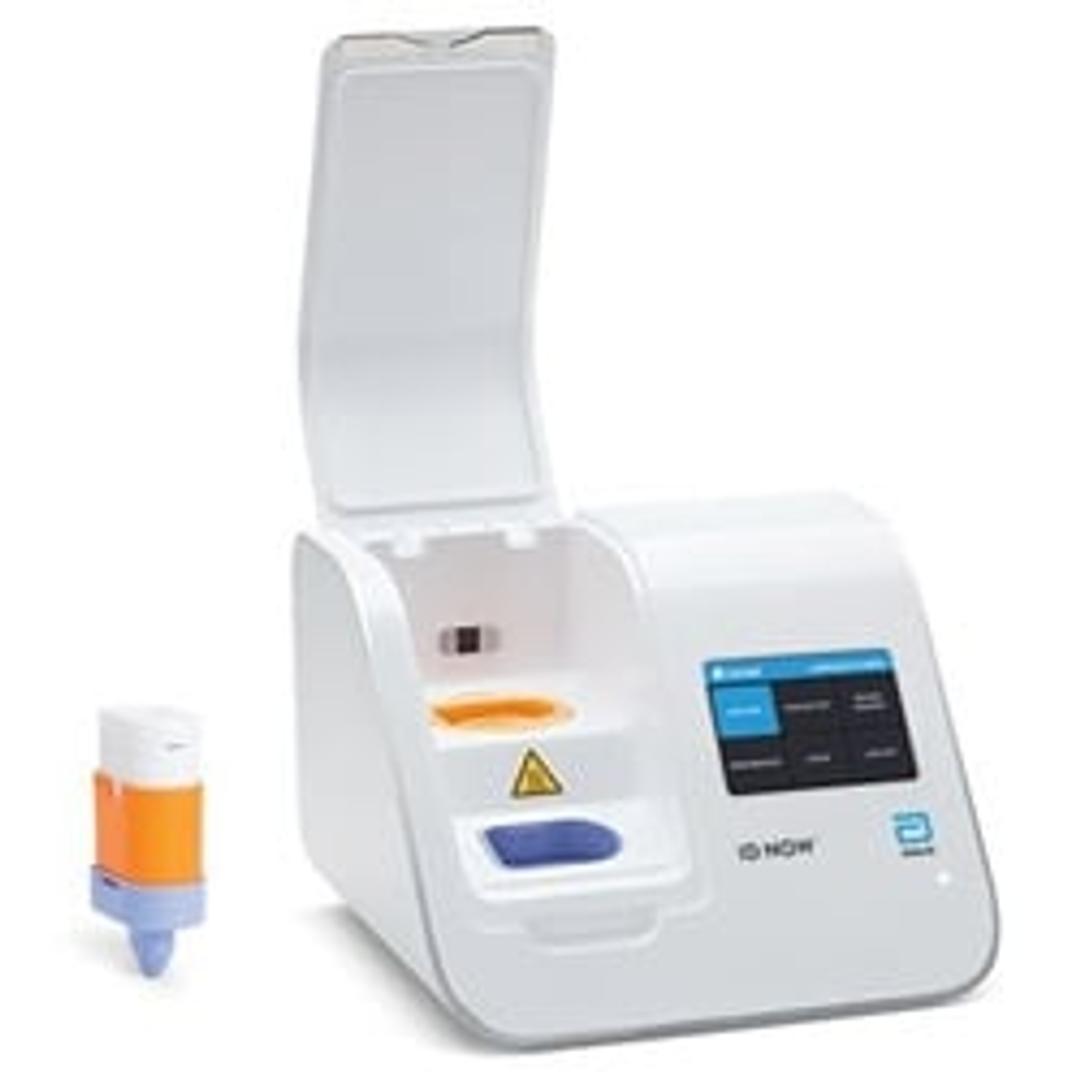
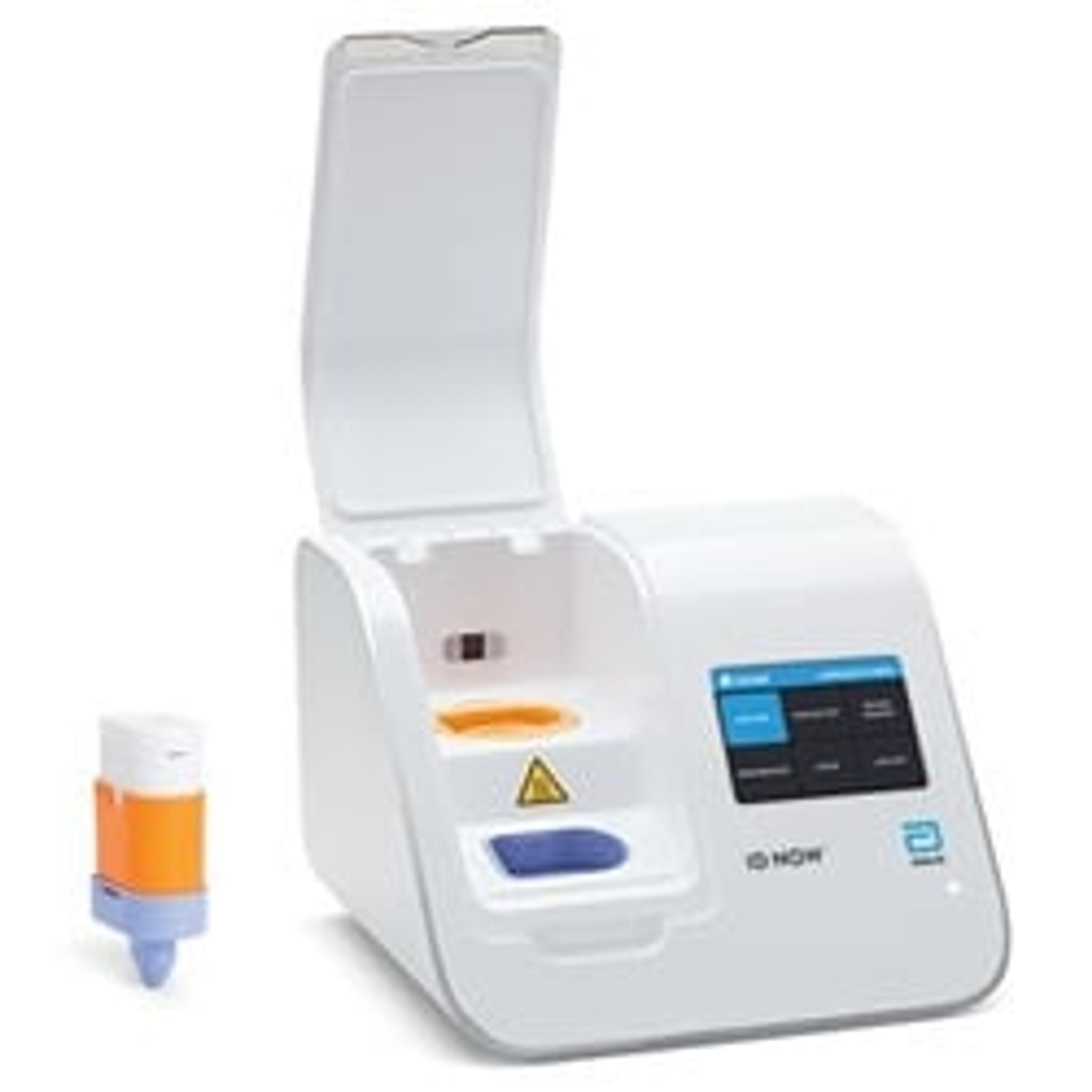
ID NOW™ Instrument (formerly Alere™ i)
A rapid, instrument-based, isothermal system for the qualitative detection of infectious diseases.

The supplier does not provide quotations for this product through SelectScience. You can search for similar products in our Product Directory.
