Tools for chromatographic success: Ensuring data quality in routine analysis
Hear from the experts about how the new Waters Arc HPLC System easily addresses the challenges of pharma analysis, quality assurance, and compliance
4 Oct 2020
A recent report from Deloitte indicates that data integrity violations account for over 70% of the warning letters issued globally. Violations are frequently attributed to a lack of complete data, suspect data, and improper audit trails.
Many pharmaceutical QC laboratories face a multitude of challenges in ensuring the integrity and quality of their data in routine analysis. At the same time, scientists often find themselves restricted by regulators when auditing their peak integration practices. Complying with these “right-first-time” expectations could be easily solved by modernizing and improving the performance of chromatographic analyses. However, optimizing separation methods, which are part of regulatory submissions, presents yet another series of challenges as regulatory licensing and method validation costs often discourage any substantial change to these procedures.

Therefore, what simple improvements can be made to better trust the integrity and compliance of your data, while making life easier for both the analysts and the data reviewers? The time for making improvements is now, as new regulatory opportunities for post-approval change management are imminent. The updated guidelines will influence how chromatographic methods are developed and documented, resulting in more examination and oversight of system suitability and reproducibility requirements to reduce measurement error and further ensure overall data quality.
In this popular SelectScience webinar, Heather Longden, Senior Marketing Manager, and Dr. Paula Hong, Principal Consulting Scientist at Waters Corporation, share insights on how to address the most common chromatographic challenges and ensure the highest data quality and integrity in routine pharmaceutical analysis, while simultaneously setting you and your lab up for success in anticipation of the new guidelines.
Current trends and challenges in analytical regulations
In the webinar, Heather Longden first outlines common issues that regulators come across when dealing with chromatographic analysis. She examines how test methods were often found to be incapable of confidently demonstrating drug purity because the analysts struggle to get accurate peak integration right first time. In one specific regulatory observation reported by FDA, data was found to have been “reprocessed up to 12 times, and only the final result was included in the report for review by Quality Assurance.”
As well as being a drain on scarce resources and time, this activity clearly triggers concerns about whether the data secured or submitted was complete. In fact, it is extremely common and scientifically sound to need to optimize peak integration parameters, but this was interpreted by the regulator as “extensive data manipulation”, especially when the manager’s response was that it was “typical to play with integration parameters”. There was a marked inconsistency concerning the integration of low-level impurities in chromatograms between users and the absence of an approved protocol for manual integration may lead to differences in peak results. These test methods also led analysts to inconsistently integrate co-eluting peaks. Inconsistent peak integration and inadequate analytical methods can lead to a failure to detect impurities, resulting in the release of adulterated drug products to the market.
Webinar poll question: What is the right peak integration?
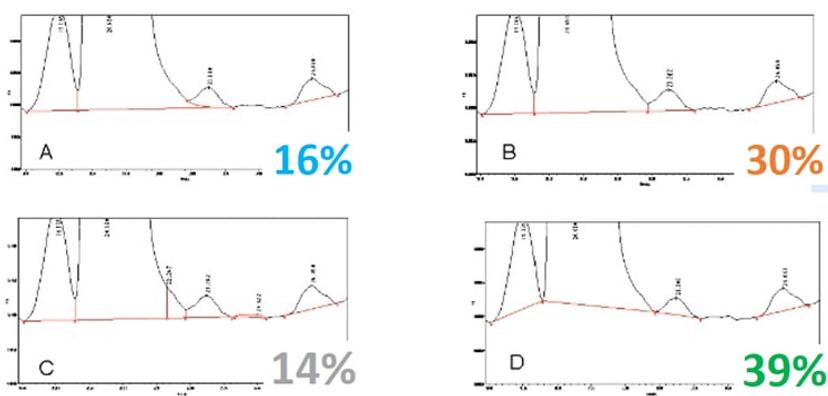
“If you can improve the method so that it’s not too burdensome, you can reduce the manual manipulation,” Longden explains. ”This ultimately reduces the concerns regulators have on data integrity issues, such as raising suspicions that the data was being manipulated into compliance.”
Longden goes on to explain that the most common root cause of these kinds of data integrity concerns is that the chosen analytical methods are often old and have not been modernized since initial development. Ultimately, this may make them inadequate for use in a modern analytical laboratory.
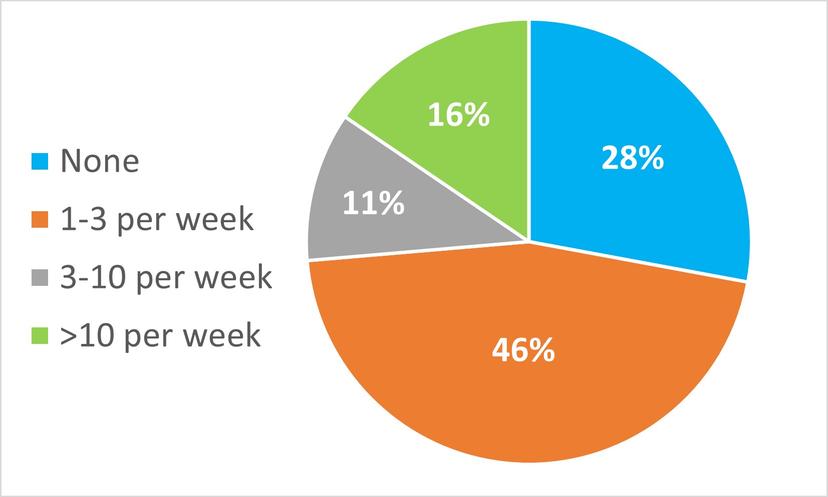
Webinar poll question: In your lab, how many sample sets per week need to be invalidated and repeated because the system suitably test fails?
Only 28% of poll respondents reported rarely having to invalidate and re-run results. 16% reported more than 10 sample sets per week needing to be invalidated and re-run, leading to a significant amount of wasted time and cost. Having robust methods that run reliably on high-performance systems to mitigate system suitability failure is key.
Despite the data integrity concerns, Longden notes that efforts by global regulators and the International Conference on Harmonization (ICH) paint a positive outlook for facilitating post-approval changes to improve analytical methods. Longden describes the ongoing efforts as “a time of change, where users should have more flexibility, providing appropriate upfront knowledge gathering and risk assessment are performed.” Among the key guidelines is the Product Lifecycle - ICH Q12 approach, which specifically includes examples of how changes can be made to analytical methods without requiring regulatory pre-approval. Secondly, the global regulators and ICH are working on new guidance in Analytical Method Development – ICH Q14, which looks to incorporate more method knowledge into the submission of an analytical method. Finally, an ICH working team is revisiting Method Validation – ICH Q2 (R2) to cover enhanced validation and validation of modern analytical techniques.
Webinar poll question: When transferring methods into QC, do you typically:

Ensure success with your HPLC system
With its ability to improve performance, the Arc HPLC System is designed to address these key issues. The Arc HPLC System is designed to run both existing HPLC methods and improved methods on smaller particle-sized HPLC columns, such as 3.5 µm. Hong describes how the Arc HPLC System “gives highly reproducible and precise injections, so you can be more confident that you’ll meet your relative standard deviation (RSD) specifications, without having to re-run your samples,” thus eliminating the need to constantly optimize peak integration parameters and reducing the time spent investigating abnormal results.

The new Waters Arc HPLC System is specially designed to meet common chromatographic challenges by offering researchers a seamless upgrade to legacy quaternary and binary systems, featuring comparable separation quality and retention times. Upgrading to a modern system like the Arc promises to improve existing methods by providing more flexibility and boosting efficiency, with higher injection precision and backpressure limits.
In addition, improvements in HPLC instrument performance and robustness can also contribute to the overall improvement of data quality and integrity. The webinar sheds light on the importance of meeting system suitability criteria for routine methods, such as USP monographs. Not only that, the Arc HPLC also implements increased backpressure tolerance to give users the flexibility to use high flow rates with smaller particle columns, reducing run time, costs, mobile phase consumption, and most importantly, achieving greater peak resolution and sensitivity. Modern instruments such as the Arc HPLC system are also designed to provide reproducibility for HPLC shallow gradients, as typically used in many regulated (USP) methods.
Insights from the audience:
- 69% of respondents cited modernizing and maximizing efficiency as their primary driver for replacing LC assets for routine analysis; 31% cited minimizing risk.
- Reliability, robustness, easy transfer of existing methods, and easy operation and maintenance were noted as the top three criteria when choosing or replacing LC assets for routine analysis.
- Respondents were most likely to replace their HPLC system if they could not achieve system suitability or if the vendor support had been discontinued, indicating that while many labs strive for better performance and have the desire to leverage new technology, a significant segment may struggle to secure resources to routinely update their fleet of lab instruments and obsolescence may be their major driver to replace equipment.
“Finding ways to easily improve your methods and ensure ‘right-first-time’ analysis gives you greater confidence in your data and confidence that regulators trust the results you are presenting or submitting,” states Longden. “Leveraging Waters as a trusted partner helps ensure success in all areas of routine analysis.”
Want to learn more?
Join us on Wednesday, October 7, for an expert panel discussion on how new guidelines and modern chromatographic solutions converge to ensure data quality in the pharmaceutical laboratory. Register for this free SelectScience webinar to hear the latest insights from Heather Longden, Dr. Phil Borman (GSK), Dr. Horacio Pappa (United States Pharmacopeia - USP), and James Pound (Medicines and Healthcare products Regulatory Agency - MHRA).
Plus, find more expert resources for your HPLC analysis below:

