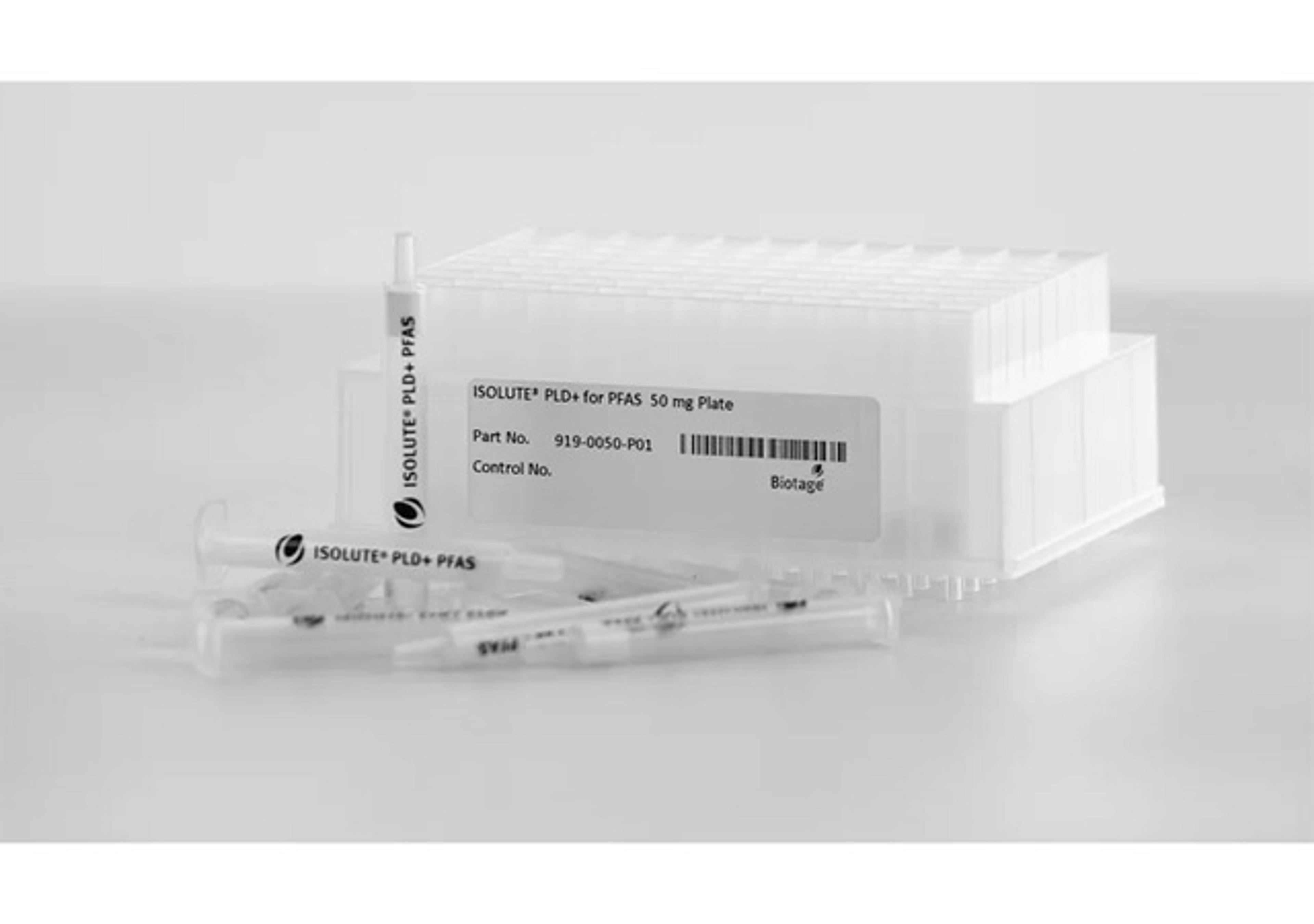
MAS-100 CG Ex® Microbial Air Sampler
The MAS-100 CG Ex® is the only microbial air sampler that enables you to test compressed gases in Zone 2 explosion hazard areas – safe, easy and accurate. Active air sampling is reliable and safe method for identifying microbial contamination in compressed gases. At numerous points in production compressed gases can come into contact with a product. Air sampling ensures a realistic total bioburden count.Microbial contaminatio…

The supplier does not provide quotations for this product through SelectScience. You can search for similar products in our Product Directory.
Great.
Air sampler in manufacturing area
Effectiveness is near 100%. It is helpful to those who want environmental air assays to maintain HACCP Plan in Industry.
Review Date: 23 Jun 2022 | Merck KGaA, Darmstadt, Germany
Very good investment.
Air monitoring
It is very easy to use. Despite being very expensive, the saving in time and materials in the long run makes it a good investment.
Review Date: 8 Jun 2020 | Merck KGaA, Darmstadt, Germany
The MAS-100 CG Ex® is the only microbial air sampler that enables you to test compressed gases in Zone 2 explosion hazard areas – safe, easy and accurate.
Active air sampling is reliable and safe method for identifying microbial contamination in compressed gases. At numerous points in production compressed gases can come into contact with a product. Air sampling ensures a realistic total bioburden count.
Microbial contamination can exist in the valves or lines that are used to feed gas and air into critical areas, such as e.g. high care foods, inert packaging lines and aseptic manufacturing environment. To ensure correct sampling results testing has to be as close to conditions of use as possible. So if the gas is injected into an inert package line or a drink at 3 bar (45 psi) then it should be tested at the same pressure at a constant air flow rate and consistent air pressure for accurate and reliable sampling. The selection of test points should be part of the overall HACCP plan following the rule of testing at the source and as near to the point of use as possible.
For the selection of a compressed gas monitor an automated operation and conversion of the compressed air or gas sample should be provided. It can be at pressures of up to 10 bar (150 psi), to atmospheric conditions after the sample has been protected in the nutrient media. Automated operation, short-duration and high-volume sampling reduces the time and effort required for monitoring and validating aseptic conditions. Furthermore, all gases used in the production environment can have different densities. The MAS-100 CG Ex® utilizes two sensors, one to measure mass flow and the other for pressure, the unit collects samples under the pressure of gas supply.
MAS-100 CG Ex® Microbial Air Sampler Features:
- Fully validated according to EN ISO 14698.
- Collects samples under gas pressure; after sampling, gradual decompression occurs, avoiding possible damage to microorganisms.
- Sampling at two different flow rates: suitable for low applications.
- Only compressed gas microbial air sampler approved for use in Zone 2 explosion hazard areas.
- Automatic calibration function: reduces risk of errors.
- Unique mass flow sensor technology: no need to calibrate for specific gases.
- Automated collection process: saves time.
- Pre-programmed for a range of different gases: no further calculations needed.
- Can store up to 10 gas protocols.
With active air monitoring products by Merck Millipore you can be sure of getting the most accurate sample solutions available. Our easy to use monitoring systems fully comply with all international standards and regulations – ensuring reproducible and audible results.