‘Our Mission to Meet the Increasing Demand for New Recombinant Antibodies in Pharma’
As the global antibody market hits new heights, meet one scientist working to bridge the gap between biomarker discovery and antibody development with high-speed phage-display technology
29 May 2019

The latest tools and technologies continue to reveal a deeper insight into genomic and proteomic changes in disease, in more physiologically relevant models, hence many new protein targets and biomarkers have emerged in recent years. This has led to a pressing need for drug compounds to be developed for these novel entities and, as a result, therapeutic antibodies and antibody fragments are an extremely fast-growing class of drugs – both in the innovator and biosimilar industries. What’s more, there is a considerable need for novel antibody probes to support the characterization of lead compounds, and monitor patient response to drug-target complexes, in pre-clinical and clinical trials.
Engineering highly specific and customized antibodies for such applications is a highly skilled process that is both time-consuming and expensive, as well as extremely sensitive to reproducibility issues. In a compelling interview with Andre ten Haaf, specialist scientist within the custom antibodies service of Bio-Rad Laboratories, SelectScience finds out how rapid, reproducible antibodies can be generated, in an animal-free environment, to address the widespread need for new recombinant antibodies.
SS: How did you become interested and involved in the development of recombinant antibodies, and what does your work entail?
ATH: After completing my Ph.D. at the Justus-Liebig University in Gießen, I started working as a scientist in Bio-Rad’s custom antibody services team in June 2016. Our focus is the use of antibodies as critical reagents in research and diagnostic assays, and to support drug discovery and development. Within this, I am responsible for designing and executing new projects related to antibody generation and characterization.
Specifically, these projects include the generation of highly specific, high-affinity anti-idiotypic antibodies to support preclinical research, clinical trials and patient monitoring for innovator and biosimilar products. Furthermore, our expertise facilitates the successful development of therapeutic antibodies and antibody fragments by employing robust bioanalytical methods for characterization. My role involves coordinating the assays for antibody characterization, including affinity determination and epitope binning experiments, for example.
SS: Why is there such a considerable gap between target proteins and available antibodies?
ATH: The use of new technologies in genomics and proteomics has led to the identification of many new protein targets and biomarkers for which no antibodies are available, and there are several reasons for this. First, conventional in vivo methods of antibody generation for commercial use leads to the production of variable clones with unpredictable target affinities.
Second, there are often batch-to-batch inconsistencies that arise, requiring extensive post-production testing and quality control to be implemented. Third, antibodies can be especially challenging to generate when the binding site is required to target protein complexes, such as drug molecules bound to the antigen. Essentially, the complex and therefore time-consuming nature of antibody development has produced a critical need for rapid antibody production services that can achieve reproducible and highly specific reagents for diagnostic, therapeutic, and research use.
One other important point to note is that therapeutic antibodies and antibody fragments are an increasingly popular class of drugs – dominating the biopharmaceutical industry for novel and biosimilar products in recent years. They have a widening application in medicine due to improvements in engineering abilities and are often able to circumvent methods of drug resistance for existing therapies. Plus, with many first-line monoclonal antibodies coming to the end of their patents, the biosimilar market has boomed.
SS: What are anti-idiotypic antibodies, and how could they impact drug discovery and development?
ATH: Anti-idiotypic antibodies are important for pharmacokinetic studies and for the development of immunogenicity assays. They are a class of antibodies that bind to other antibodies via the idiotope – the unique antigenic determinants of the terminal variable region. We have developed different classes of anti-idiotypic antibodies, offering options for flexibility in assay design: inhibitory antibodies (Type 1), non-inhibitory antibodies (Type 2), and drug-target complex-specific antibodies (Type 3). The Type 3 antibodies are especially challenging to generate and are unique to Bio-Rad.
These different classes of anti-idiotypic antibodies allow detection of several forms of the therapeutic antibody present in the patient sample, either free drug, partially-bound or fully bound.
