ResourceSeparations
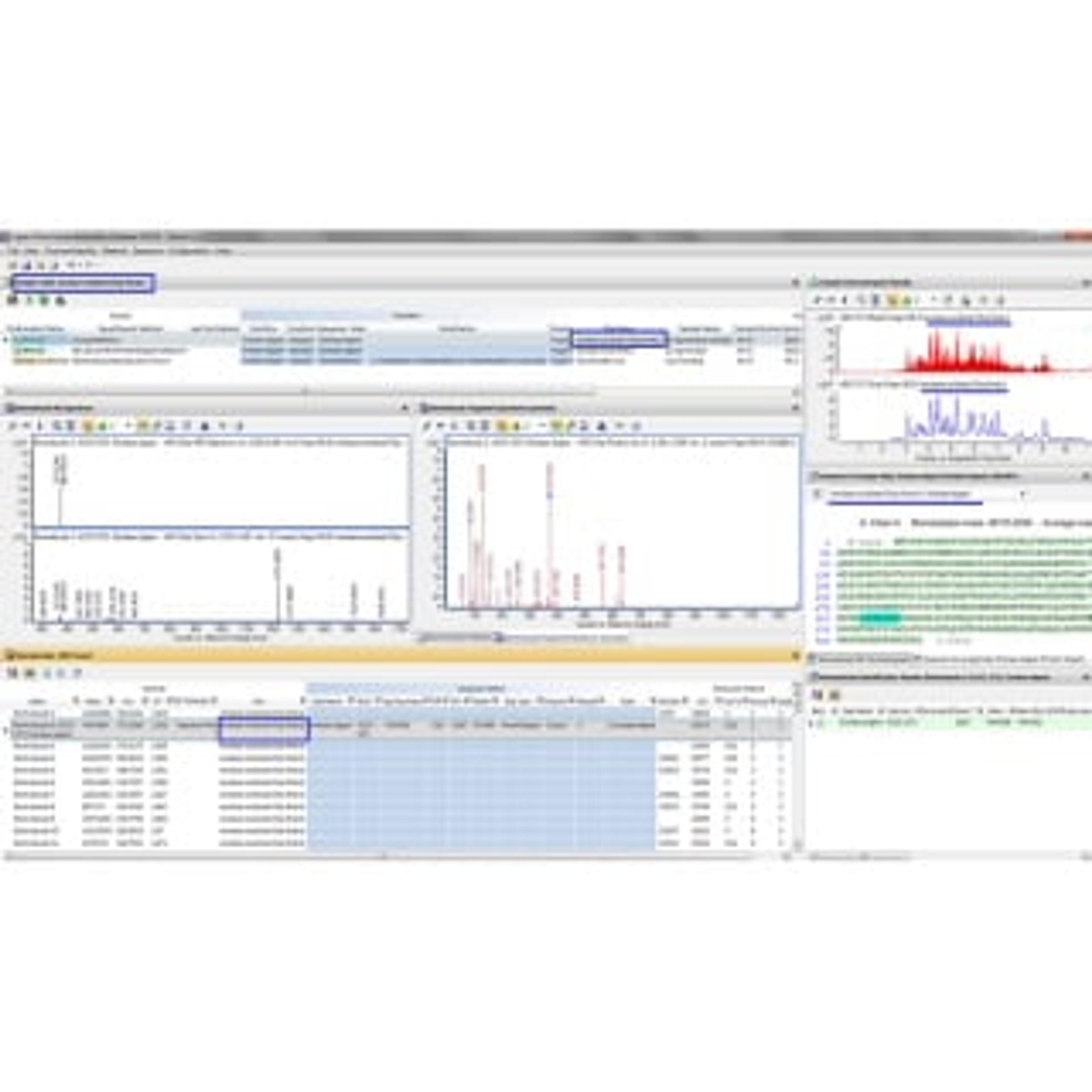
Integrated Workflow for Intact and Subunits of Monoclonal Antibody Accurate Mass Measurements
22 Jan 2018Monoclonal antibody (mAb) based entities represent a rapidly growing class of biologics that require extensive characterization to obtain approval for clinical trials and subsequent market release. Accurate mass measurement is a challenging step in the analytical characterization of antibodies because of their large size and the presence of post-translational modifications such as glycosylation. These characteristics also make determining the location of modifications more complex.