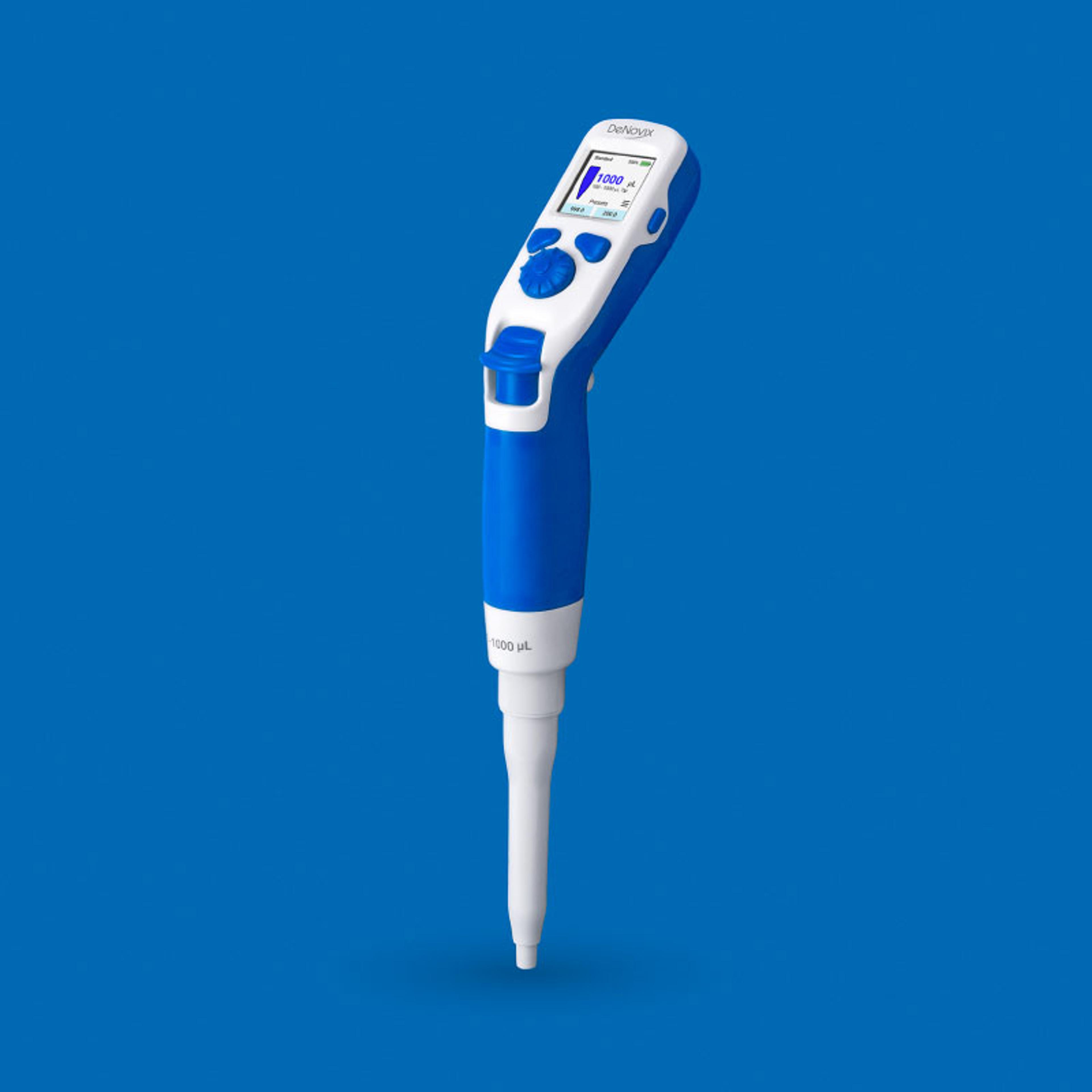
Thermo Scientific™ CryoMed™ Controlled-Rate Freezer with OPC UA, General Purpose
For customers in cell and gene therapy and vaccine production, the CryoMed Controlled-Rate Freezer (CRF) with OPC UA provides precise, repeatable freezing results that protect samples from intracellular freezing. The CryoMed CRF features OPC UA serial communication capabilities, with standard factory certifications included. In addition the unit provides enhanced data traceability via a touch screen user interface and offers c…

The supplier does not provide quotations for this product through SelectScience. You can search for similar products in our Product Directory.
Great results
Cell storage
It is very easy to use, maintains the correct temperature, the service personnel is very careful and checks the device every 6 months, it was not very expensive and it is a must for freezing biological samples for research/clinical use.
Review Date: 12 Jul 2016 | Thermo Fisher Scientific
Bad results, I suggest not to buy Freezers from Thermo Fisher.
Storage of samples (oncology)
We bought -80C and -140C Freezers from Thermo Fisher in 2017. However, all of them were sent back for fixing in 2020. What's worse, the -140C has crashed down twice. It's really terrible. In my 4 Labs experience, the quality of the -80C freezers we've bought in the last 8 years is bad.
Review Date: 11 Jul 2016 | Thermo Fisher Scientific
For customers in cell and gene therapy and vaccine production, the CryoMed Controlled-Rate Freezer (CRF) with OPC UA provides precise, repeatable freezing results that protect samples from intracellular freezing. The CryoMed CRF features OPC UA serial communication capabilities, with standard factory certifications included. In addition the unit provides enhanced data traceability via a touch screen user interface and offers customizable freezing profiles while supporting 21 CFR part 11 and GMP needs.
