Identification of Microglial States in Neurological Diseases - Immunostaining with JIR Secondary Antibodies
How a researcher at UC San Diego is elucidating the role of microglial dysfunction in brain trauma and disorders
19 Apr 2018

Dr. Hui-quan Li, senior postdoctoral fellow at the Section of Neurobiology, Division of Biological Sciences at UC San Diego, USA, studies microglial phagocytosis and pinocytosis in brain trauma and disorders
SS: Briefly introduce yourself and your place of work
HL: I am a senior postdoctoral fellow at the Section of Neurobiology, Division of Biological Sciences, UC San Diego, and study neuroplasticity in the adult brain with Dr. Nicholas Spitzer, who has made important discoveries of neurotransmitter switching in both developing and adult central nervous system (CNS). Neurotransmitter identity in mature neurons has long been thought fixed, but this dogma has been challenged recently by findings of neurotransmitter switching, where neurons demonstrate plasticity by changing their transmitters, adapting to ever-changing environments. I am leading a group of highly intelligent and motivated people to identify transmitter switching in the adult rodent brain and we study how transmitter switching regulates behavior. I obtained my Ph.D. from the Institute of Neuroscience, Chinese Academy of Sciences, Shanghai, under the mentorship of Dr. Shumin Duan, who has made great achievements in studies of neuroplasticity, glial function and neuron-glia crosstalk. During my Ph.D., I studied the immune function of microglia, which are known as resident macrophages in the CNS.
SS: Describe your research on microglia in brain infection or injury. Why is this research important?
HL: Microglia are the resident immune cells in the CNS and play important roles in both developing and adult brains. Microglial dysfunction is associated with pathogenesis in brain trauma and disorders. Knowing the mechanism underlying microglial phagocytosis and pinocytosis is the basis of understanding microglial functions in both health and disease. By using primary rat microglia cultures and transgenic mouse lines, I found that purines (both ATP and UTP) trigger microglial pinocytosis via P2Y4 receptors and this purine-mediated pinocytosis mediates clearance of soluble β-amyloid, a key mediator in Alzheimer’s disease. This study reveals the mechanism of β-amyloid clearance by microglia and provides new targets for developing therapeutic approaches for Alzheimer’s disease. In addition to their roles as scavengers, microglia are also antigen-presenting cells. Upon infection or in disease, microglia present antigens to T cells to prime immune responses. By using multidisciplinary approaches, alongside my colleagues, I found a size-based sorting mechanism for endocytic contents in microglia and we demonstrated that this size-based sorting machinery is necessary for presentation of exogenous antigens. This work identified a novel mechanism for molecule transport in endocytic pathway and provides a new aspect in understanding antigen presentation.
SS: Your research involves a lot of immunostaining. Can you explain the benefits of immunostaining to study the role of microglia?
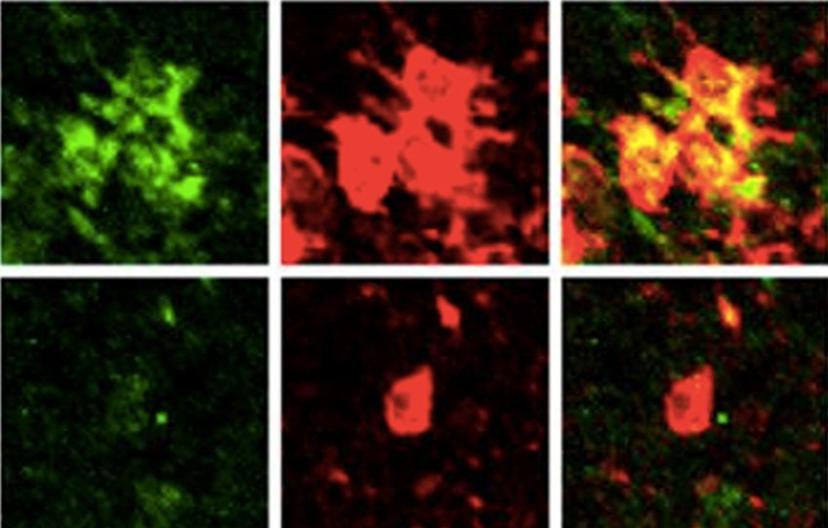
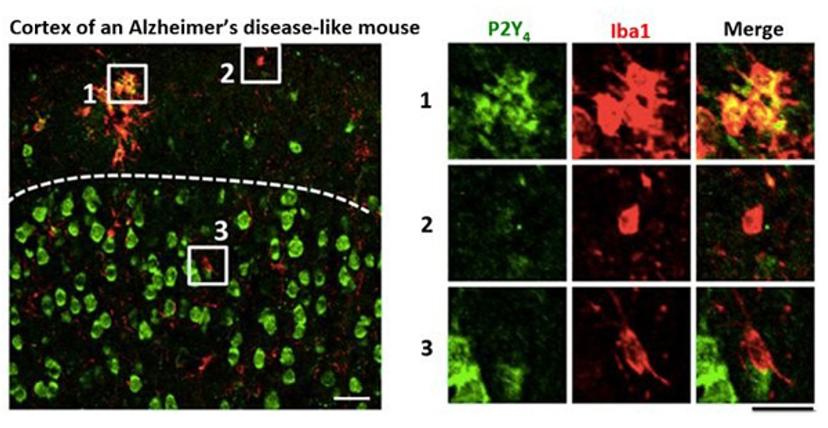
HL: Immunostaining is an irreplaceable technique in biological studies, with numerous benefits. It is an easy and reliable approach that allows in situ identification of microglial states and gene expression patterns. In our studies of purine-mediated pinocytosis, I co-stained P2Y4 purinoreceptors and Iba1, a microglial marker, in brain sections of APPswe/PS1dE9 mice, a transgenic model of Alzheimer’s disease and found that resting microglia – that are far away from amyloid plaques – look ramified, and expressed an undetectable-to-low level of P2Y4. However, levels of P2Y4 expression in microglia that were concentrated around amyloid plaques and transformed into an amoeba-like morphology were dramatically enhanced. This result associates P2Y4 with its function, i.e. mediating clearance of β-amyloid. Immunostaining is also an excellent approach to specify subcellular localization of proteins. For example, in the same article, by immunostaining, I identified that P2Y4 receptors and phosphorylated Akt were both enriched on ATP-induced membrane ruffles and newly-formed pinosomes. These results suggest the potential role of these molecules in pinosome formation and lead us to test the necessity and sufficiency of the activation of the P2Y4-PI3K-Akt pathway in purine-induced pinocytosis.
