ResourceLife Sciences
Meet FDA guidelines in GMP/GLP labs
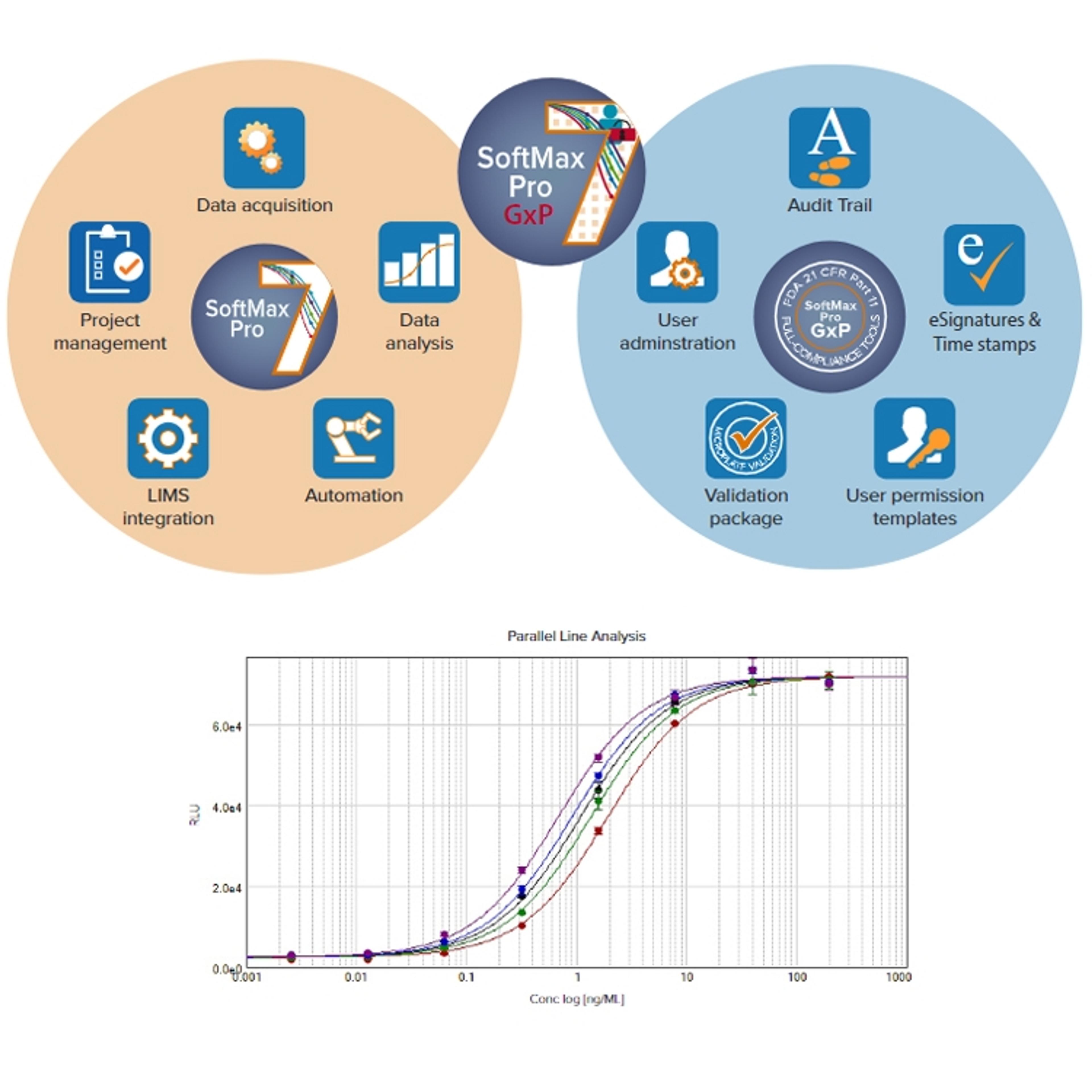
11 Mar 2020SoftMax® Pro 7.1.1 GxP Software is the latest, most secure software to achieve full FDA 21 CFR Part 11 compliance with streamlined workflows to ensure data integrity. Every step is optimized to simplify analysis and reporting to support Molecular Devices microplate readers.