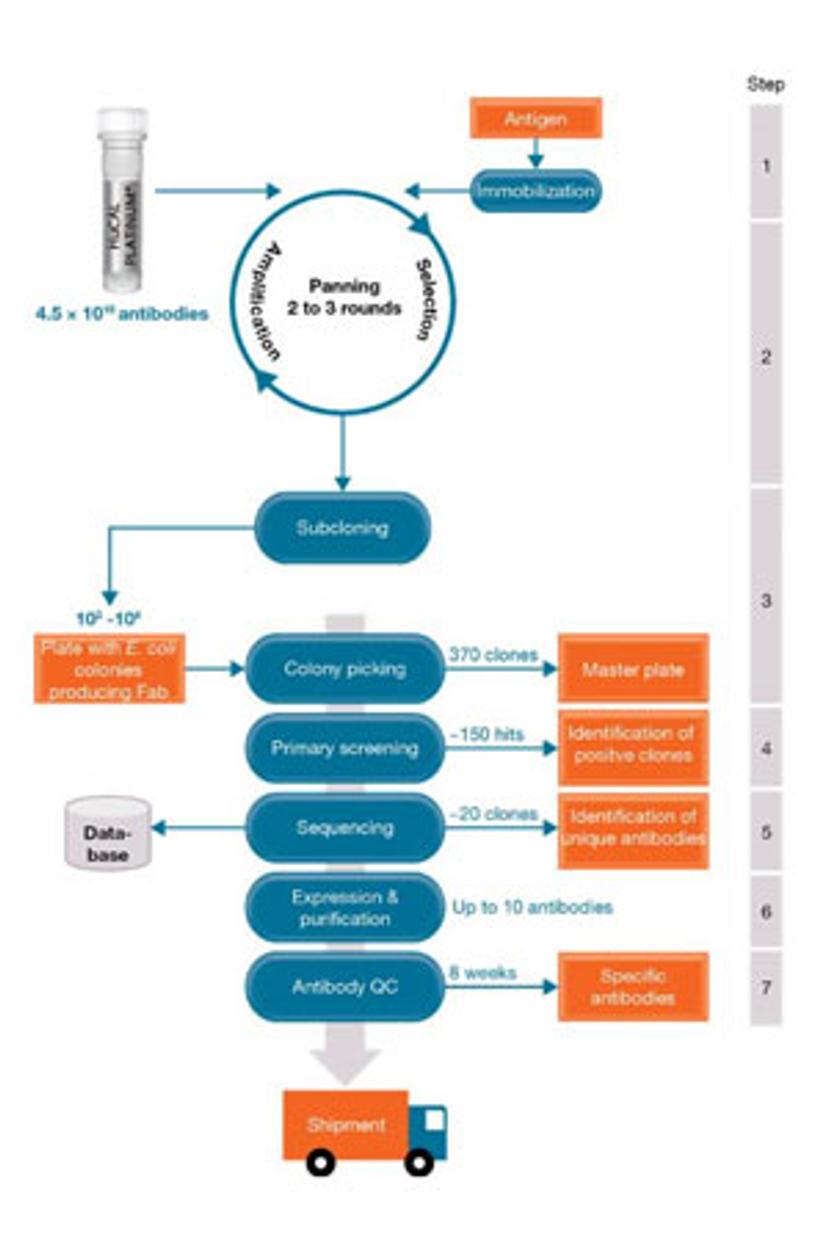
SS: How do you achieve antibody production in a way that overcomes the limitations of conventional approaches?
ATH: Our team uses phage display technology to generate highly specific, monoclonal antibodies in as little as eight weeks. We are using the well-known, high-quality HuCAL® antibody library, and by drawing on many years of experience, we can design guided selection strategies to identify highly specific antibodies from the library. This allows customers to design and develop unique or improved assays.
The benefits of our approach are abundant: phage-display mediated production of antibodies is a process that occurs in vitro, thus reducing the use of animals in research. Furthermore, the modular composition of the HuCAL® library gives us the option for rapid affinity optimization if needed. For additional flexibility, the recombinant antibodies can be produced in various formats, including monovalent and bivalent Fab fragments with a choice of epitope tags, or full-length human and chimeric immunoglobulins.
All of this wouldn’t be possible without the simultaneous sensing and high-throughput capabilities of our lab instruments. We use the Octet Red384 by ForteBio, for example, at four different steps of the antibody generation and characterization process, making it an integral tool to our work. These steps include (1) off-rate ranking, (2) affinity determination, (3) epitope binning, and (4) quantitation of immunoglobulin content in cell supernatant.
SS: What do these processes involve, exactly?
ATH: Off-rate ranking is a screening procedure which enables ranking of the antibodies coming from a particular project according to their dissociation rate (kd) in crude E. coli lysate. Clones with preferred kd rates (high, medium, low or mixed) are then selected for further characterization.
Affinity determination of the purified antibodies provides important information about the binding behavior and the binding strength of an antibody. With the epitope binning we establish whether two antibodies that are specific for the same target recognize different epitopes. Finally, the quantitation in cell supernatant enables the determination of the immunoglobulin concentration after production.
Visit our new Engineered Antibodies Special Feature to find out more >>
SS: What is the Octet Red384, and why does it benefit antibody production specifically?
ATH: The Octet Red384 is a microplate-based screening and analysis system that benefits our processes due to its fast measurement, and 16 simultaneous sensors, enabling high throughput. This is important for us since we produce many different antibodies in a short period of time that need to be analyzed quickly. We can measure hundreds of customized antibodies in a relatively short time.
Furthermore, the microchannels don’t get clogged; the sensors can be regenerated for re-use; and we can analyze E. Coli lysate in a label-free manner. Another great benefit is that consumption of reagents is low – only a minimal amount of antigen and antibody is required for our assays which is hugely beneficial to our multi-step processes. Lastly, the software is very intuitive; the ability to rank the antibodies by their dissociation rate at an early stage improves the chances of a successful outcome for the project in a shortened timeline. All these advantages would lead me to recommend the instrument for users with similar requirements.
SS: With the global monoclonal antibody market hitting new heights, what are your thoughts and predictions for the future?
ATH: Interesting and important protein targets and biomarkers continue to be identified for which no antibodies are available, so we are confident that our antibody generation services and recombinant antibody products will continue to be necessary for scientists working at the forefront of proteomic research, diagnostic assay development, and in drug discovery and bioanalysis.
Phage display technology and other in vitro antibody selection techniques will continue to play a crucial role in this field, primarily because they overcome the limitations of traditional animal immunization methods. They enable the generation of highly specific antibodies against almost any given target, including low immunogenic, toxic, and complex antigens, in a matter of weeks rather than months. Ultimately, our work is, and will continue to, accelerate novel therapeutic discoveries.
Visit our Engineered Antibodies Feature for more on the latest advances in the field >>