SS: Please share your experience using Jackson ImmunoResearch Secondary Antibodies. What do you like about them?
HL: When designing immunostaining experiments, I look for primary antibodies from multiple vendors but for secondary antibodies I usually order from Jackson ImmunoResearch. They have a broad collection of secondary antibodies. Among them, I can always find the one that matches my needs (e.g. species, isotypes, conjugated fluorophores, etc.). Their antibodies give sharp signals, negligible background and consistently nice quality. We use immunohistochemistry as a semi-quantitative approach. Reliable antibodies ensure decent quality and consistency of our experimental results.
SS: What other projects are you working on?
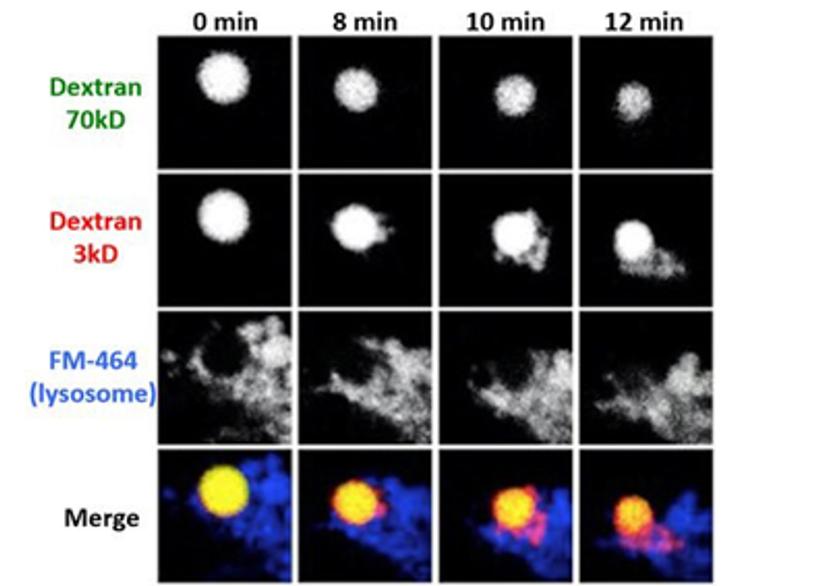
HL: Another project I’d like to share is my microglial antigen-presentation project. By using live-cell imaging, I found that polysaccharide molecules of either small (M.W. < 10 kD) or large size (> 40kD) were both internalized by microglia via pinocytosis and they were enclosed in the same pinosomes. However, after around eight to ten minutes, smaller molecules exited the pinosomes and entered lysosomes, leaving the large molecules behind.
By using immunostaining and live-cell imaging, we then found that both fusion and fission between pinosomes and lysosomes were required for this size-based sorting to occur. We were also able to demonstrate that the size-based sorting process is necessary for microglial presentation of exogenous antigens.
SS: What is the future of your research?
HL: Subtypes and functions of microglia are still mysterious. Discoveries have been made in the past decade that uncovered important functions of resting microglia during development (e.g. pruning synapses) and in adults (e.g. monitoring neuronal activity). However, the underlying molecular mechanisms remain largely unknown. Purines are crucial messengers in, but not restricted to, the CNS. Purinergic receptors play important roles in reactive microglial functions, e.g. P2Y12 in migration, P2Y6 in phagocytosis, and P2Y4 in pinocytosis. It would be interesting to test whether purines and their receptors also contribute to the newly identified functions of resting microglia. Another interesting direction is to study crosstalk between microglia and T cells in both healthy and diseased brains. T lymphocytes have long been thought to invade the CNS only in inflammatory neurological diseases such as multiple sclerosis, vasculitis, etc. However, recent findings have identified their roles in maintaining CNS homeostasis under healthy conditions. In fact, around 150,000 T cells can be found in the cerebrospinal fluid (CSF) of healthy humans. Given that the size-based sorting that we have identified occurs both in vitro and in vivo and is required for antigen presentation, verifying the contribution of this sorting machinery to antigen presentation in the intact brain would be prominent to understanding the function of microglia-T cell crosstalk in both health and disease.
Do you use Jackson ImmunoResearch Whole IgG Affinity-Purified Secondary Antibodies in your research? Write a review today for your chance to win a $400 Amazon voucher.

