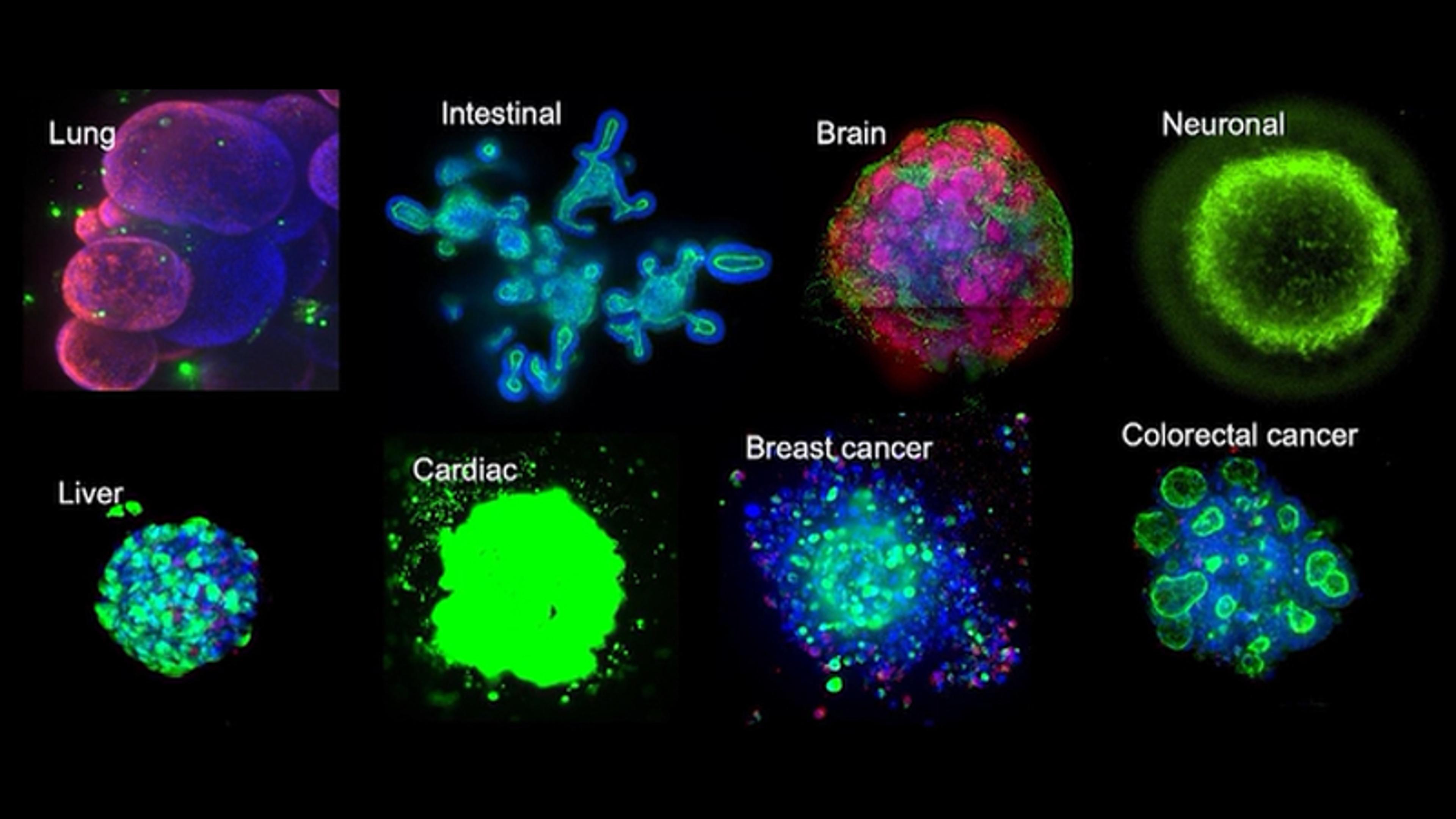
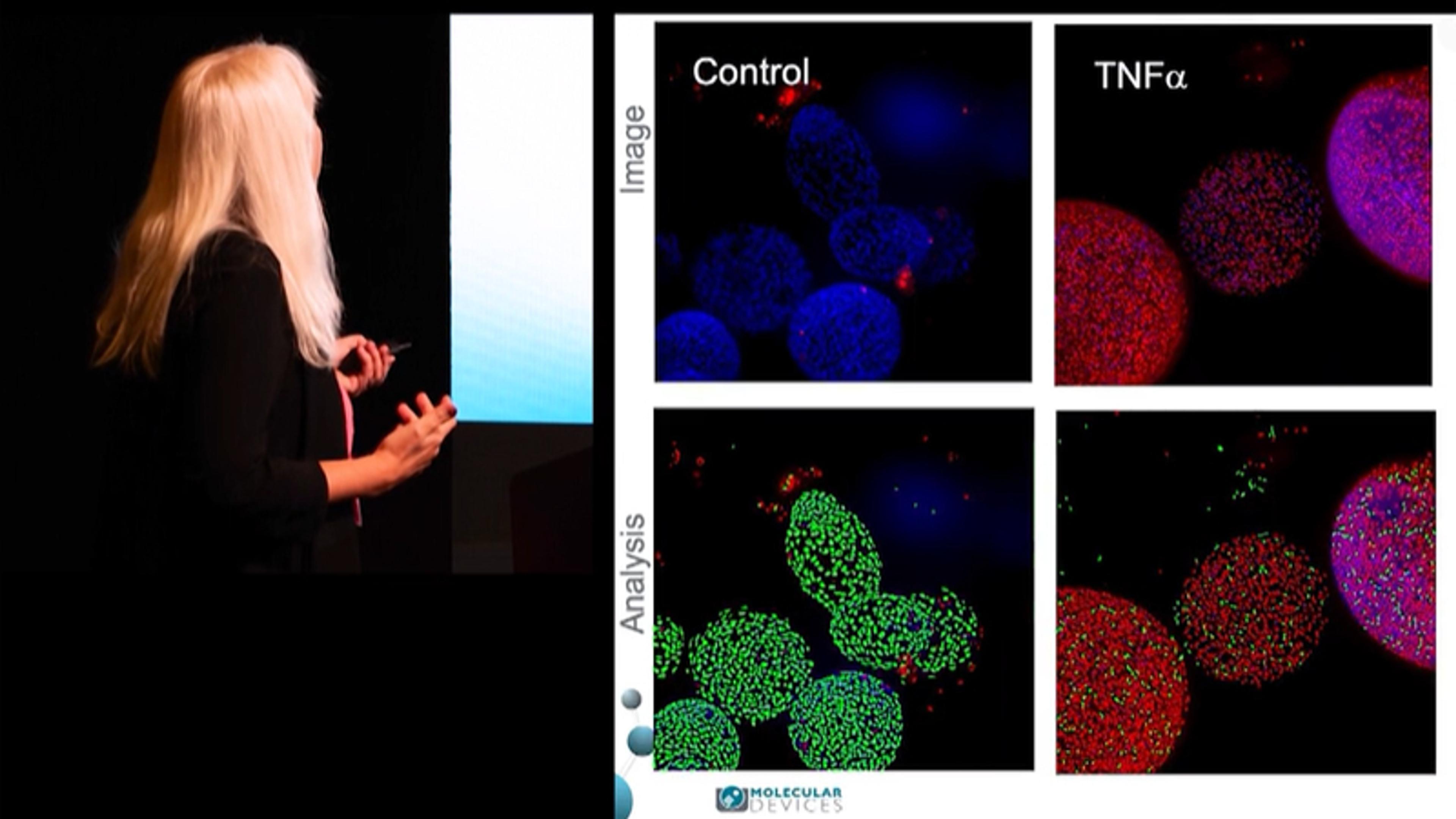
Product NewsLife Sciences
Adapt your lab microplate reader for bacterial endotoxin testing
3 Apr 2025
Join Timothy Francis, from Fujifilm, as he explores the adaptation of microplate readers for bacterial endotoxin testing. This presentation delves into key techniques, including gel clot, turbidimetric, and chromogenic methods, with a special focus on the importance of kinetic measurement approaches. Learn how SoftMax Pro Software ensures compliance with GLP and GMP standards and discover a step-by-step protocol for the Kinetic Turbidimetric assay.