Fapon expands one-stop solution for the IVD industry and sets sights on next-generation therapeutics
Fapon’s new Shine i8000/9000 CLIA analyzer caters to high-volume clinical testing needs, while the company is also scaling up efforts to bring advanced cancer therapies to market
14 Sept 2023
Fapon has launched its latest chemiluminescence immunoassay analyzer, the Shine i8000/9000 series, at this year’s Association for Diagnostics and Laboratory Medicine (formerly known as AACC) Annual Scientific Meeting & Clinical Lab Expo. The Shine i8000/9000 series brings exceptionally high-throughput testing capabilities in an open and modular format, adding to Fapon’s diverse one-stop solution of analyzers, reagents, and critical raw materials for in vitro diagnostics (IVD) developers and clinical labs. In addition to acting as an IVD enabler, Fapon is also gaining footholds in the biopharma industry, where it is working to unlock the potential of artificial intelligence (AI), circular RNA, and other emerging technologies to bring more affordable and efficacious therapeutics to market.
We recently talked to Fapon about its key products and offerings, how it is forging close partnerships with IVD and biopharma innovators to help shape the future of healthcare, and to discover the advantages of the Shine i8000/9000 series.
What is Fapon?
Fapon is a leading global life sciences company dedicated to providing integrated solutions and services for diagnostics, biopharma, and biotherapy. Integrating a one-stop solution on raw materials, reagents, and innovative open instrument platforms, the company has grown into a leading supplier in IVD. Fapon boasts advanced innovations in therapeutic technologies and AI algorithm platforms with data insights that contribute to a better and healthier world.
“Fapon has been dedicated to the IVD upstream raw materials field for over 22 years, and we are the top supplier of IVD core materials in China,” says Jielun Zhu, CFO & CIO of Fapon. “We also strive to co-create diagnostic ecosystems with our partners globally to accelerate the development of technologies in this industry.”
While founded in diagnostics, since 2015, Fapon has also been gaining ground in the biopharma arena. Here, its focus lies in developing and manufacturing novel biologics and cell therapy products against cancer and autoimmune diseases. “We have developed world-leading technology platforms of antibody discovery and bispecific antibody development to support a pipeline of over 20 projects targeting different stages of the cancer immunotherapy cycle,” says Zhu. “Our overall mission is to enable more reliable and affordable biologic therapies.”
A springboard for IVD innovators
Where Fapon distinguishes itself from other suppliers is its commitment to delivering highly integrated products and services, known as ‘one-stop solutions’. This holistic approach encompasses a comprehensive range of core raw materials, reagent solutions, and open instruments, along with original equipment manufacturer (OEM) and contract development and manufacturing organization (CDMO) services.
“We can be considered similar to a kitchen maker,” explains Dr. Sandeep K. Vashist, Senior Global IVD Product Director of Fapon. “We provide the cooking instruments (our open analyzers), the ingredients (the raw materials and the reagents), and the recipes (the procedures or the protocols that we have optimized).”
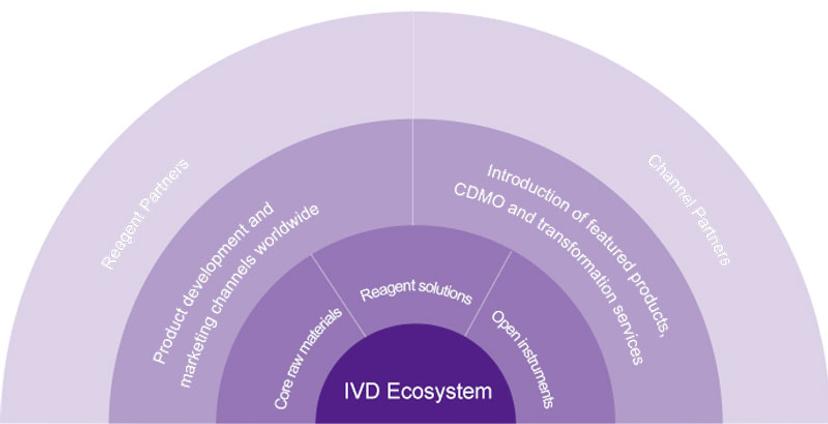
“In cases where customers have their own raw materials or reagents, we can also bulk manufacture these in accordance with stringent quality control standards,” he continues. “We provide all these services as a one-stop solution, so customers don’t have to look for many different suppliers as they have everything under one umbrella.”
Through this model, Fapon aims to empower both IVD developers and manufacturers to improve their abilities in product development, build independent IVD manufacturing capabilities, and efficiently break into local and global markets. “We call this ecosystem co-creation,” explains Zhu. “It's not just selling products and services, but establishing a much closer, longer-term relationship with our customers by working with them as an IVD enabler.” This goes so far as offering technical services such as factory construction design, product registration support, quality assurance systems, and product aftercare services.
“Over the last decade, we have forged many successful partnerships across the globe, including in China, India, Korea, Brazil, England, and several other countries,” adds Kevin Liu, Global Sales Director of Fapon. “A point of particular pride for us has been our role in improving local healthcare systems, for example, by working with partners to help them to build up their own technology to detect local diseases such as dengue, malaria, and HIV.”
