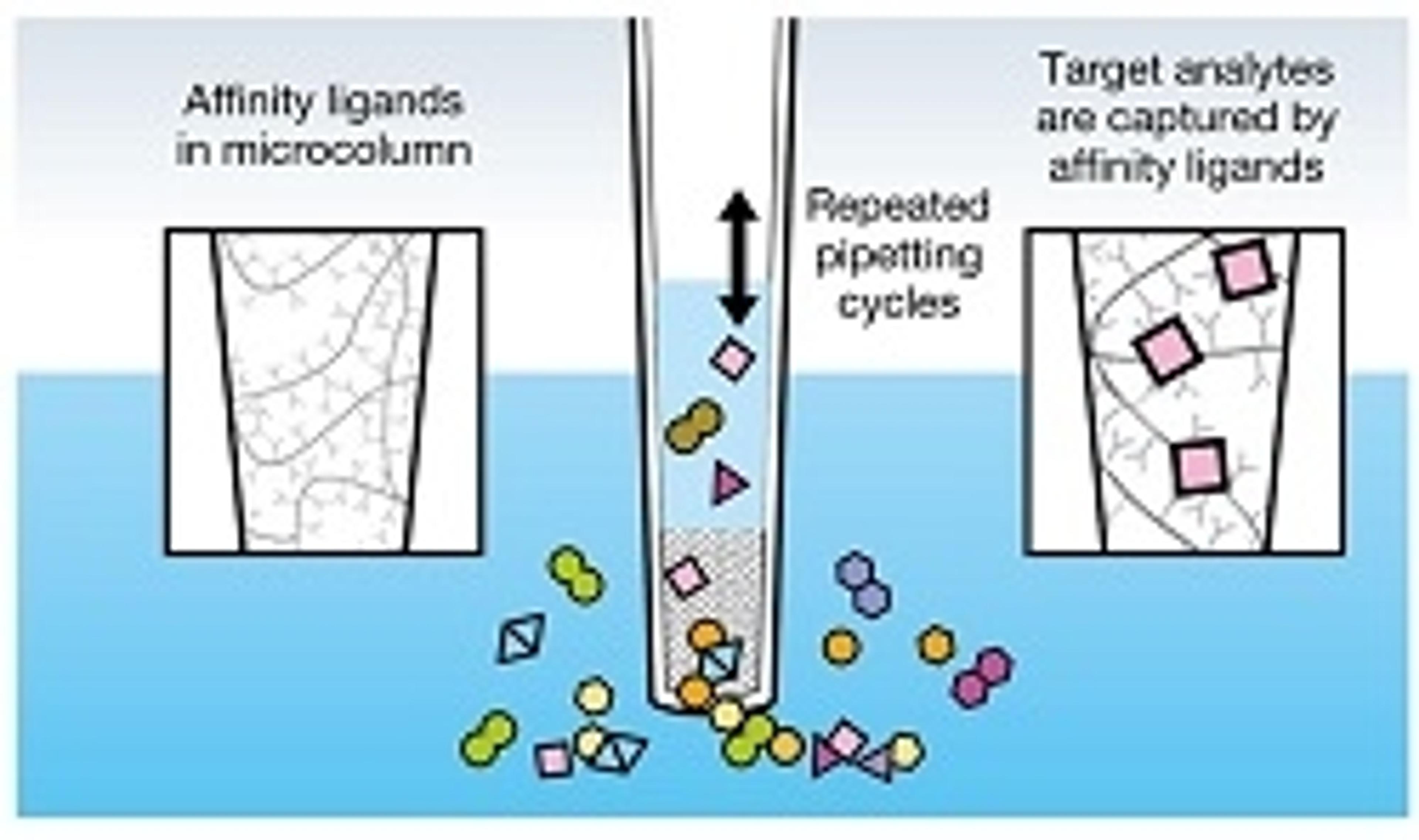
MSIA™ Microcolumns for the Freedom EVO Platform
Next generation large molecule bioanalysis made easier by combining the Thermo Scientific™ innovative MSIA™ technology with Tecan advanced, robust and easy-to-use liquid handling. MSIA affinity microcolumns, designed for Freedom EVO platform (MCA 96 head), provide innovative enablement in the areas ranging from bioanalysis of therapeutic mAbs, antibody-drug conjugates (ADC), biotransformation [in-vivo modifications, drug-an…

The supplier does not provide quotations for this product through SelectScience. You can search for similar products in our Product Directory.
