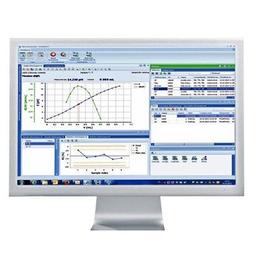
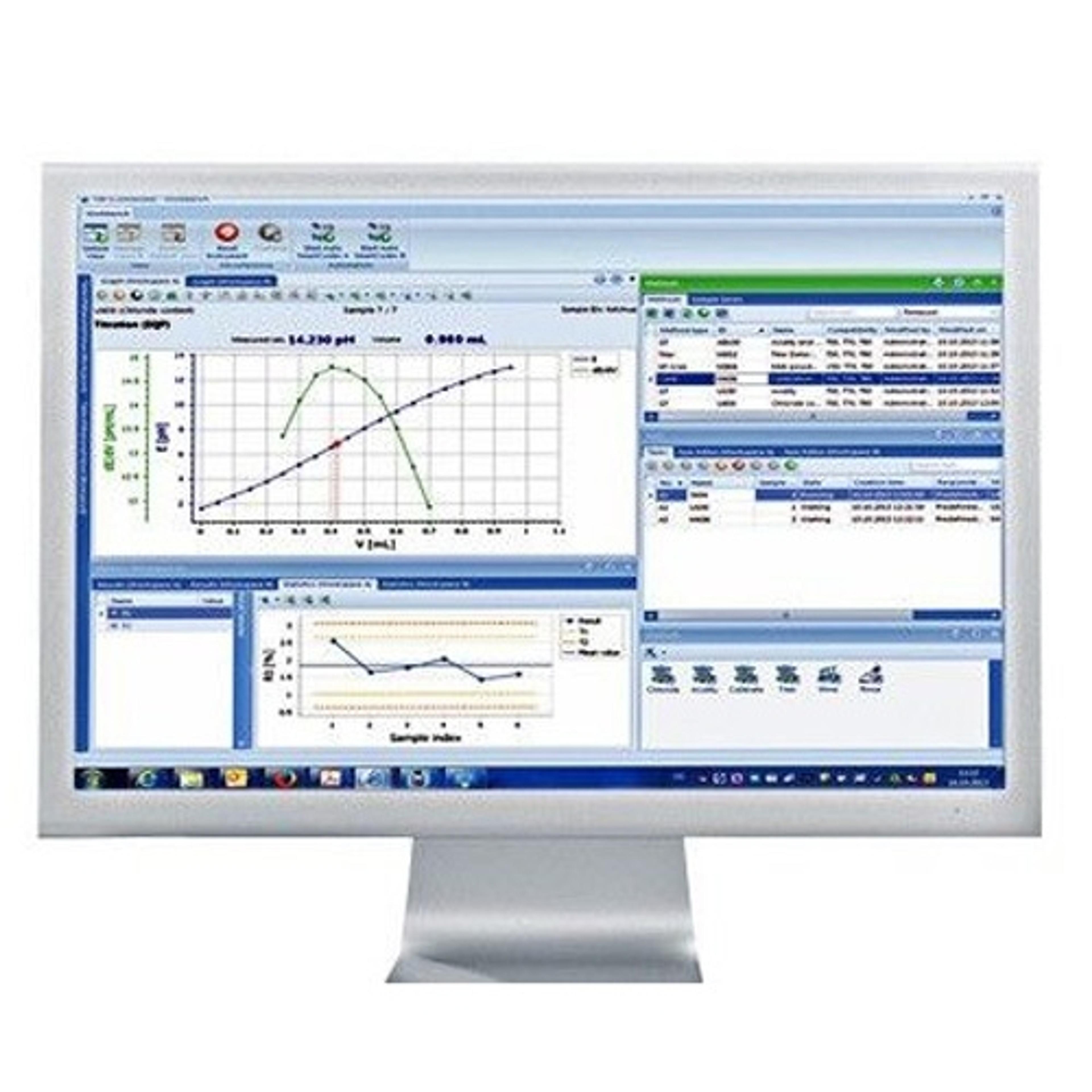
LabX™
Featured solutions for data integrity and workflow efficiency - LabX laboratory software
Great software, minor tweaks could be done to improve it.