User-friendly operation with enhanced data traceability
- Intuitive touchscreen display allows for easy set-up, operation, and review of a freezing run
- OPC UA (ethernet) serial communication protocol
- Factory-issued certificates
- Six pre-set freezing profiles and space for up to 14 user-defined, “custom” freeze profiles
- Integrated touchscreen UI logs usage and events to support 21 CFR part 11 requirements
- USB data export of .pdf file run logs support 21 CFR part 11
- User security: 3 levels of user accessibility
- “Run last” feature allows the same profile to be run consecutively with the touch of a button
- PC interface software provides remote operation capabilities, run review, and custom profile creation
Real-time run monitoring for sample protection
- Chamber and sample temperatures are monitored by Type T thermocouples, eliminating lag time and providing “real time” responsiveness
- Standard alarms to alert users of thermocouple failures, heater malfunction, high/low temperature limits, temperature tracking, power failure, and completion of run
- Remote alarm contacts
- Optional thermal printer
Reliable temperature performance for high throughput usage
- Dual solenoid valves are designed to balance LN2 injection volume for precise temperature control and accelerated freezing
- Preventative maintenance indicator for LN2 solenoid replacement limits downtime
- Temperature control and uniformity achieved via an air-handling and liquid nitrogen injection ring system
- Compatible with VHP cleaning techniques
- Environmentally friendly insulating foam
- Type 304 stainless steel with exterior powder-coat finish
The CryoMed CRF with OPC UA is available in three sizes – 17L, 34L and 48L which can be purchased with and without a printer.
How to successfully operate a reliable and high-quality specimen biobank
Biobanks are the backbone of modern clinical and basic research, providing the long-term storage and accessibility of human biospecimens required to unlock a deeper understanding of complex diseases. However, the value of a biobank is only as high as the quality of its samples. To transform raw specimens into actionable data, laboratories must implement rigorous design standards that prioritize sample integrity, security, and traceability.
This SelectScience eBook, produced in partnership with Thermo Fisher Scientific, a global leader in laboratory equipment and life sciences solutions, offers a comprehensive guide to operating a high-performance biobanking facility. From navigating the complexities of cryopreservation to meeting modern sustainability goals, this resource provides the technical framework needed to produce reliable, high-quality research outcomes.
Inside this guide, you will explore:
- Industry-standard best practices for sample preparation, high-density storage, and precision tracking.
- Actionable advice on reducing the carbon footprint of cold storage without compromising specimen safety.
- Expert tips on selecting the right equipment to maintain stable, long-term environments for sensitive biospecimens.
- How to meet the rising demands for data transparency and chain-of-custody in modern biorepositories.
- Navigating the regulatory and ethical landscape of human specimen handling.
Whether you are designing a new facility from the ground up or refining your existing protocols, this eBook delivers the tools to help your biobank thrive in a competitive research landscape.
Download the resource now to ensure your biobank is a strategic asset for the future of medicine.
Biobank storage temperatures: An illustrative guide
In this illustrative guide from Thermo Fisher Scientific, explore the optimal temperatures for biobank storage. This infographic provides key information about the best temperature for your sample and the products you can use to ensure the preservation of your sample.
Is this Europe’s most modern biobank?
In this case study, explore how the University of Szeged, Hungary, is focused on collecting and storing biological samples for educational purposes as well as for clinical, biomedical, and physiological research.
Designing effective contamination prevention strategy: Crucial factors to consider for cell therapy production
In advanced therapy medicinal products (ATMP) manufacturing, the quality and safety of the product needs to be ensured from the first stages of development. Discover how to design an effective contamination prevention strategy that will minimize risks to cell therapy products and allow you to implement appropriate control measures.
The webinar will outline the types and sources of particulates in a cell therapy production environment and their impact on product quality and safety. It will also explore the role of laboratory equipment in contamination prevention, and provide clarity around regulatory requirements, validation methodology, and the validation process for laboratory equipment.
Key learning objectives
- GMP guidance for ATMP – how to identify risks to cell therapy products and implement appropriate control measures
- The role of laboratory equipment in contamination prevention and what to look for when sourcing suitable equipment
- Types and sources of particulates in the cell therapy production environment and their impact on product quality and safety
- Regulatory requirements and validation methodology for particle emission testing
- The validation process and compatibility of lab equipment with disinfection protocols
- How to evaluate effectiveness of lab equipment, including built-in features designed to control contamination
- How to determine lab equipment suitability for GMP cleanroom environments
Who should attend?
Lab scientists, lead scientists, R&D scientists, principal investigators, engineers and lab or facility managers involved in the cell therapy development journey starting from R&D and pre-clinical / clinical testing (translational institutes, medical institutes, academic incubators, GMP production facilities) through to the later stages of cell therapy development, such as commercialisation / scale up and QC testing, usually taking place in Biotech, Biopharma, Contract Research Organisations (CRO), Contract Manufacturing Organisations (CMO) & Contract Development and Manufacturing Organizations (CDMO).
This webinar will be of interest to principal investigators, lead scientists, research scientists, lab managers, facility managers, process engineers, and others working in this field.
Certificate of attendance
All webinar participants can request a certificate of attendance, including a learning outcomes summary, for continuing education purposes.
Thermo Fisher Scientific biobanking solution
In this video, Thermo Fisher Scientific shares its biobanking sample collection, preparation and automation equipment. Biobanking services give you the power to secure and preserve your samples, track you inventory, streamline productivity, and meet your toughest laboratory challenges.
Thermo Fisher Scientific unveils first off-the-shelf validated software solution to automate cell therapy manufacturing workflows
Gibco™ CTS Cellmation™ Software digitally connects instruments from across Thermo Fisher’s cell therapy portfolio to streamline and automate manufacturing
Meeting good manufacturing practice guidelines for cell and gene therapy products: Why your equipment matters
Discover the most common challenges faced when implementing new equipment in your manufacturing workflow and what to look for when choosing new equipment