ResourceClinical Diagnostics
Overcome bottlenecks in NGS panel reporting
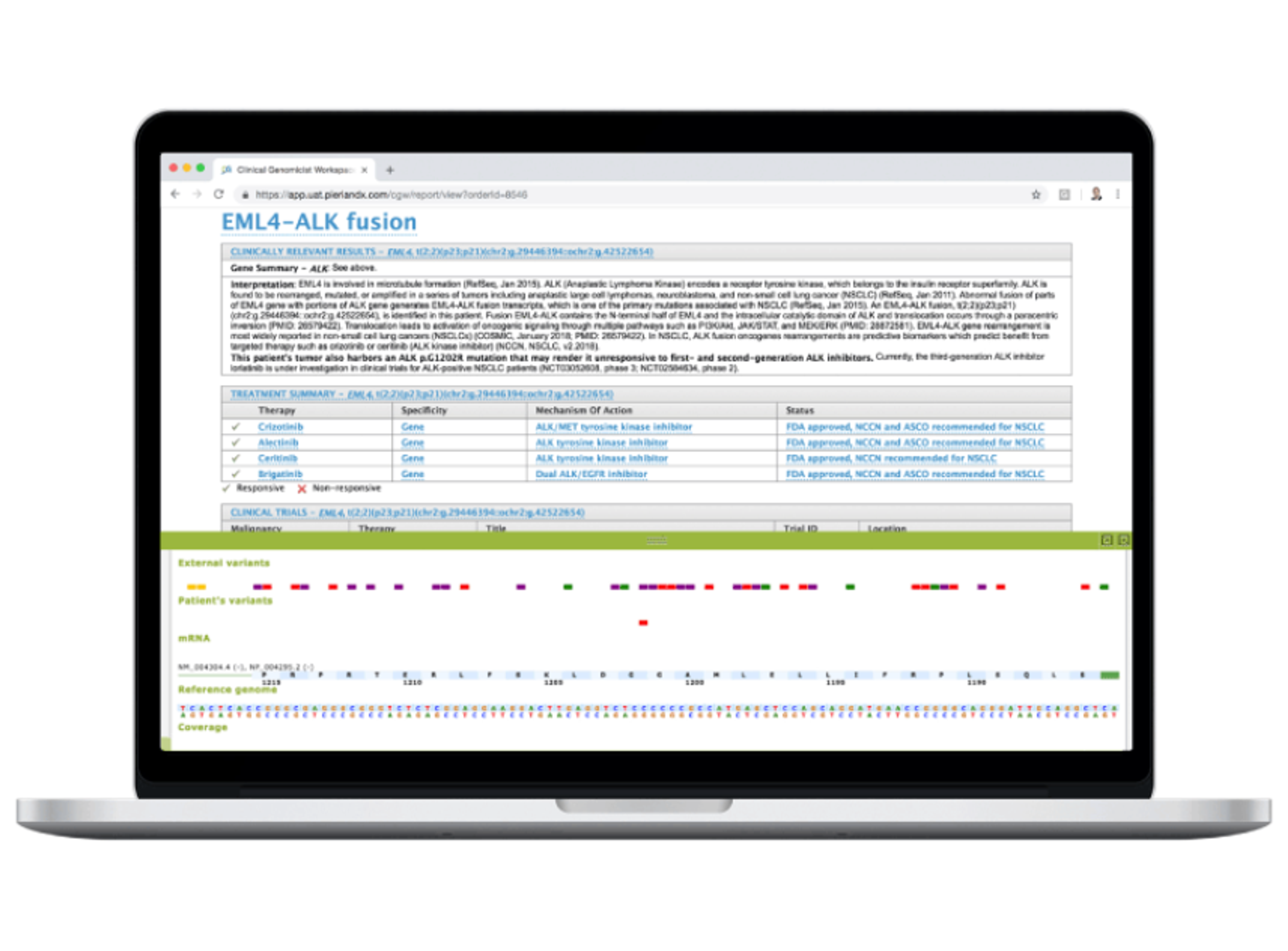
5 Mar 2025Next-generation sequencing (NGS) panel reporting presents challenges in efficiently identifying, reviewing, and reporting clinically relevant variants. With so many variants to assess, how can you ensure accuracy while optimizing turnaround time?
Inside this guide, you'll learn how to:
- Identify concise, accurate clinical content for reporting
- Quickly review large variant sets for quality and clinical relevance
- Build upon past decisions for faster, more consistent reporting
Find a solution that empowers you to:
- Customize quality and clinical evidence workflows
- Set preferences and customizations at the panel level
- Streamline filtering to focus on the most relevant variants
- Leverage a smarter knowledgebase for automated interpretations
- Facilitate VUS triage with variant pathogenicity insights