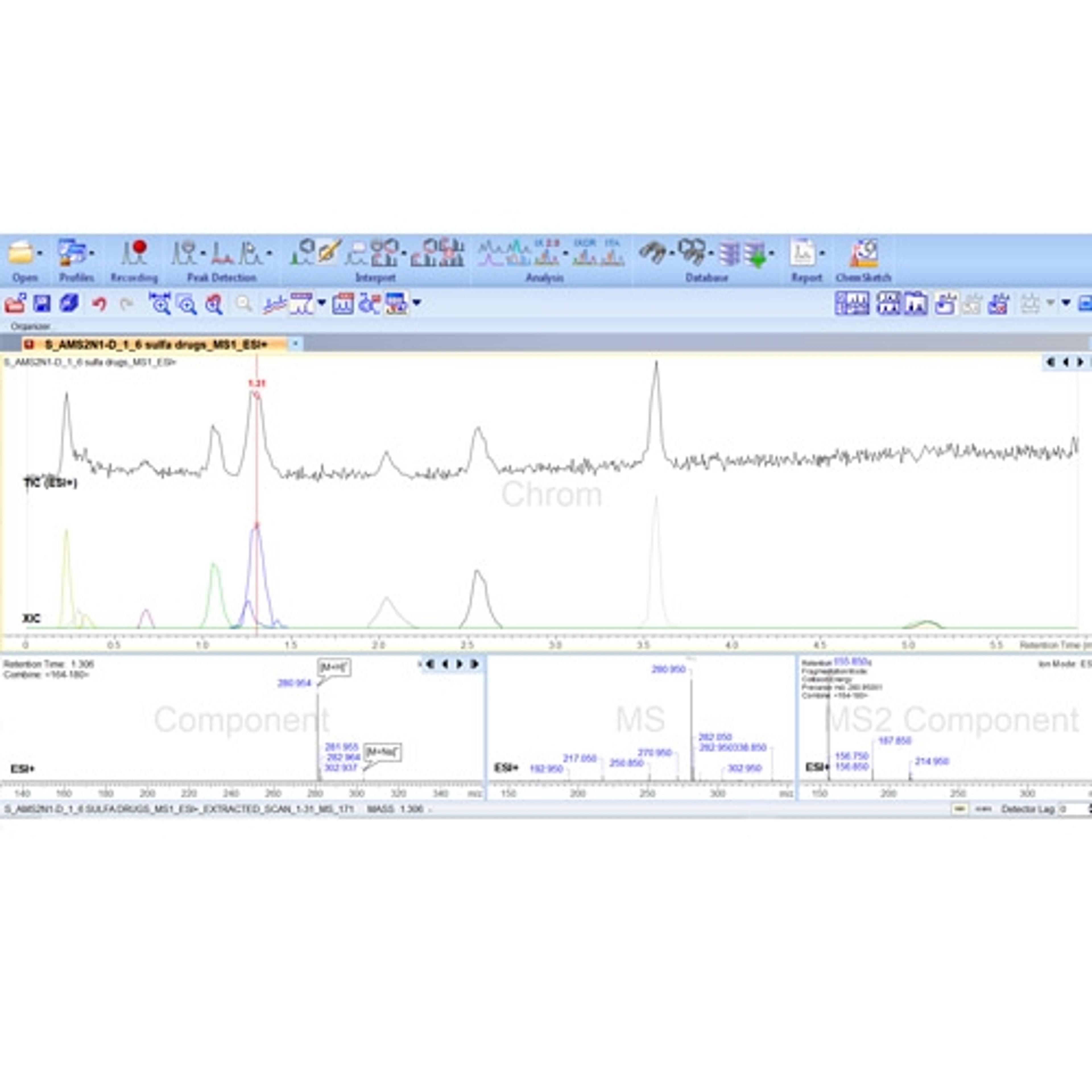
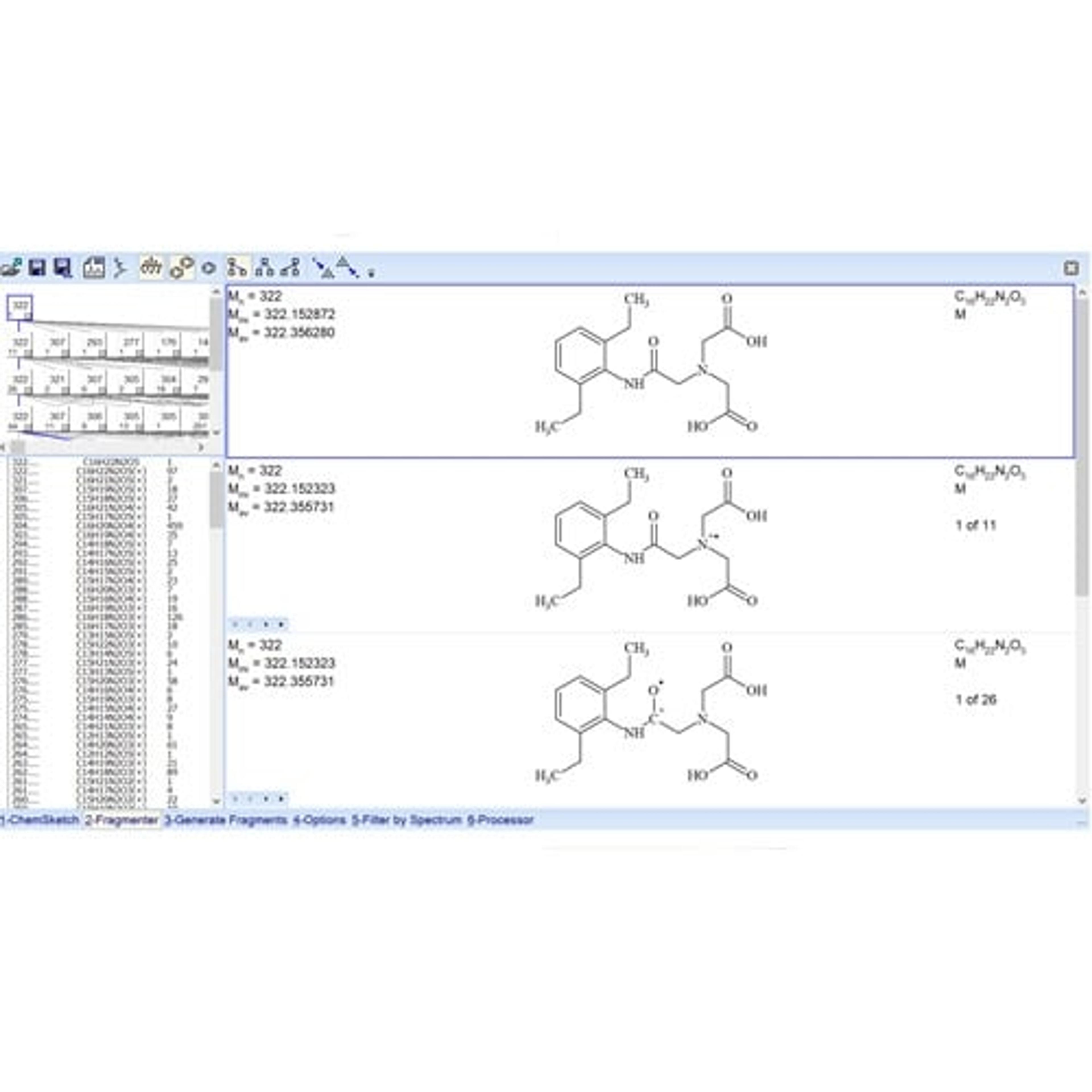
Unified Laboratory Intelligence (ULI) is a new category of laboratory R&D informatics that provides a technology framework to improve chemical identification, characterization, and optimization. This application note outlines how ULI principles embodied into the ACD/Spectrus Platform are applied to enable active development and management of chromatographic methods and data used for impurity resolution.
ACD/Labs Releases Updates to its Foundational Spectrus and Percepta Platforms
The release of ACD/Labs’ v2018.1 software includes major enhancements to its analytical knowledge management solutions for pharmaceutical development, MS and NMR dereplication workflows, and chromatography offerings