ResourceClinical Diagnostics
Develop and deliver your IVD with confidence
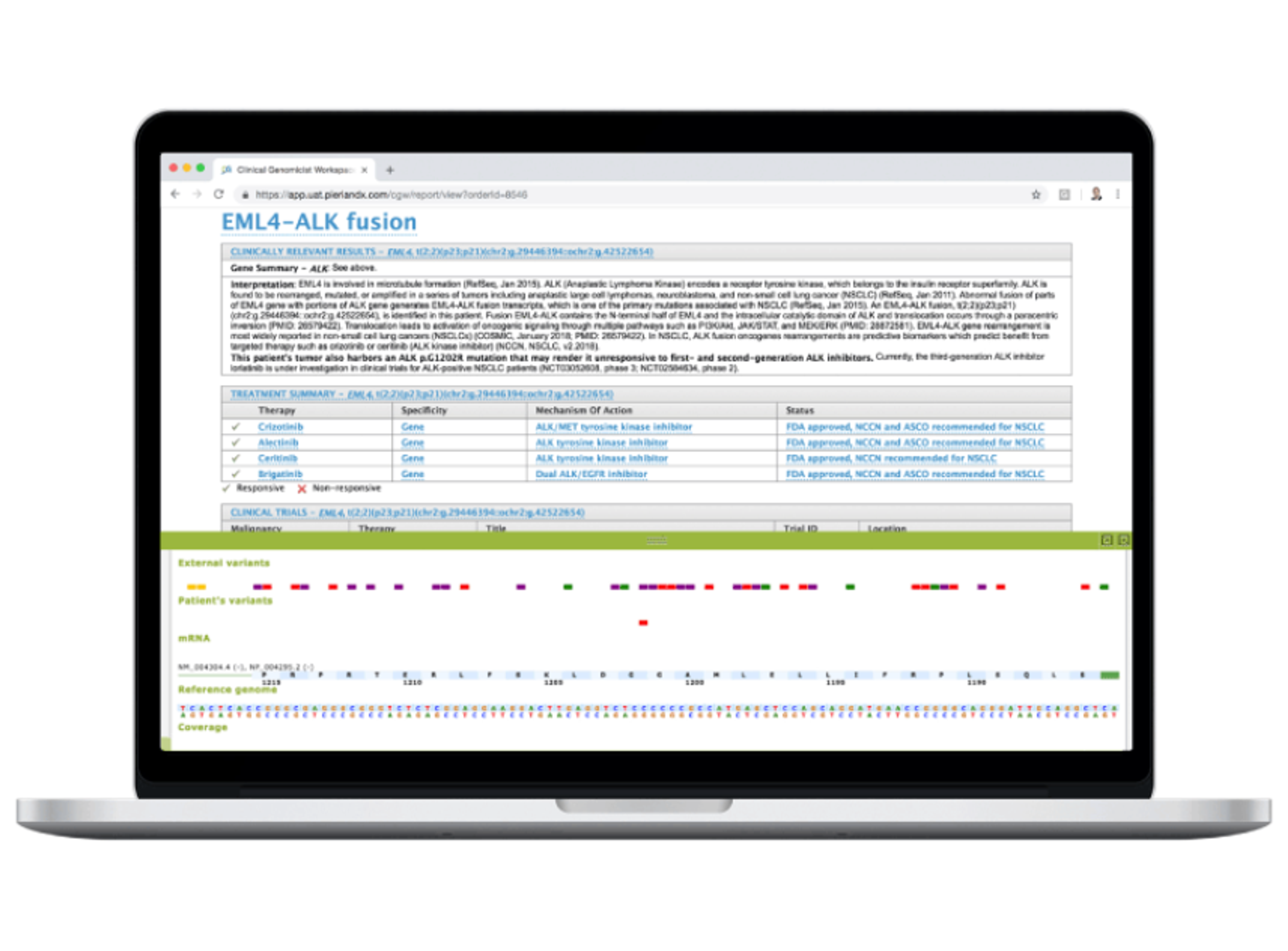
27 Feb 2025Bringing an in vitro diagnostic (IVD) solution to market requires precision, efficiency, and regulatory confidence. Discover how Velsera's advanced informatics platform and Clinical Genomics Workspace (CGW) provide a seamless, end-to-end solution for IVD development.
Explore how to:
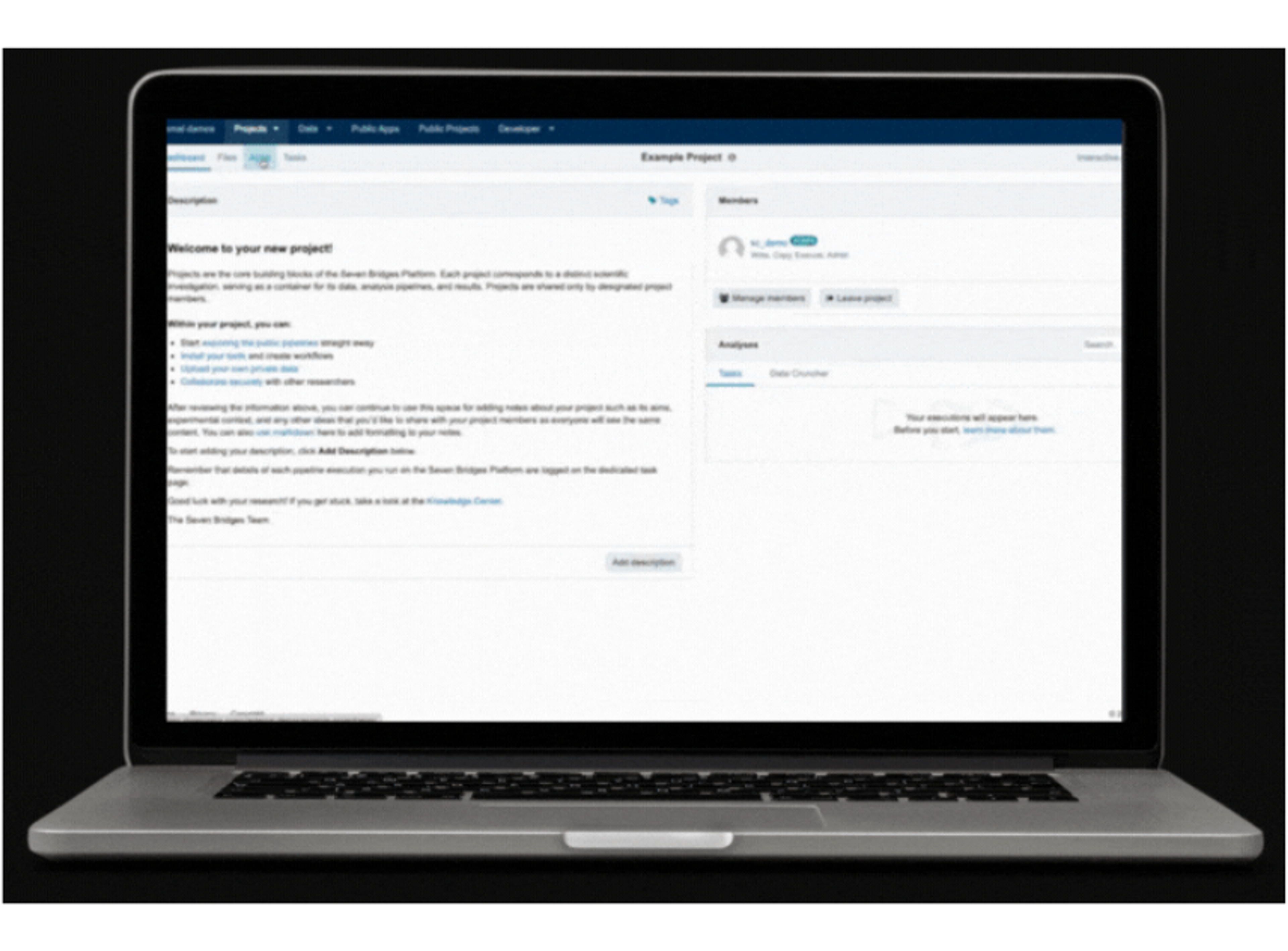
- Build and validate your workflow on the Seven Bridges analysis platform
- Deploy your IVD’ s analysis pipeline to Velsera's Platform
- Run your clinical workflow with Velsera