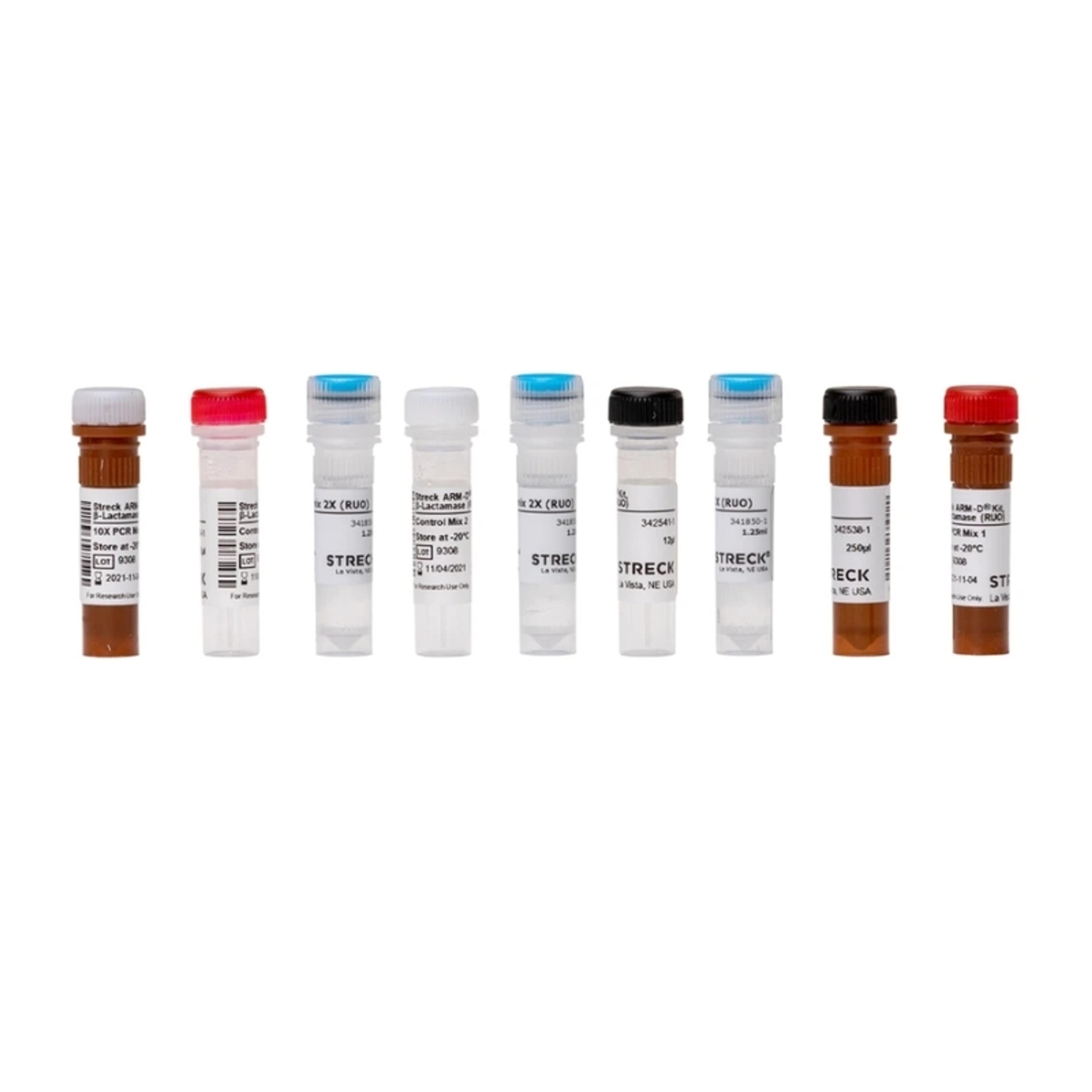
MDx-Chex® for BCID2
MDx-Chex for BCID2 is a first-of-its-kind quality control designed to evaluate the entire analytical process of the BIOFIRE ® Blood Culture Identification 2 (BCID2) Panel for sepsis. With MDx-Chex for BCID2, you can track lot-to-lot performance of the BCID2 assay and reduce the occurrence of incorrect results due to instrument or assay failures.
MDx-Chex for BCID2 is a patient-like full process control designed to validate the entire analytical process of the BIOFIRE BCID2 Panel, including cell lysis, DNA extraction, purification and removal of PCR inhibitors in addition to qPCR amplification, detection and analysis. MDx-Chex for BCID2 covers all targets tested on the BIOFIRE BCID2 Panel: 43 bacteria, yeasts and antimicrobial resistance gene targets packaged in two separate vials, one for Gram (-) bacteria and one for the Gram (+) bacteria and yeasts. Microorganisms are intact, inactivated and suspended in a matrix of stabilized red blood cells, white blood cells and blood culture media components, providing you with a control that can be mixed and pipetted like a positive patient sample, reducing the risk of cross-contamination, pipetting errors or other pre-analytical variables that may cause incorrect results.