“By supporting partners in countries where those diseases have been prevalent, we’ve helped them bring more affordable diagnostic solutions to the market,” he adds. “As a result, early stage and rapid detection of diseases has been growing very quickly by local providers, and now many countries have very good control of those diseases.”
Fapon unveils the Shine i8000/9000, its new ultra-high-throughput CLIA analyzer
Fapon’s one-stop solutions span a wide range of product lines, including ELISA, rapid tests, PCR, NGS, and immunoturbidimetry, accompanied by over 1400 antibodies, antigens, enzymes, blockers, and other critical reagents. Among these, a highlight is its chemiluminescence immunoassay (CLIA) portfolio, which features a range of fully automated open CLIA analyzers.
The Shine i8000/9000 series is a pivotal addition to this lineup and represents the company’s highest throughput system to date, with the capacity to perform up to 900 tests per hour. “The Shine i8000/9000 can handle 410 samples in a single run and has a modular design, allowing up to four units to be connected in tandem, increasing the throughput to over 1,600 samples per run,” explains Dr. Vashist. “The analyzer can run up to 60 different tests, with most assays completed within 10 minutes, meaning it can handle more than 10,000 samples per day.”
The ultra-high throughput of the Shine i8000/9000 makes it particularly well suited to centralized clinical testing labs, while Fapon’s previous Shine instruments offer benchtop options for healthcare settings with low-to-mid throughput requirements. “The Shine i1000 has the smallest footprint and can run 120 tests per hour and up to 10 different assays, which makes it very useful for decentralized labs, point-of-care settings, or physician office labs,” says Dr. Vashist. “The Shine i2000 can perform 200 tests per hour and run up to 25 different assays, making it best suited for mid-throughput labs, which require several hundred samples to be tested in a day.”
According to Dr. Vashist, the most significant benefit shared across all of Fapon’s Shine CLIA analyzers is their open architecture. This enables users to leverage Fapon's proprietary raw materials and reagents or their own, unlocking the potential to develop highly specialized tests that address local clinical needs.

At the forefront of novel and emerging technologies
A cornerstone of Fapon’s success and industry leadership is its strong commitment to research and development. With over 880 existing patents, the company has established an extensive range of proprietary technologies across its instrument platforms and raw materials. “Most of the technologies with our CLIA analyzers are proprietary, and in terms of raw materials, we recently introduced protein-free blockers and heterophilic antibody interference blockers for IVD assays,” says Dr. Vashist. “Our protein-free blocker, being 15 to 20 times smaller in size than conventional blockers such as bovine serum albumin, provides much better blockage of solid surfaces, thereby minimizing non-specific interactions in IVD assays. Because it is chemically synthesized, we can produce it in bulk, and it isn’t prone to denaturation, biofouling, or other effects that you can expect in protein blockers.”
The company has also built-up advanced expertise in antibody phage display technology, a genetic engineering technique that enables the animal-free production of antibodies through their presentation on the surface of bacteriophages. “We’re using our capabilities and propriety technologies in phage display to not only advance antibody development for IVD applications but also develop therapeutic antibodies against our biopharma targets,” explains Dr. Vashist.
“We're also looking at cutting-edge AI algorithms for the early screening and diagnosis of cancers and identification of novel biomarkers, as well as AI-enabled drug discovery and screening,” adds Zhu.
To facilitate its wide-ranging initiatives, Fapon has a global R&D footprint, which Zhu refers to as the ‘six plus one roadmap’. The six refers to established R&D hubs located across China, the US, and Europe. Each site has a different focus, with teams working on areas including next-generation enzymes, circular RNAs, NGS, lab automation, and AI technologies. “The plus one refers to a potential new site we're looking at in San Diego,” Zhu adds. “This will be mostly focused on innovative diagnostic platforms, and there is currently a small ecosystem of several interesting diagnostic companies based out of San Diego.”
The path forward for IVD: Trends and insights
When asked what trends he sees shaping the future of the IVD industry, Zhu highlights the shift towards earlier detection and integrated treatment. “When you intervene in a patient’s disease cycle, you want to both diagnose and treat at the same time,” he says. “Companion diagnostics are also very important, because many drugs are blunt tools, meaning we need ways to tailor treatments to individual patient responses. We're in the early stages of doing that, and in the future, diagnosis and treatment will go hand in hand.”
Liu, on the other hand, emphasizes the emerging trend of IVD localization. “Since the COVID-19 pandemic, many governments have realized the importance of the local development of IVD companies, and several countries have introduced policies to encourage localized production, enabling the provision of affordable, region-specific products,” he says. “Fapon is excited to see this because we have been working with local partners for decades. We believe that we can empower them to be more successful in the local market and even become global.”
While seeing vast potential in advanced technologies such as AI and machine learning, Dr. Vashist underscores that their integration into clinical practices is an arduous process, with challenges extending to regulatory approvals, market readiness, and cybersecurity. “The adoption of any new technology or test hinges on clinical evidence and validation which can take many years, and even then, products need to be approved by reimbursement agencies to be commercially viable,” he says. “There are many examples of IVD products that are FDA approved but don’t make it to market, so right now, all the big players are learning and keeping a keen eye on emerging applications. On the discovery side, things are going great, but to see these in actual practice, I think we’ll have to wait and see how the field will shape up.”