Love it and so easy to use
Testing samples for COVID-19
We received this placement analyzer and it has been a great addition to our in house testing for COVID-19. We may look at some of the additional testing available.
Review Date: 29 Mar 2022 | Abbott
Easy to use, easy to train new operators
Point of Care Testing
Great after purchase service, high ease of use.
Review Date: 20 Dec 2021 | Abbott
Really like the analyzer. we have several.
NP
Results are good, analyzer is very easy to use, not too long on test time.
Review Date: 16 Mar 2021 | Abbott
Supplies not available or on backorder.
Clinical
Easy to use and quick. Can't get supplies, they're always on backorder.
Review Date: 16 Oct 2020 | Abbott
Perfect outcomes and easy to use it.
Clinical
Excellent tool for research and clinical application.
Review Date: 22 Jul 2020 | Abbott
Ease of use is very good.
Metabolism tissue samples
Very effective in all manners, everyone should go for this product. It's reliable and cost effective. Ease of use is very good.
Review Date: 15 May 2020 | Abbott
Easy to use, but...
COVID-19 viral testing
This little analyzer is easy to use and gives quick results, especially compared to sending the samples out for testing, but we are a little skeptical about the LOD, especially since Abbott recently sent out a notice that "Negative results should be repeated with another method."
Review Date: 15 May 2020 | Abbott
Fast and easy to use for qualitative answers.
COVID-19 nasopharyngeal swab testing
This instrument is simple to use and includes step by step instructions on the screen to walk you through each test. Results seem reproducible and accurate so far, but I have only had the instrument for a month. Great for fast individual testing. Not recommended for volume test situations.
Review Date: 15 May 2020 | Abbott
Great instrument for COVID-19 tests.
Molecular tests
Excellent platform for COVID-19 molecular tests without special qualification.
Review Date: 10 May 2020 | Abbott
Our new favorite analyzer.
Molecular diagnostics
The training was extremely helpful, we were taught the old school way buy using scenarios that stick in your mind. We were using the ID Now as a confirmation on negatives that were run on a rapid kit. We found about 35% of negatives on rapid tests were positive on the ID NOW!
Review Date: 10 Feb 2020 | Abbott
ID NOW™ is a rapid, instrument-based, isothermal system for the qualitative detection of infectious diseases. Our unique ID NOW™ isothermal nucleic acid amplification technology provides molecular results in just minutes, allowing you to make effective clinical decisions sooner. ID NOW™ is significantly faster than other molecular methods and more accurate than conventional rapid testing.
Available Tests:
ID NOW™ Influenza A&B delivers molecular flu results in less than 15 minutes on our unique ID NOW™ platform. Rapid diagnostic tests with increased sensitivity are essential for the reliable detection of influenza A and B and enable immediate, effective treatment decisions.
ID NOW™ Strep A provides molecular results in 8 minutes or less on the unique ID NOW™ platform; allowing you to confidently test, and with a positive result, treat during the patient’s visit. The Strep A Molecular Test Now CLIA-Waived.
ID NOW™ RSV delivers molecular RSV results in 13 minutes or less on our unique ID NOW™ platform. Traditional laboratory methods and rapid antigen testing for RSV diagnosis have considerable shortcomings in terms of turnaround time or performance. ID NOW™ RSV detects 25% more true positives than Rapid Antigen Detection Tests (RADTs) and allows you to make real-time clinical decisions which impact patient care.
Features & Benefits:
- Easy to use with only minimal training requirements
- Large visual touchscreen displays results, eliminating transcription errors and the need for printing
- Small footprint saves you bench space and can be used in any healthcare setting
- Offers exceptional performance
- Eliminates interpretation and transcription errors
- Gives you the confidence to make clinical decisions sooner
- Facilitates effective patient management
- Enables prompt initiation of infection control measures
- Aids targeted antiviral therapy and Antimicrobial Stewardship
- Reliable near-patient testing reduces overall healthcare costs
TO GET MORE FROM YOUR POC TESTING, CONNECT IT.