Features
- Patient-like matrix of inactivated microorganisms, blood cells and culture media components
- Can be directly used on the BIOFIRE FilmArray® Torch and 2.0 platforms
- Kit of two vials that provide complete coverage of all 43 BIOFIRE BCID2 Panel targets
Benefits
- Challenges and verifies the entire analytical process of the BIOFIRE BCID2 Panel assay
- Reduces preanalytical variables such as pipetting errors and cross-contamination
- Easy to use - simply pipette like a patient sample
Brochures
MDx-Chex quality controls for sepsis testing
In this brochure, discover how you can simplify your sepsis testing quality control and reduce bench time with MDx-Chex® controls from Streck
MDx-Chex controls for molecular-based testing
The FDA-cleared, class II assayed, patient-like full process controls, MDx-Chex™, contain intact, inactivated microorganisms suspended in a matrix of blood culture media components. The controls aim to address the gap in coverage left by traditional quality controls and ensure reliable and accurate sepsis testing. Streck discusses the development of MDx-Chex controls for use in automated DNA-based sepsis tests, and highlights data on the precision and reproducibility of these controls across multiple sites and systems. Overall, MDx-Chex controls provide a more comprehensive measure of assay performance than traditional controls and help maintain assay quality and compliance with accrediting standards and guidelines.
Tampa General Hospital’s journey to sepsis QC excellence
May 28, 2025 - 16:00 BST / 17:00 CEST / 11:00 EDT / 08:00 PDT
Join Dr. Luciano Soares, Clinical Scientist at Tampa General Hospital, as he shares how his lab seamlessly transitioned to MDx-Chex from their previous quality control (QC) method, and learn about the practical insights and lessons he gained along the way. Streck's innovative MDx-Chex® controls were meticulously designed to verify every step of the BIOFIRE® BCID2 and Luminex VERIGENE® BC-GP and BC-GN sepsis tests, ensuring comprehensive coverage of every detectable target. Dr. Soares will highlight how these features have greatly improved his lab's accuracy and reliability.
Don't miss this opportunity to elevate your lab's diagnostic capabilities.
Key learning objectives:
- Understand the key differences between an external quality control (EQC) and an internal quality control (IQC)
- Learn about the ideal composition of an EQC with regards to micro recommendations (IQCP)
- Discover how an EQC with the recommended composition of intact microorganisms in a patient sample-like matrix can detect issues with assay reagents that nucleic acid-based QC cannot
Who should attend?
- Clinical microbiology lab managers/directors
- Quality management
- Infectious disease clinicians
Certificate of attendance
All webinar participants can request a certificate of attendance, including a learning outcomes summary, for continuing education purposes.
If you view the on-demand webinar, you can request a certificate of attendance by emailing editor@selectscience.net.
Molecular diagnostics at a crossroads with future insights from the experts
Molecular diagnostics is reshaping how we detect and manage infectious diseases, with far-reaching implications for both patient care and translational research. In clinical settings, rapid molecular testing enables faster, more targeted treatment decisions that can significantly improve outcomes. In research laboratories, the same technologies are unlocking deeper insights into pathogen behavior, resistance mechanisms and emerging threats.
But with every advancement comes a set of challenges. Laboratories must weigh the benefits of adopting new technologies, like improved sensitivity, multiplexing and automation, against practical considerations such as validation complexity, workflow disruption and cost. The need for reliable, adaptable quality controls becomes even more critical as assays grow more sophisticated and regulatory demands increase.
In this expert roundtable—presented in partnership with Streck—leaders from the clinical and research community come together to share their experiences navigating this evolving landscape. They’ll discuss how molecular diagnostics is driving real-world impact, how their labs are adapting to new pressures, and how thoughtful implementation of innovation can yield both scientific and operational rewards.
Key learning objectives:
- How molecular diagnostics is transforming treatment strategies for serious infections
- Risks and rewards of adopting new molecular technologies
- How quality controls are adapting to support complex, multiplexed assays and evolving regulatory demands
- Practical steps laboratories are taking to improve efficiency, ensure reliability and prepare for what’s next
Whether you're optimizing workflows, evaluating new platforms or rethinking your QC approach, this session delivers expert perspectives to help you navigate the future of molecular diagnostics.
Certificate of attendance
If you attend the live forum, you will automatically receive a certificate of attendance, including a learning outcomes summary, for continuing education purposes. If you view the on-demand forum, you can request a certificate of attendance by emailing editor@selectscience.net.
Streck's MDx-Chex controls for sample to answer QC needs
Sample to answer molecular diagnostics instruments play a vital role in patient diagnosis. Any error can lead to significant impacts, making the use of external controls to monitor performance essential. Explore MDx-Chex® controls from Streck. Ideal for quality control needs, they provide an all-in-one solution for verifying the performance of the BIOFIRE® BCID2 and VERIGENE® BC-GP and BC-GN sepsis tests. Comprehensive and easy-to-use, MDx-Chex controls evaluate the entire assay process, from cell lysis, DNA extraction and purification to nucleic acid detection and analysis.
How external quality controls are transforming molecular diagnostics
Molecular diagnostics experts share why EQCs are no longer optional if you’re looking to elevate your molecular diagnostics
Molecular diagnostics treat infectious disease with confidence
Find out how Streck is adapting to evolving clinical lab needs by providing quality controls for automated molecular diagnostics
Experts explore how molecular diagnostics are reshaping infectious disease testing and patient care
From sepsis to syndromic panels, this SelectScience forum discussed how molecular diagnostics is setting new standards for accuracy, efficiency, and impact
Full-process, patient-like quality controls advance molecular diagnostic testing
One lab shares its experience switching to Streck’s quality control solution for molecular diagnostics testing