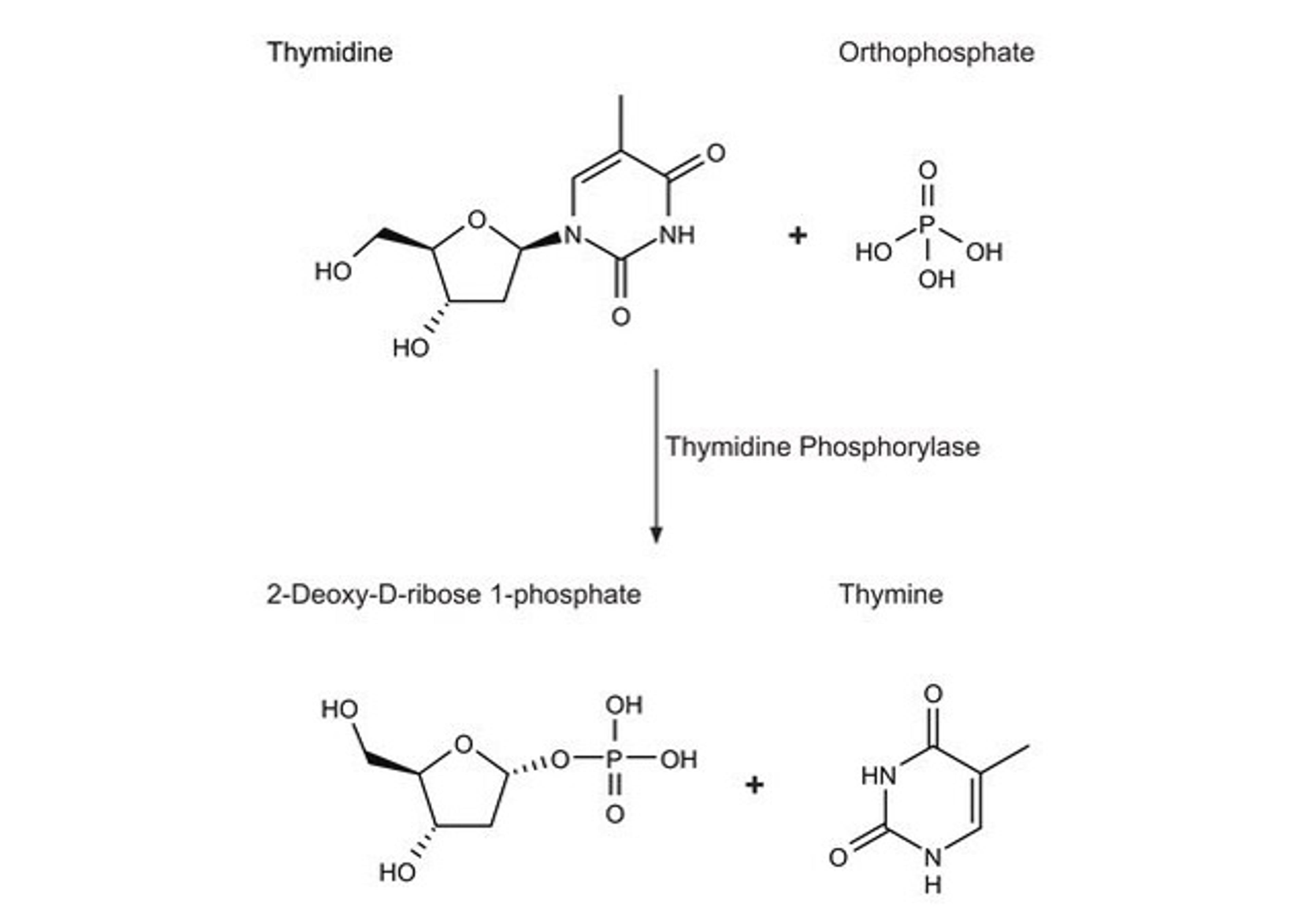
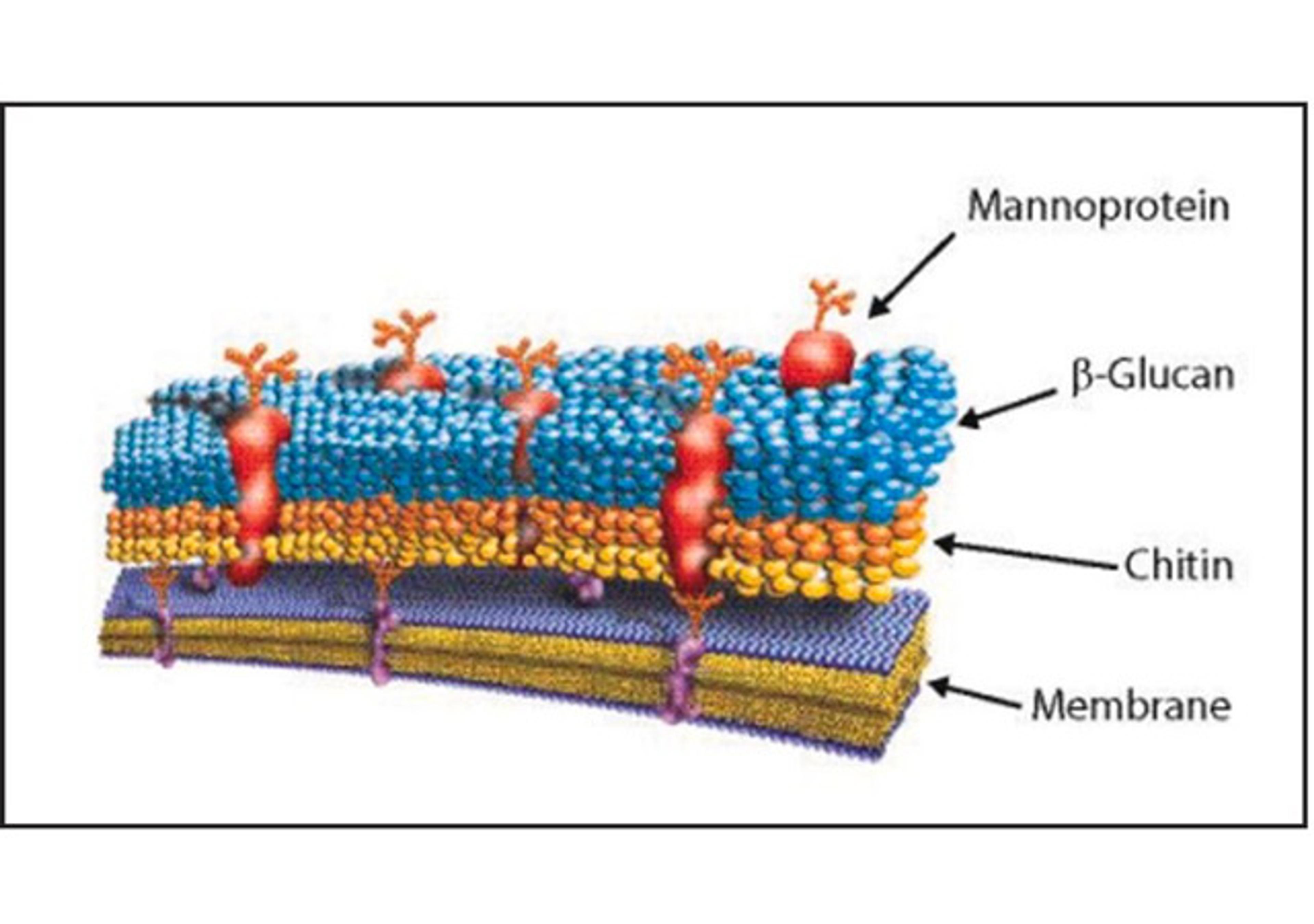
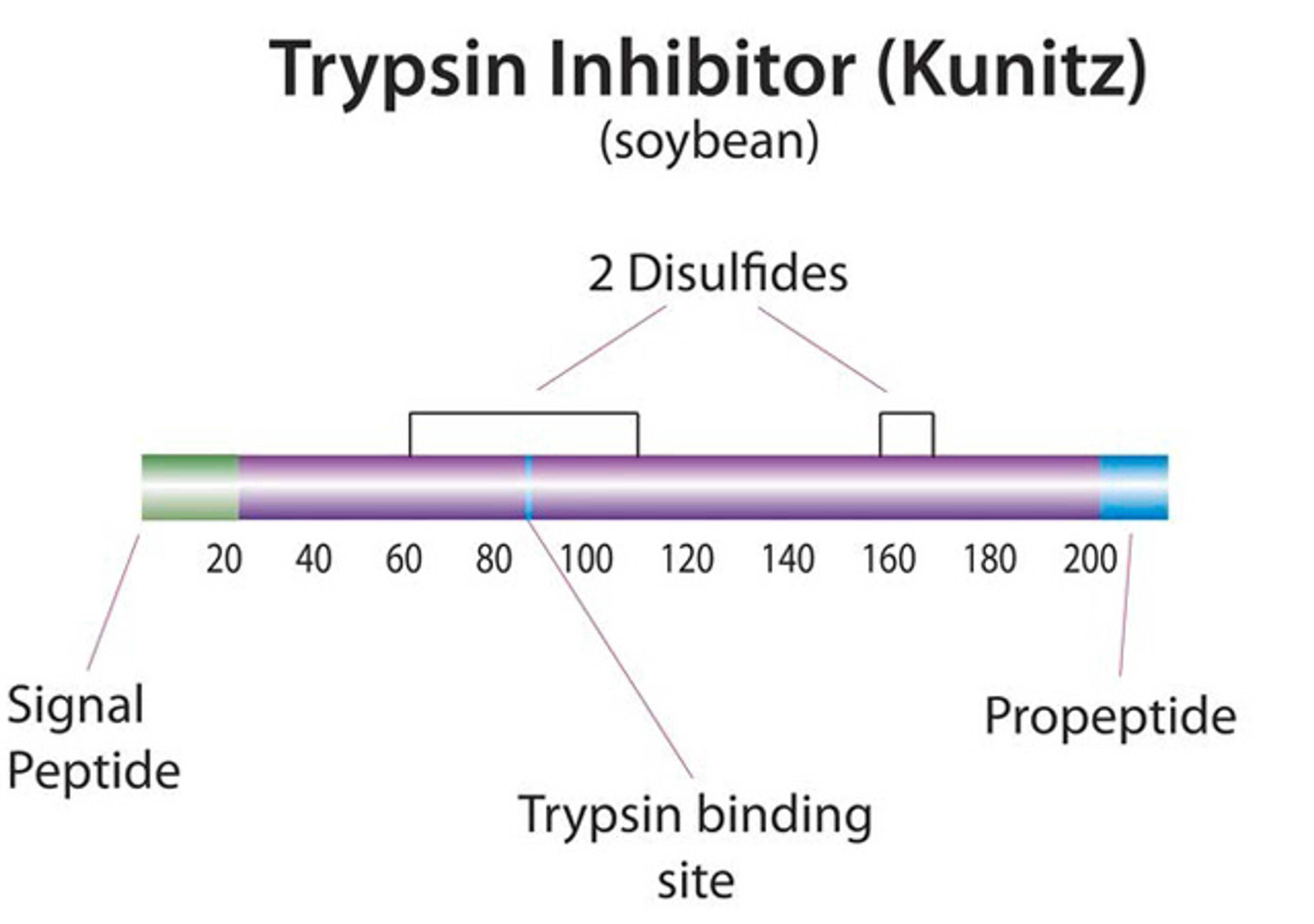
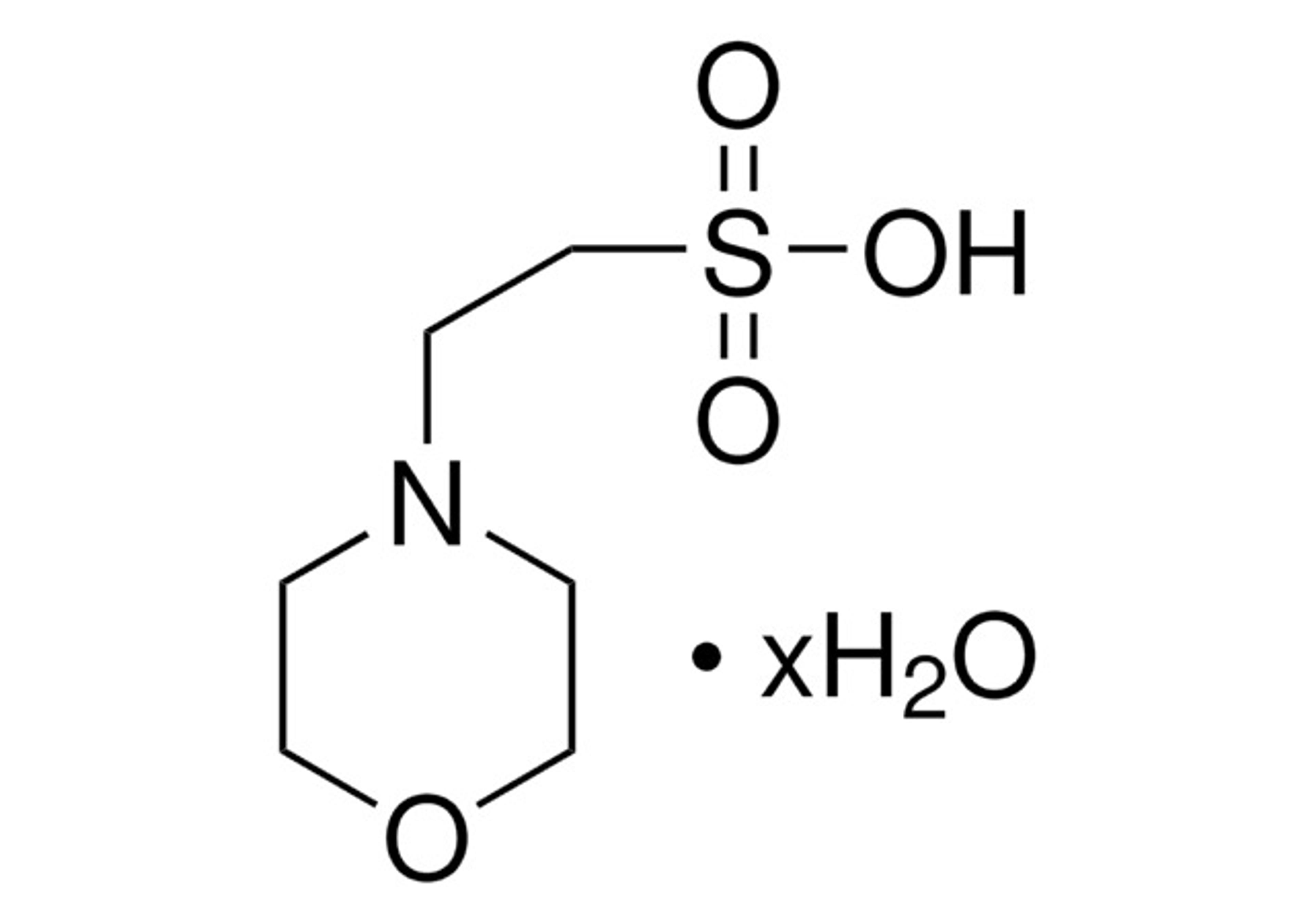
Covance Regulatory Services
Superior Regulatory Service Helps Transform Your Clinical Trials. Strong regulatory strategies are pivotal in getting products to market. While many countries’ drug regulations are similar, there are still significant differences. These similarities and differences are the basis of a global regulatory strategy that encompasses the needs of all regions. Serving you where you need to be, our Regulatory Affairs teams are located…

The supplier does not provide quotations for this product through SelectScience. You can search for similar products in our Product Directory.
Superior Regulatory Service Helps Transform Your Clinical Trials.
Strong regulatory strategies are pivotal in getting products to market. While many countries’ drug regulations are similar, there are still significant differences. These similarities and differences are the basis of a global regulatory strategy that encompasses the needs of all regions. Serving you where you need to be, our Regulatory Affairs teams are located throughout North America, Latin America, Asia Pacific and Europe.
As global regulatory requirements continue to evolve, Covance’s expertise helps you increase your chance of regulatory approval. With over 150 regulatory affairs professionals in more than 14 countries around the world, we help you navigate regulatory requirements throughout your product’s development lifecycle.
Services include:
- Course corrections throughout development when necessary
- Orchestrating Agency interactions
- Managing regulatory submissions, including the filing of global Marketing Applications
7 Ways to Reduce Inflammation Study Challenges
The following whitepaper offers seven approaches recommended by Covance to reduce common inflammation clinical study challenges. These proactive approaches are based on our experience carrying out clinical trials in this area, as well as an acknowledgement that the re-applicability of therapies to different Immune-Mediated Inflammatory Disorders (IMIDs) also enables operational improvements to the conduct of IMID clinical trials.