Connect More, Share More, Worry Less
A web-based, open platform, AegisPOC™ lets you better manage and share data from your POC devices on one flexible, scalable middleware. AegisPOC provides you with the ability to integrate POC devices with LIS, HIS, EMR, quality management, user management and other systems. Not available in the USA.
Fast, Accurate Detection of Influenza in the Clinical Laboratory with the Alere™ i
Dan Hanna, South Sioux City Laboratory Supervisor at Family Health Care of Siouxland, Nebraska, USA, explains how the Alere i enables his lab to accurately detect influenza in under 15 minutes, while improving the experience for both technician and patient. Hear how this system eliminates difficulties associated with traditional lateral flow assay testing, helping doctors to better care for patients.
Alere Awarded Best New Clinical Laboratory Product of 2015 Winner
KC McGrath, Respiratory Product Manager for Alere Inc. accepts the award for the Alere™ i Strep A, winner of the Best New Clinical Laboratory Product of 2015. Watch the video to learn how this product has significantly improved patient care and is redefining rapid diagnostics.
Alere Presented with Award for the Most Successful Clinical Video
Deanna Olcott, Digital Marketing Operations, Alere Inc. accepts the award for the Most Successful Clinical Video. Find out how this video has enabled scientists all around the world, to see the molecular Alere i system in use, helping healthcare professionals to improve patient care.
Lab Quality Molecular Testing Available at the Point-of-Care: Alere at AACC 2015
The Alere™ i Molecular Platform is not only incredibly easy to use, but also provides high quality, rapid molecular testing in just minutes. Watch this interview with KC McGrath, Respiratory Product Manager, Alere Inc., to find out how this state-of-the-art technology is having such a positive impact on patient care.
Abbott introduces NeuroSphere Virtual Clinic, first-of-its-kind remote neuromodulation patient-care technology in the UK
NeuroSphere Virtual Clinic utilizes advancements in digital technology to optimize care for patients with chronic pain or movement disorders
Abbott receives FDA clearance for its imaging technology using artificial intelligence for vessels in the heart
The new intuitive interface provides step-by-step guidance to enhance ease of use
Abbott’s Panbio Rapid Antigen Self-Test receives CE mark, increasing access to fast, reliable COVID-19 testing in the United Kingdom
The Panbio self-test features a minimally invasive nasal swab and delivers results in as early as 15 minutes with no instrumentation, using proven Abbott lateral flow technology
Abbott has confirmed capacity to support expanded use of HeartMate 3 heart pump
Access to mechanical circulatory support devices is critical for advanced heart failure patients and can improve survival, clinical outcomes and quality of life
Abbott begins shipping BinaxNOW COVID-19 Ag Self-Test to retailers
The BinaxNOW Self-Test will be sold in 2-count packs for an MSRP of $23.99, making it an affordable over-the-counter COVID-19 rapid test available in the U.S.
Abbott has announced its Pandemic Defense Coalition: A global network of expert collaborators designed to help prevent future pandemics
Abbott will rapidly develop tests that can be used to help identify, isolate, and contain outbreaks when potential new virus threats are identified
Abbott introduces next-generation 3D cardiac mapping platform in Europe and Australia
The EnSite X EP System is a new cardiac mapping system designed from electrophysiologist input to optimize cardiac ablation procedures
Abbott receives CE mark for MitraClip heart valve repair device to treat mitral regurgitation
CE mark for MitraClip G4 offers physicians an innovative next-generation system with more options for mitral valve repair using proven clip-based technology
Abbott's 15-minute, $5 COVID-19 antigen test receives FDA emergency use authorization
Test delivers results in just 15 minutes with no instrumentation, using proven lateral flow technology with demonstrated sensitivity of 97.1% and specificity of 98.5%
FDA approves Abbott’s iOS-compatible app for chronic pain patients
The NeuroSphere Digital Care offers flexible virtual care options as the COVID-19 global pandemic impacts how healthcare is administered