The value of mutation markers for personalized medicine in neuropathology
Discover how MilliporeSigma is expanding its portfolio of antibodies for tissue diagnostics to help better classify gliomas and other tumors of the nervous system
17 Jan 2023
Every cancer is characterized by a unique set of genetic alterations, including amplifications, rearrangements, translocations, or indels (additions or deletions). These alterations drive tumor development and can be used as markers to define the tumor type, the stage of development, and even predict the risk of metastasis and potential response to therapy. These cancer biomarkers – or mutation markers – are currently the subject of intense research as clinicians can use the valuable information they provide to help diagnose, classify tumors, and track their prognosis. In addition, these markers can inform precise therapeutic approaches that are personalized to the individual patient and tumor type.
In this article, we speak with experts at MilliporeSigma to find out how new antibodies, developed using its innovative Cell Marque™ Tissue Diagnostics technology, are helping neuropathologists to classify gliomas, brain tumors that originate from the glial cells (the supporting cells) of the central nervous system.
Our goal is to remove the guesswork and bring confidence into the diagnosis.
Brent Heller
MilliporeSigma
Bringing confidence to tissue diagnostics

“Mutation markers are a key step in the greater distinction of malignancies,” says Brent Heller, the Head of Sales for Tissue Diagnostics in the Western part of North America. However, they are not without their challenges. “At MilliporeSigma, we see the value in these unique targets, but developing accurate assays for easy use in the lab remains a challenge,” Heller explains. “You need mutation tissue to develop the assay, but you need an assay to identify the tissue, so which one comes first?”
To address this challenge, researchers need to confirm the identity of the tissue using alternative testing. One way to detect mutation markers is to sequence the DNA of a tumor sample. However, this approach is expensive and requires a relatively large amount of tissue, which is not always available, especially for tumors of the brain and spinal cord. Furthermore, this approach can also give false negative results when there are few tumor cells in the sample compared to the number of non-neoplastic cells. Immunohistochemistry (IHC) using antibodies targeting specific mutations is a convenient and cost-effective alternative screening method that provides genetic information in the context of the morphology of the tumor.
Using Cell Marque Tissue Diagnostics technology, clinicians can now perform the required experiments in a more cost- and time-effective manner for their customers. “We invest the time and energy upfront to ensure these mutation assays are specific,” says Heller. Several different approaches are used in the tissue diagnostic development process at MilliporeSigma, including sequencing tissue themselves, working with partner labs on known mutation cases, and using known cell lines with the same mutations.
A key part of the company’s approach is to custom design and produce rabbit monoclonal antibodies through a unique B-cell cloning technique. The traditional method to manufacture monoclonal antibodies is to inoculate a molecule of interest into a rabbit or a mouse. Researchers then collect and isolate the resulting antibodies and test their ability to bind to the target molecule with high affinity. However, there is a lot of variability in this process. “The animal’s immune system is going to break the target molecule down into fragments or antigens,” Heller explains. “You don't get to control that.”
Along with being an animal-free process, B cell cloning allows MilliporeSigma to take an exact input – the amino acid sequence of the mutation of interest – and produce a highly specific product. This is integral for accurate diagnostic testing. “Our goal is to remove the guesswork and bring confidence into the diagnosis,” says Heller. “B-cell cloning produces a monoclonal cell line for the stable production of the antibody and helps to ensure sustainable supply and performance”.
With traditional classification, the diagnostic practice suffered greatly from poor predictive performance and poor reproducibility.
Dr. Qin Su
MilliporeSigma
Reliable antibodies for glioma classification
Gliomas represent 30% of all tumors of the central nervous system.