Quaternary compound quantification
Great software. Managing results, altering raw data, titrant information, EQP set up is pretty easy, software is pretty intuitive. Gives you a whole lot more freedom with your raw data and results in terms of reprocessing, something you can't do if your device only has a terminal. Keeps an audit trail, GxP compliant in regards to that. Biggest downfall is that it cannot be operated by multiple users simultaneously.
Review Date: 2 Oct 2019 | METTLER TOLEDO
Very compliant and easy to use and control
Data integrity, pharmaceuticals
It did enable high quality data integrity throughout the instruments connected and the pre and post sales is often very high
Review Date: 11 Jul 2019 | METTLER TOLEDO
Effortless Data Integrity and central management of data, workflows and user profiles. Connect all your Excellence instruments to LabX software for full support with regulatory compliance and increased workflow efficiency. With SOP user guidance on the balance or instrument display, automated recording, calculation and transfer of data (including metadata), and a secure centralized database, LabX helps you meet MHRA and FDA 21 CFR part 11 requirements for data integrity. Use LabX as the central control point for all your laboratory instruments: titrators, balances, pH meters, refractometers, melting point devices and more.
Brochures
Seamless lab compliance for pharmaceutical manufacturers
The pharmaceutical industry is highly regulated, with standards and regulations such as good manufacturing practice becoming increasingly important. METTER TOLEDO provides an overview of the regulatory landscape related to analytical and weighing processes in pharmaceutical labs, and recommends solutions that meet standards and support compliance. The guide also highlights how the pharmaceutical industry can utilize state-of-the-art products incorporating high-tech functions and clever design details to ensure regulatory compliance throughout the entire product lifecycle.
Mastering food regulations and quality control
Food regulations aim to establish uniform food processes to ensure food trade and protect the public from illness, contaminants, and other food hazards. They address every aspect of the food supply chain. In this guide, METTLER TOLEDO explains how appropriate food safety testing and instrument choice are key to the success of any manufacturer or food testing laboratory.
Areas covered include:
- Food regulations and standards
- Understanding food regulation worldwide
- Choosing your instrument
- Common food quality control analyses
- Standards and norms by products and food categories
Case study on the determination of the acid dissociation constant
The determination of the acid dissociation constant (pKa) is an important step in the development of new pharmaceutical products. Explore the fundamentals of pKa and how METTLER TOLEDO titrations, in combination with the LabX Titration Software, allow for the measurement to be routinely performed and the data documented and exported according to GLP.
Determination of the acid dissociation constant
Understanding the acid dissociation constant (pKa) is essential for drug development, regulatory compliance, and predicting molecular behaviour in biological environments. Accurate pKa determination supports formulation decisions and helps assess how drug molecules interact with target sites under varying pH conditions.
Discover how spectrophotometric techniques enable reliable pKa measurement, supporting pharmaceutical, life sciences, and agrochemical research with precision and efficiency.
Management of weighing data: From paper to digital
It may not seem like the most exciting step, but weighing is often one of the most important parts of a lab procedure. While paper logs for recording weighing data have traditionally been the norm, some level of automated result capture and storage is desirable to help reduce the risk of errors.
In this application note, METTLER TOLEDO compares manual transcription with digital solutions for improving the accuracy and efficiency of data transfer and management, assessing the strengths and weaknesses of each approach.
The concept of 21 CFR Part 11 without a PC
In this whitepaper, METTLER TOLEDO explores the common misconceptions surrounding electronic records on standalone instruments and maintaining 21 CFR Part 11 compliance. Discover how LabX®, an integrated software solution, can ensure your electronic records are secure and validated, with minimal documentation and qualification effort.
Pharmaceutical quality control ensuring excellence and compliance
Pharmaceutical laboratories navigate rigorous guidelines and must maintain exhaustive documentation to ensure public health and meet regulatory compliance. Inefficient quality control can lead to significant operational setbacks, including product release delays, increased storage costs, regulatory penalties, and production bottlenecks.
