SelectScience InterviewsClinical Diagnostics
Landmark Troponin Assay to Give Results at the Point-of-Care
27 Nov 2013
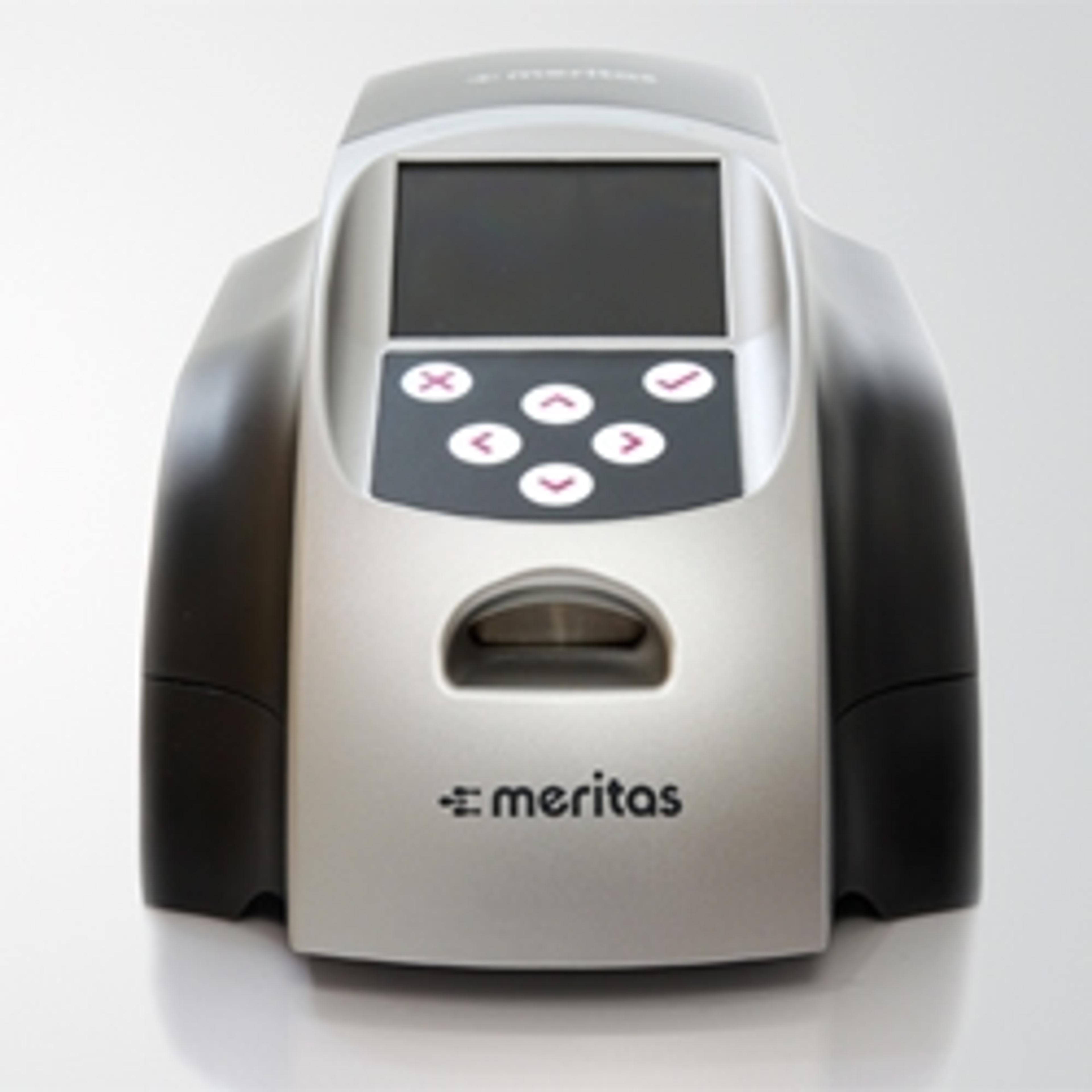
Find out more about the new Meritas® Troponin I point-of-care test, showcased by Trinity Biotech at Medica this year. Enabling fast, accurate triage and intervention of patients at the emergency department, this assay has the potential to generate significant cost savings, reduce turnaround times, and have a massive impact on patient care. Filmed by SelectScience at MEDICA 2013.