Traditional classifications are based on –
Lineage:
i. astrocytic
ii. oligodendroglial
iii. oligoastrocytic
Histology:
i. grade II (benign)
ii. grade III (low-grade malignancy)
iii. grade IV (high-grade malignancy)
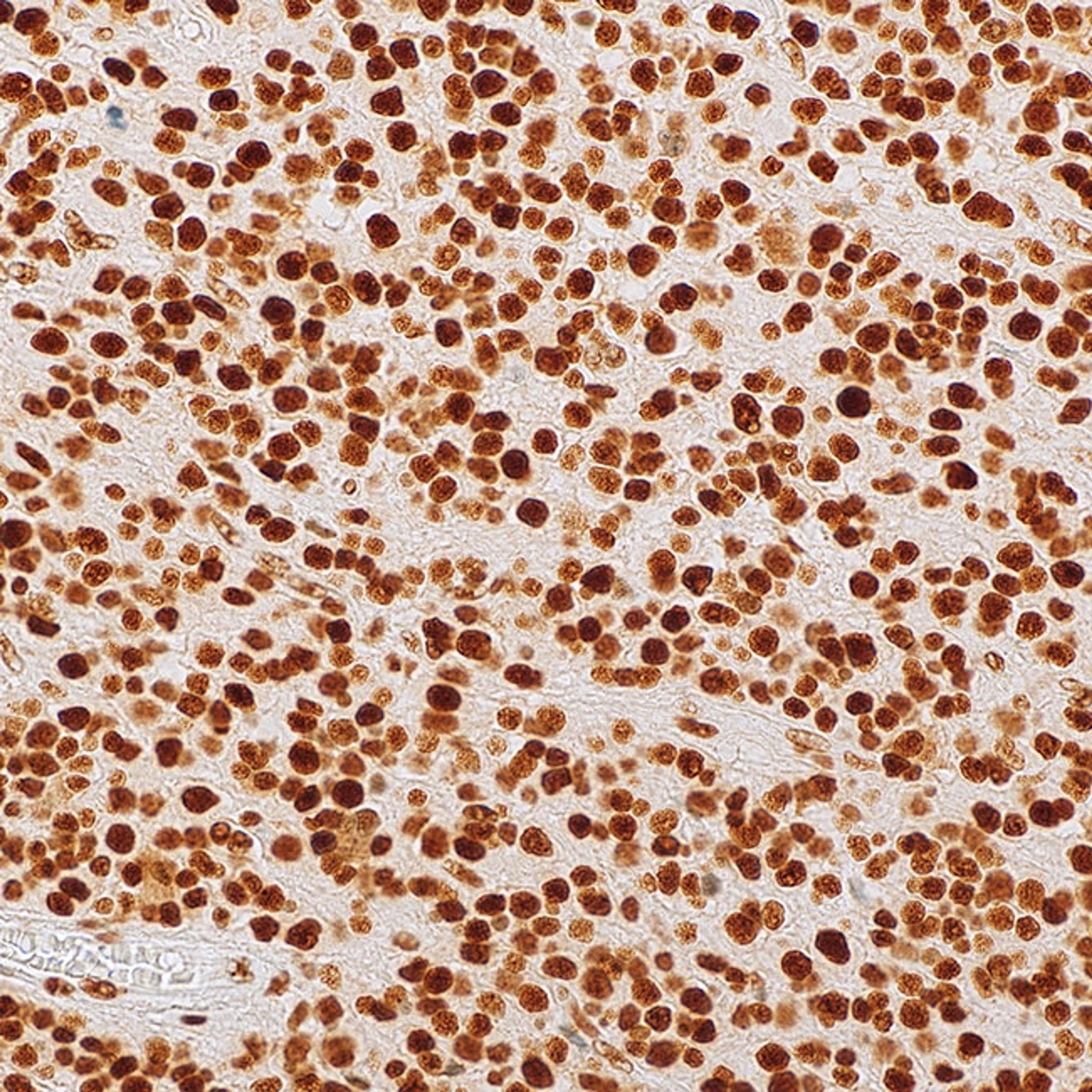
One of the team’s newest antibodies, IDH1 R132H (MRQ-67), is helping clinicians to identify and classify adult-type diffuse gliomas.