Next generation large molecule bioanalysis made easier by combining the Thermo Scientific™ innovative MSIA™ technology with Tecan advanced, robust and easy-to-use liquid handling.
MSIA affinity microcolumns, designed for Freedom EVO platform (MCA 96 head), provide innovative enablement in the areas ranging from bioanalysis of therapeutic mAbs, antibody-drug conjugates (ADC), biotransformation [in-vivo modifications, drug-antibody ratio (DAR)], preclinical and clinical drug trials (PKPD, DMPK, and ADME) to biomarker and proteins analysis. This unique solution is designed to deliver the accuracy, reliability and automation you need to be successful.
Features:
- Robust Automation: Simply program the Freedom EVO to elute the analyte from complex biological samples and let the automated platform carry the load.
- Highly sensitive: Analyze proteins of interest down to femtomole level.
- Enhanced characteristics: Detects multiple protein variants for qualitative and quantitative bioanalysis in a single assay.
- Contaminant-free: proprietary molecular trapping microcolumn technology prevents matrix injection that would impact LC-MS function.
- High signal-to-noise ratio: Innovative sample interface technology reduces matrix loss, reagent carryover and non-specific binding.
- Time Savings: Microfluidic channels drive kinetic interactions significantly decreasing incubation time required to process the samples, while the MCA 96 head enables parallel processing of 96 samples.
- Reproducible: Consistent performance for assay reproducibility.
- Preserving samples: Small sample volumes (as low as 10μL) and a reduced requirement for repeating experiments.
- MSIA Streptavidin EVO microcolumns provide end user flexibility to analyze their specific targets of interest.
A Universal LB-MSIA Workflow using Freedom EVO Platform for the Quantitative Analysis of Intact Therapeutic Antibodies of Different Allotype Subclasses
This application note demonstrates the effectiveness of quantitatively analyzing therapeutic mAbs of different constructs and allotype subclasses from rodent plasma using the Thermo Scientific™ enhanced capacity MSIA™ Streptavidin EVO microcolumns. This pre-clinical bioanalytical solution, on the Tecan Freedom EVO® platform, is based on mass spectrometric detection, specific for the bioanalysis of humanized, fully human and chimeric therapeutic monoclonal antibodies.
A Universal LB-MSIA Workflow Using Freedom EVO Platform for the Pre-Clinical Analysis of Therapeutic Antibodies of Differing Allotypes in Rodent Plasma
This application demonstrates the effectiveness of analyzing therapeutic mAbs of different allotypes from rodent plasma using the Thermo Scientific™ LB-MSIA™ workflow on the Tecan Freedom EVO® platform utilizing enhanced capacity Thermo Scientific™ MSIA™ Streptavidin EVO microcolumns. This pre-clinical bio-analytical solution is based on mass spectrometric detection, specific for the bioanalysis of humanized, fully human and chimeric therapeutic monoclonal antibodies.
Learn more about Thermo Scientific™ MSIA™ Streptavidin EVO microcolumns.
Discover the Latest Developments in Biomarker Research
Next-Generation Large Molecule Affinity Purification Solution Improves Biotherapeutic, Disease Biomarker and Sports Anti-Doping Bioanalysis
Thermo Scientific MSIA Streptavidin EVO microcolumns simplify large molecule bioanalysis
What’s New in Biopharmaceutical Research?
Discover the latest technologies advancing biopharmaceutical analysis