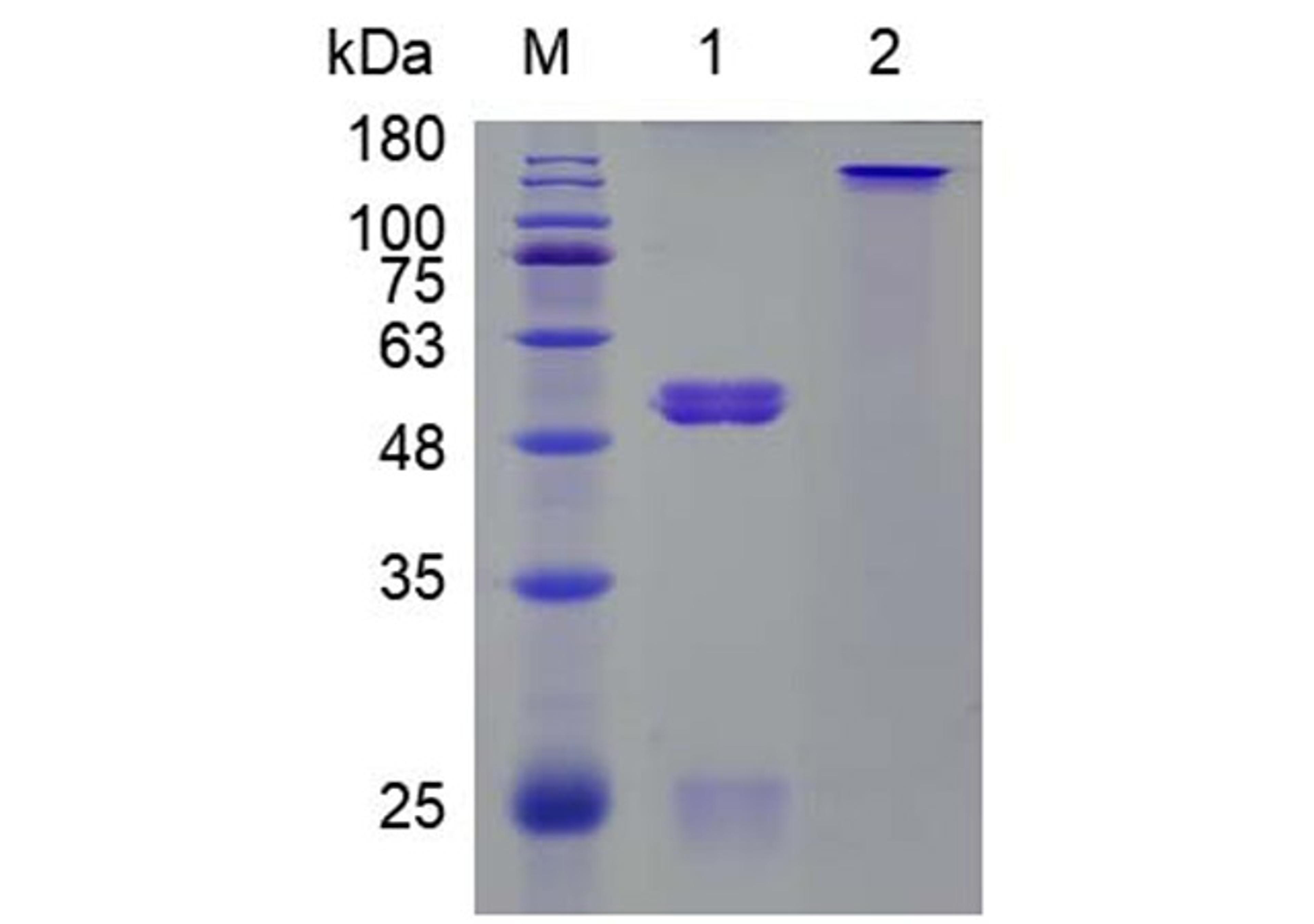
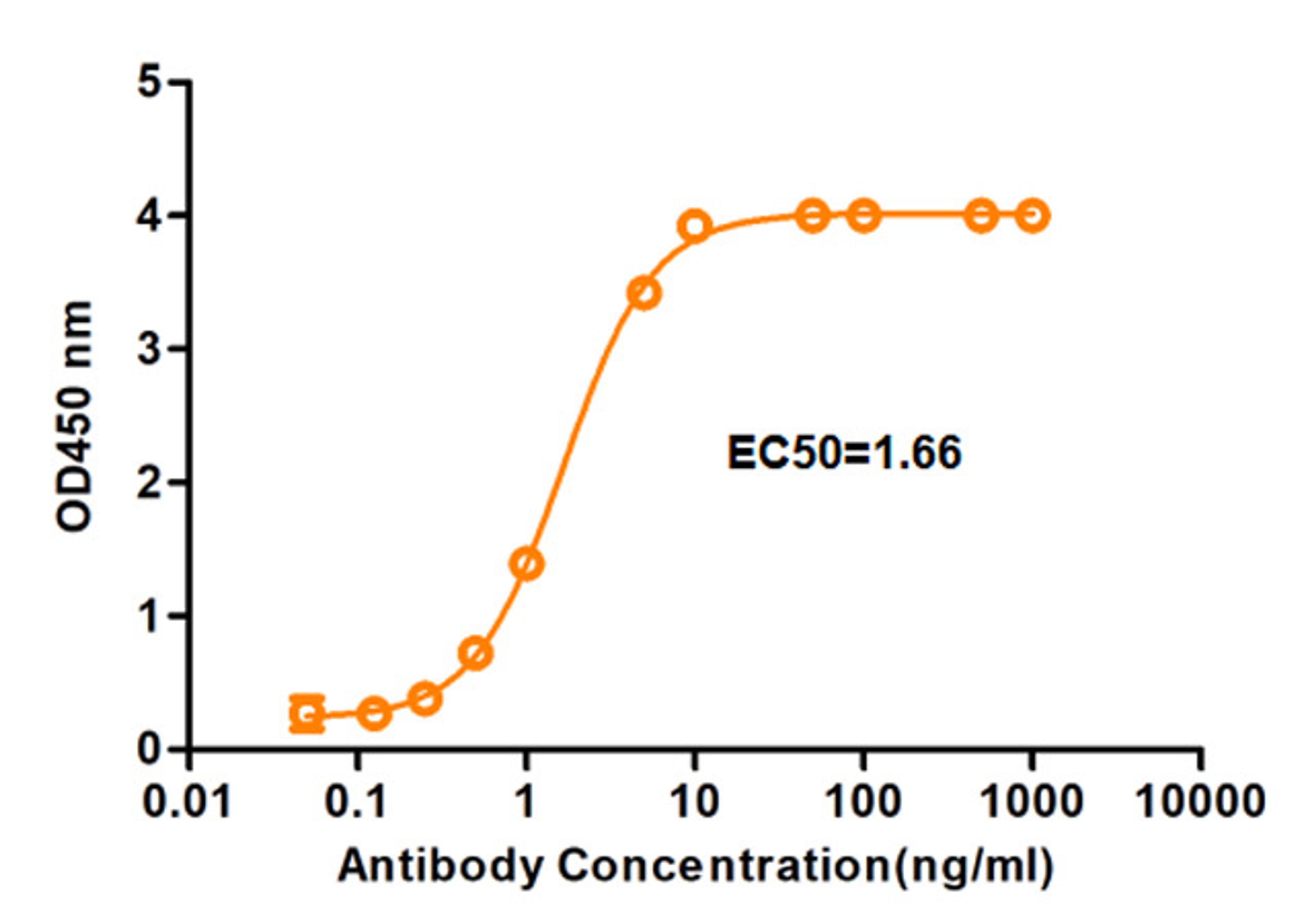
One-Stop CAR-T Therapy Development
Creative Biolabs delivers an end-to-end CAR-T development pathway—from target/scFv discovery, CAR design and vectoring, and cell engineering to in-vitro/in-vivo efficacy, IND authoring, and manufacturing support. Modular and GMP-aligned, the platform spans autologous and allogeneic routes (incl. TRAC/Cas9 and mRNA) to de-risk translation for hematologic and solid tumor programs.