“Traditionally, diffuse gliomas were predominantly classified based on morphology – only microscopical observation – with three lineage types and three histologic grades,” explains Dr. Qin Su, a staff pathologist at MilliporeSigma. “With this classification, the diagnostic practice suffered greatly from poor predictive performance and poor reproducibility.”
However, since 2016, the WHO classification of gliomas now integrates morphology along with the status of the following key molecular alterations that are often found in these types of tumors: IDH1 mutation, ATRX presence or loss, and 1p/19q codeletion.
As an example, IDH1 encodes an enzyme called isocitrate dehydrogenase 1 (IDH1), which is part of the citric acid cycle in all cells under normal conditions. Mutations in IDH1 are common in several malignancies including diffuse and anaplastic gliomas and secondary glioblastomas. An arginine to histidine substitution at codon 132 (R132H) of this protein is found in over 90% of adult-type gliomas but is also present in some rare types of myeloid leukemia.
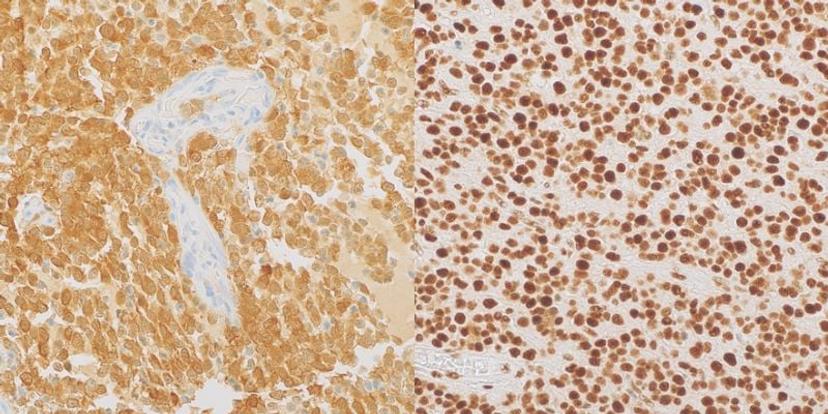
MilliporeSigma soon realized there was a gap in the market for in vitro diagnostic antibodies for gliomas and used its B cell cloning technology to develop a monoclonal antibody (dubbed MRQ-67) to target the R132H mutation of IDH1. This antibody has been shown to specifically recognize the mutation in both adult-type gliomas and myeloid leukemia. Furthermore, MRQ-67 outperformed a widely available mouse monoclonal antibody that also targets the same mutation. Download the comparative study >>
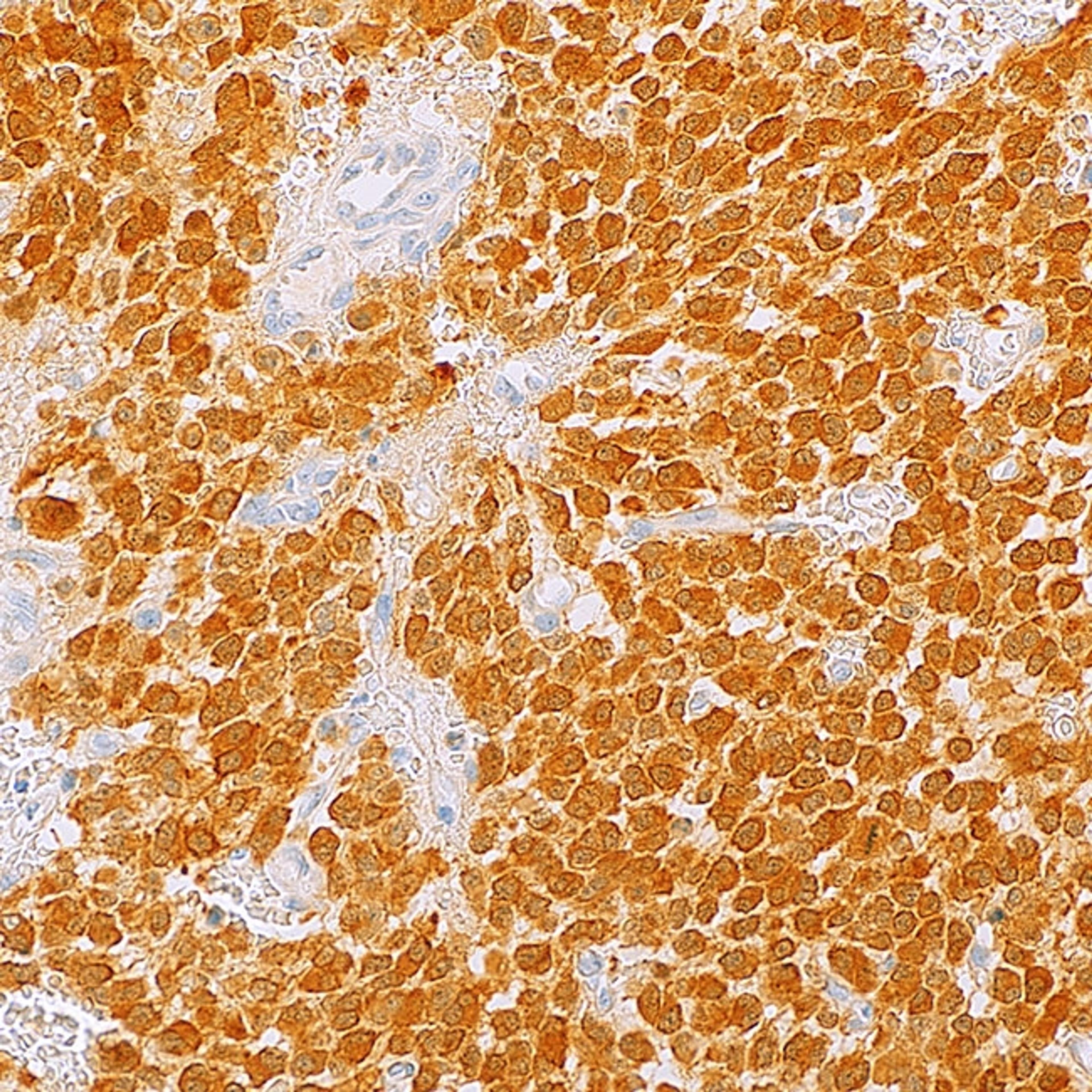