Explore how modernized practices such as automation, data integrity, analytical service, quality control, and the importance of service and training are crucial for maintaining accuracy, efficiency, and compliance of pharmaceutical operations.
Additionally, learn about the advancements in personalized medicine and the enabling technologies that are shaping its development.
Laboratory data integrity guide
In manufacturing industries, strict regulatory oversight and Current Good Manufacturing Practice (cGMP) guidelines demand modern technologies and data integrity to pass inspections, due to potential impacts on public health. Hybrid systems reliant on manual data input risk non-compliance, while streamlined electronic processes enhance productivity and compliance. METTLER TOLEDO offers insights into navigating the regulatory landscape and improving analytical procedures for data integrity and operational efficiency.
Efficient culture media formulation
In this application note from Mettler Toledo, discover the key points in biopharma media formulation and how innovative weighing solutions can help achieve efficient results.
Data and Workflow Management in the Lab
Labs can take advantage of a variety of different levels of data capture. Each level offers a certain amount of security, error mitigation, and time savings not afforded by a primarily handwritten, paper-based system. Whether you choose to add a printer, connect to a PC, or provide full integration with a workflow and data management system such as LabX®, capturing data at the point of origin is the foundation of any effort to enhance the efficient use of data. Discover more in this guide.
TechTalk: Insights from voice-of-customer interviews on transforming labs with robotics
Monday, January 19 at 15:00 GMT | 16:00 CET | 10:00 EST | 7:00 PST
METTLER TOLEDO and ABB Robotics bring you an exclusive three-part TechTalk Series full of insights from in-depth interviews with industry leaders and experts.
Part 1: Insights from voice-of-customer interviews on transforming labs with robotics
In this kickoff session, hear from industry stakeholders about the challenges and barriers they face in transforming laboratory workflows with robotics. Discover practical strategies to turn these bottlenecks into successful projects and lay the groundwork for more flexible, future-ready labs.
Don't miss other related TechTalks in our series below over this month:
- Part 2: How to build a vision to transform labs with robotics – Explore how to create a clear and actionable vision for your laboratory transformation. Learn how to lay the foundations for successful lighthouse projects that deliver maximum impact and value.
- Part 3: How to choose the right partner to transform labs with robotics – This final session will guide you in selecting the ideal partner for your robotics initiatives. Understand how to ensure collaborations align with your goals and accelerate your lab’s journey toward a fully optimized, future-ready workflow.
Join us to learn from real-world experiences and prepare your lab for the future with robotics innovation!
Key learning objectives:
- Understand the main challenges laboratories are facing today.
- Learn from real experiences and feedback collected in the voice-of-customer interviews.
- Explore five key insights that can help guide improvements in labs.
- Find out how to lead change to support important Lighthouse projects.
- See how using digital solutions can boost ROI and long-term lab performance.
Who should attend?
This TechTalk is tailored for laboratory managers, quality managers, automation specialists, R&D leaders, operations managers, and decision-makers responsible for digitalization and automation in scientific and industrial laboratories.
Certificate of attendance
If you attend the live TechTalk, you will automatically receive a certificate of attendance, including a learning outcomes summary, for continuing education purposes.
If you view the on-demand TechTalk, you can request a certificate of attendance by emailing editor@selectscience.net.
TechTalk details
- Cost: Free to attend
- Location: Online
- Duration: 20 minutes
Registration is required to secure your place. If you register but can’t attend live, you will receive a link to the on‑demand recording once it becomes available
TechTalk: How to build a vision to transform labs with robotics
Friday, January 23 at 15:00 GMT | 16:00 CET | 10:00 EST | 7:00 PST
METTLER TOLEDO and ABB Robotics bring you an exclusive three-part TechTalk Series full of insights from in-depth interviews with industry leaders and experts.
Part 2: How to build a vision to transform labs with robotics
In this second session, we will explore how to create a clear and actionable vision for your laboratory transformation. Learn how to lay the foundations for successful Lighthouse Projects that deliver maximum impact and value.
Don't miss other related webinars in our series below over this month:
- Part 1: Insights from voice-of-customer interviews on transforming labs with robotics - Hear from industry stakeholders about the challenges and barriers they face in transforming laboratory workflows with robotics. Discover practical strategies to turn these bottlenecks into successful projects and lay the groundwork for more flexible, future-ready labs.
- Part 3: How to choose the right partner to transform labs with robotics - This final session will guide you in selecting the ideal partner for your robotics initiatives. Understand how to ensure collaborations align with your goals and accelerate your lab’s journey toward a fully optimized, future-ready workflow.
Join us to learn from real-world experiences and prepare your lab for the future with robotics innovation!
Key learning objectives:
- Understand the main challenges laboratories are facing today.
- Learn from real experiences and feedback collected in the voice-of-customer interviews.
- Explore five key insights that can help guide improvements in labs.
- Find out how to lead change to support important Lighthouse projects.
- See how using digital solutions can boost ROI and long-term lab performance.
Who should attend?
This TechTalk is tailored for laboratory managers, quality managers, automation specialists, R&D leaders, operations managers, and decision-makers responsible for digitalization and automation in scientific and industrial laboratories.
Certificate of attendance
If you attend the live TechTalk, you will automatically receive a certificate of attendance, including a learning outcomes summary, for continuing education purposes.
If you view the on-demand TechTalk, you can request a certificate of attendance by emailing editor@selectscience.net.
TechTalk details
- Cost: Free to attend
- Location: Online
- Duration: 20 minutes
Registration is required to secure your place. If you register but can’t attend live, you will receive a link to the on‑demand recording once it becomes available
TechTalk: How to choose the right partner to transform labs with robotics
Friday, January 30 at 15:00 GMT | 16:00 CET | 10:00 EST | 7:00 PST
METTLER TOLEDO and ABB Robotics bring you an exclusive three-part TechTalk Series full of insights from in-depth interviews with industry leaders and experts.
Part 3: How to choose the right partner to transform labs with robotics
This final session will guide you in selecting the ideal partner for your robotics initiatives. Understand how to ensure collaborations align with your goals and accelerate your lab’s journey toward a fully optimized, future-ready workflow.
Don't miss other related webinars in our series below over this month:
- Part 1: Insights from voice-of-customer interviews on transforming labs with robotics - Hear from industry stakeholders about the challenges and barriers they face in transforming laboratory workflows with robotics. Discover practical strategies to turn these bottlenecks into successful projects and lay the groundwork for more flexible, future-ready labs.
- Part 2: How to build a vision to transform labs with robotics - Explore how to create a clear and actionable vision for your laboratory transformation. Learn how to lay the foundations for successful Lighthouse Projects that deliver maximum impact and value.
Join us to learn from real-world experiences and prepare your lab for the future with robotics innovation!
Key learning objectives:
- Understand the main challenges laboratories are facing today.
- Learn from real experiences and feedback collected in the voice-of-customer interviews.
- Explore five key insights that can help guide improvements in labs.
- Find out how to lead change to support important Lighthouse projects.
- See how using digital solutions can boost ROI and long-term lab performance.
Who should attend?
This TechTalk is tailored for laboratory managers, quality managers, automation specialists, R&D leaders, operations managers, and decision-makers responsible for digitalization and automation in scientific and industrial laboratories.
Certificate of attendance
If you attend the live TechTalk, you will automatically receive a certificate of attendance, including a learning outcomes summary, for continuing education purposes.
If you view the on-demand TechTalk, you can request a certificate of attendance by emailing editor@selectscience.net.
TechTalk details
- Cost: Free to attend
- Location: Online
- Duration: 20 minutes
Registration is required to secure your place. If you register but can’t attend live, you will receive a link to the on‑demand recording once it becomes available
UV7+LabX: Easy and efficient compliance
Easy Compliance. All the way. Every day
Does Your Lab Lack the X Factor?
Discover why integrated laboratory software is said to be a ‘must-have’ for the modern laboratory that wants to remain competitive