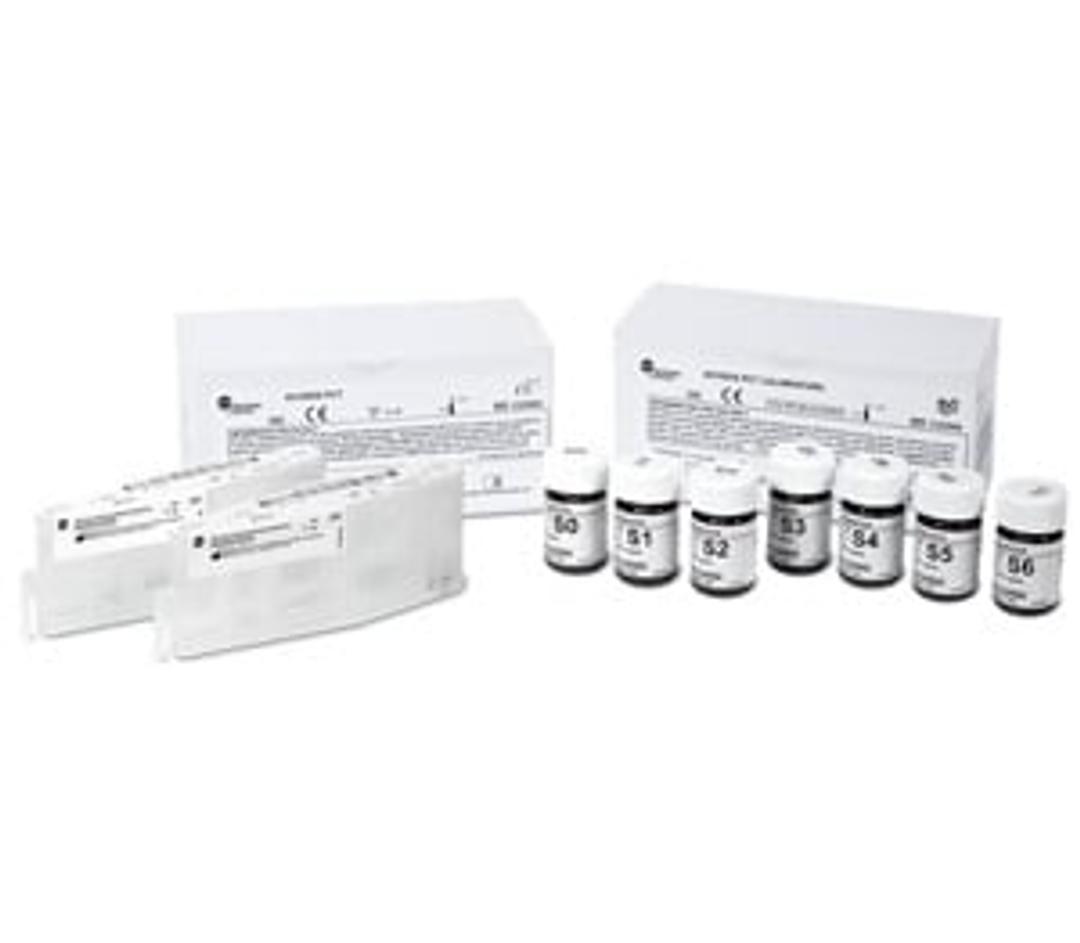
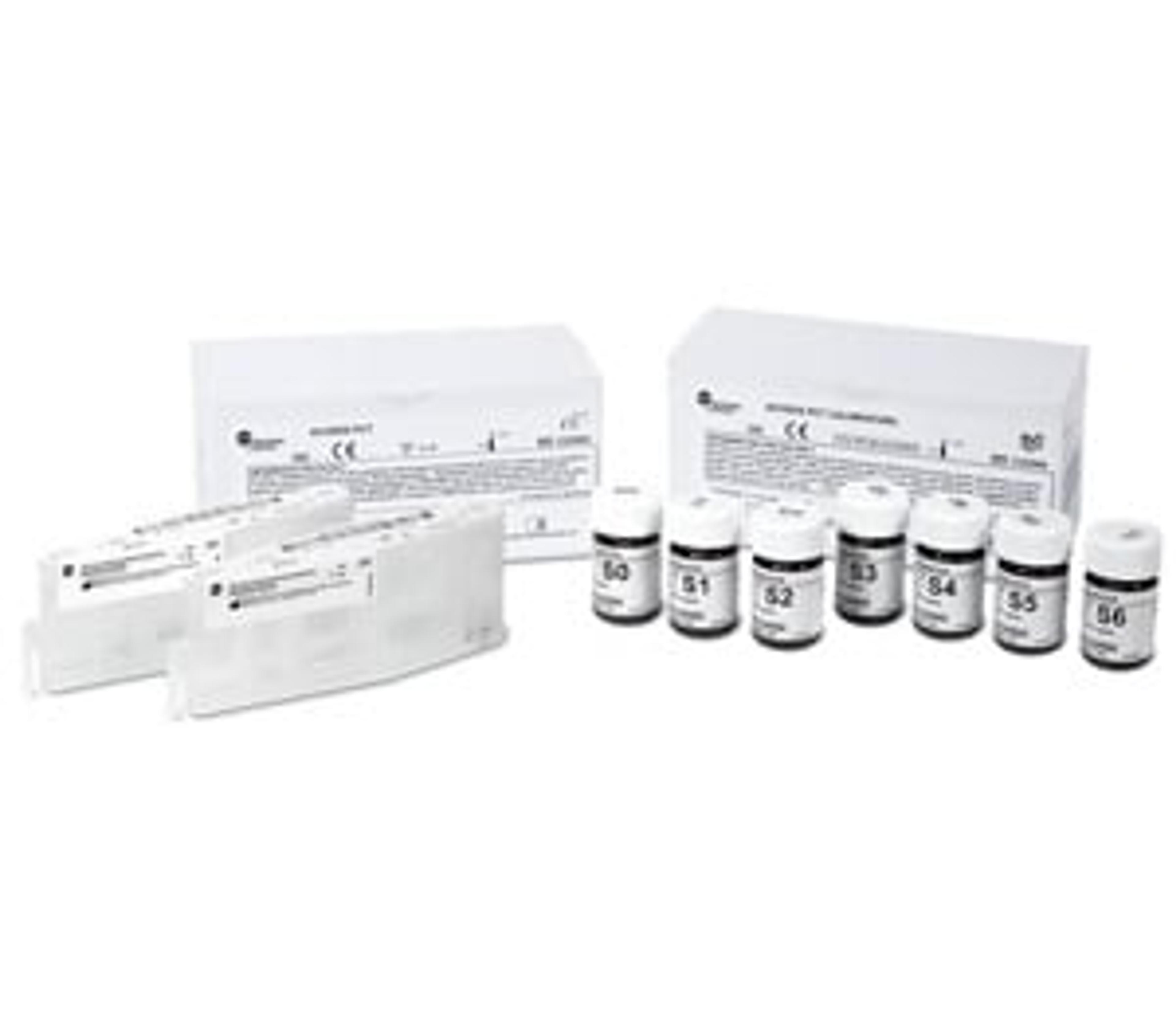
Access PCT
Timely, accurate Procalcitonin test results. Now CLIA categorized.
The Procalcitonin* (PCT) assay is intended for the quantitative determination of PCT in human serum, EDTA or lithium heparin plasma. Measurement of procalcitonin in conjunction with other laboratory findings and clinical assessments aids in the risk assessment of critically ill patients on their first day of ICU admission for progression to severe sepsis and septic shock. The PCT assay has been FDA CLIA categorized as Moderate Complexity for use on Beckman Coulter AU480, AU680, AU5800 and DxC 700 AU clinical chemistry analyzers.
MIS-C detection: Beckman Coulter partners with BARDA in rapid diagnostic for pediatric complication of SARS-CoV-2
The partnership aims to expand existing Monocyte Distribution Width (MDW) hematology-based biomarker to potentially aid in the rapid detection of MIS-C
Beckman Coulter completes first in a series of antibody neutralization studies
Access SARS-CoV-2 IgG assay shows excellent agreement with the Genscript Surrogate Virus Neutralization Test