Alongside MRQ-67, the company also developed a rabbit polyclonal antibody product for detection of ATRX that is suitable for classification of gliomas, and the team hope more antibodies for use in neuropathology and other applications will follow. “We are investing more and more into the development of tumor markers, including mutations associated with other types of gliomas,” explains Su.
The growing value of markers in personalized medicine
As the trend for personalized medicine continues to grow, mutation markers are becoming increasingly valuable across the healthcare market. “We've already seen some success with mutation markers for conditions including Lynch syndrome,” says Heller. Also known as hereditary nonpolyposis colorectal carcinoma, Lynch syndrome is the most common cause of hereditary colorectal cancer and can also lead to skin cancer and other tumors, so represents a heavy burden.
“Patients with this diagnosis are often given treatment plans that are specific to them and that tend to result in better outcomes and better prognoses compared to other colorectal carcinoma patients,” explains Heller. This same concept shows potential for success in neuropathology applications, which is especially important given the delicate nature of the brain and the obstacles this presents to any proposed surgical approaches.
Heller concludes by sharing his vision of the future: “From a treatment standpoint, if we're able to identify specific mutations and the specific proteins that arise from these mutations, leveraging existing technology for antibody-drug conjugates against those proteins may then enable us to accurately deliver and focus a drug to right where the tumor is.”