Merck Millipore Introduces New Processing Technologies in Booth 517 at the BioProcess International Conference & Exhibition
12 Oct 2014
Product news
Merck Millipore, the Life Science division of Merck, invites you to visit booth 517 at the BioProcess International Conference & Exhibition October 20-23 in Boston to learn about its new biopharmaceutical processing solutions. These products add to Merck Millipore's complete range of upstream and downstream products, helping to improve manufacturing productivity and accelerate time to market.
The new technologies on display at the BioProcess International Conference & Exhibition will include:
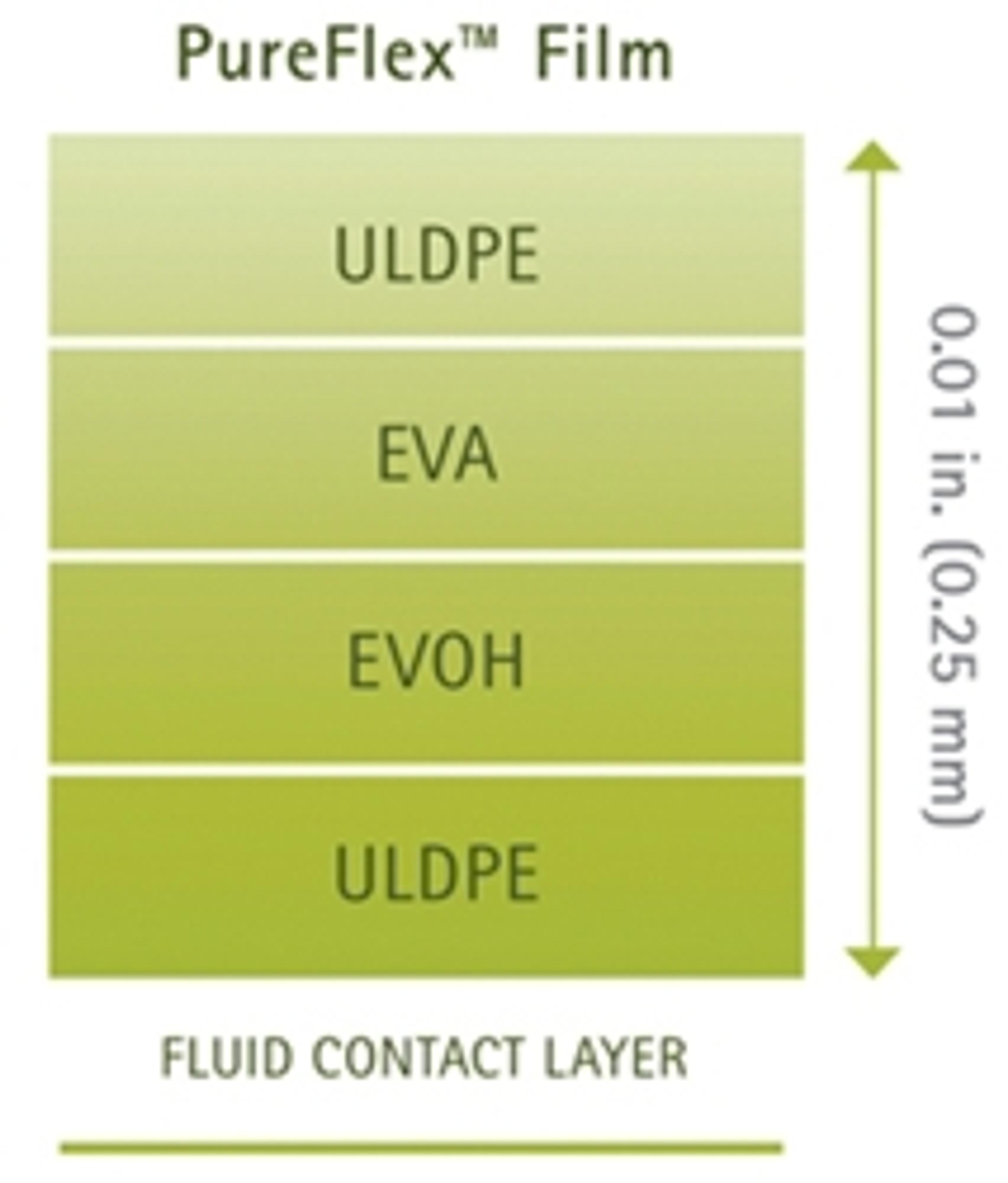
PureFlex™ Plus Single-Use Process Container Film
Merck Millipore is introducing PureFlex™ Plus film, a robust, durable film used in the construction of Mobius® single-use process containers for biopharmaceutical manufacturing. The film's rugged outer layer offers strong resistance to leak formation, making it ideal for the demanding applications often encountered in large volume operations.
The PureFlex™ Plus film can be used throughout the entire manufacturing process. It builds upon Merck Millipore's established PureFlex™ film technology by adding a new, more robust linear low density polyethlene (LLDPE) outer layer to reduce susceptibility to leaks through abrasion, puncture, stretching and tearing.
Cellvento™ CHO Cell Culture Media
Merck Millipore is expanding its Cellvento™ CHO cell culture media portfolio of chemically defined, non-animal origin media formulations for batch and fed-batch mode operations. Three new products are launching to specifically address DHFR negative cell lines: Cellvento™ CHO-110 medium, Cellvento™ CHO-210 medium and Cellvento™ Feed-210 media.
Cellvento™ CHO-110 medium is recommended for batch application and seed train expansion as its rich formulation supports high cell growth. Cellvento™ CHO-210 medium and its companion feeds are effective at achieving high-density cell growth and competitive productivity with DHFR negative CHO suspension cell types and expression systems, especially recombinant CHO DG44 cell lines.
NovaSeptum® Single-Use Holders
NovaSeptum® single-use holders are a new component of Merck Millipore's NovaSeptum® fully disposable sampling systems, which help prevent contamination when sampling fluids from aseptic and sterile processes. The new single-use holders are compatible with NovaSeptum® containers, helping reduce validation requirements, and can be filled with any type of container using a 2mm trigger.
Ready-to-use, flexible and fully validated, the single-use holders are ideal for manufacturers dealing with recombinant proteins, vaccines and mAbs who need to autoclave before starting their processes.
Chromabolt® Chromatography Resins
Merck Millipore is announcing the availability of three new chromatography resins for its Chromabolt® prepacked columns. These pre-validated chromatography columns can come packed with either strong cation exchangers, weak cation exchangers or strong anion exchangers, freeing up end users' time and resources by eliminating manual column packing and cleaning.
Chromabolt® columns are available in three sizes - 10cm, 20cm and 32cm inner diameter (i.d.) - and are designed for pilot and early clinical manufacturing. The three new resins are Fractogel® EMD SE Hicap, Fractogel® EMD COO(M), and Eshmuno® Q.