Creative Biolabs delivers true end-to-end CAR-T development—spanning target/scFv discovery, CAR design and vector construction, cell engineering, in‑vitro/in‑vivo efficacy testing, IND dossier preparation, and manufacturing support—so you can streamline vendors, de‑risk tech transfer, and move faster to clinic.
Built on modular, rigorous quality practices, the program supports hematologic and solid‑tumor pipelines and accommodates advanced routes (allogeneic/TRAC‑Cas9/mRNA/γδ T) to address safety, consistency, and scalability.
Key Features / Benefits
- End‑to‑end workflow: discovery → CAR design/construction → cell engineering → in‑vitro/in‑vivo efficacy → IND → manufacturing support
- Multiple technology paths: allogeneic, TRAC/Cas9 knock‑in, mRNA/LNP, γδ T options
- Regulatory enablement: IND consulting and documentation preparation
- Modular services that integrate into existing pipelines
Relevant Applications
- Hematologic malignancy programs from discovery to IND
- Solid‑tumor CAR‑T with design optimization and robust efficacy packages
- Next‑gen modalities (mRNA/LNP, TRAC/Cas9, allogeneic) for manufacturability and access
Next-generation CAR-T design: Overcoming TME barriers with armored payloads and logic-gated precision
CAR-T therapy has already had an impact in outcome expectations in B-cell malignancies, demonstrating how engineered immune cells can leverage antigen accessibility, trafficking, and effector function for dramatic clinical benefit. In solid tumors, however, the same rules do not apply. The tumor microenvironment acts less as a passive target and more like an active defense system, characterized by stiffened extracellular matrix, aberrant vasculature, hypoxia-driven metabolic collapse, and layered immunosuppressive signaling. As a result, CAR-T programs that show strong cytotoxicity in vitro often experience attenuated trafficking, accelerated exhaustion, and functional shutdown in vivo.
In this white paper, Creative Biolabs examines how in 2025-2026 the design philosophy has changed, moving the field towards system-level CAR design. ‘Armored CAR’ concepts are now recognized as essential strategies for bridging the gap between elegant constructs and reproducible antitumor activity in complex models. Creative Biolabs highlights key R&D hotspots in engineered cell therapy, including armored CARs, logic-gated targeting, checkpoint-evasive circuits, and TME-adaptive metabolism.
From autologous to universal: Efficiency strategies to accelerate next-generation immune cell therapy (CAR-NK/iPSC) R&D
Autologous CAR-T therapies have demonstrated remarkable clinical success; however, operational friction can arise due to vein-to-vein logistics, manufacturing variability, and complex supply chains. This creates bottlenecks, limiting scalability and commercial viability. Therefore, now more than ever, operational efficiency is becoming just as critical as innovation in the field of cell therapy.
In this white paper, Creative Biolabs explores the shift from patient-specific cell therapies toward scalable, universal immune cell platforms. Universal strategies, particularly those utilizing CAR-NK and iPSC-derived immune cells, offer advantages such as improved safety profiles, manufacturing scalability and engineering potential. Creative Biolabs outlines a framework to accelerate CAR-NK/iPSC development while minimizing downstream rework, emphasizing early CMC integration, high-efficiency gene delivery and expansion, streamlined vendor workflows and pre-validated component strategy. These strategies demonstrate how cell therapy can be transformed into a repeatable product paradigm built on standardized inputs, controlled variation, and predictable outputs.