Bioprocessing Salt Active Nucleases - Physiological Conditions
Nonspecific endonuclease is highly active at physiological conditions (125–200 mM) for DNA removal during protein production, vaccine manufacturing, and viral vector preparation. M-SAN High Quality can be used directly in cell media, which improves efficiency and yield. Designed to replace Benzonase without workflow changes. M-SAN HQ excels in ensuring higher purity and efficiency. High purity (≥ 99%). Compatible with ELISA ki…
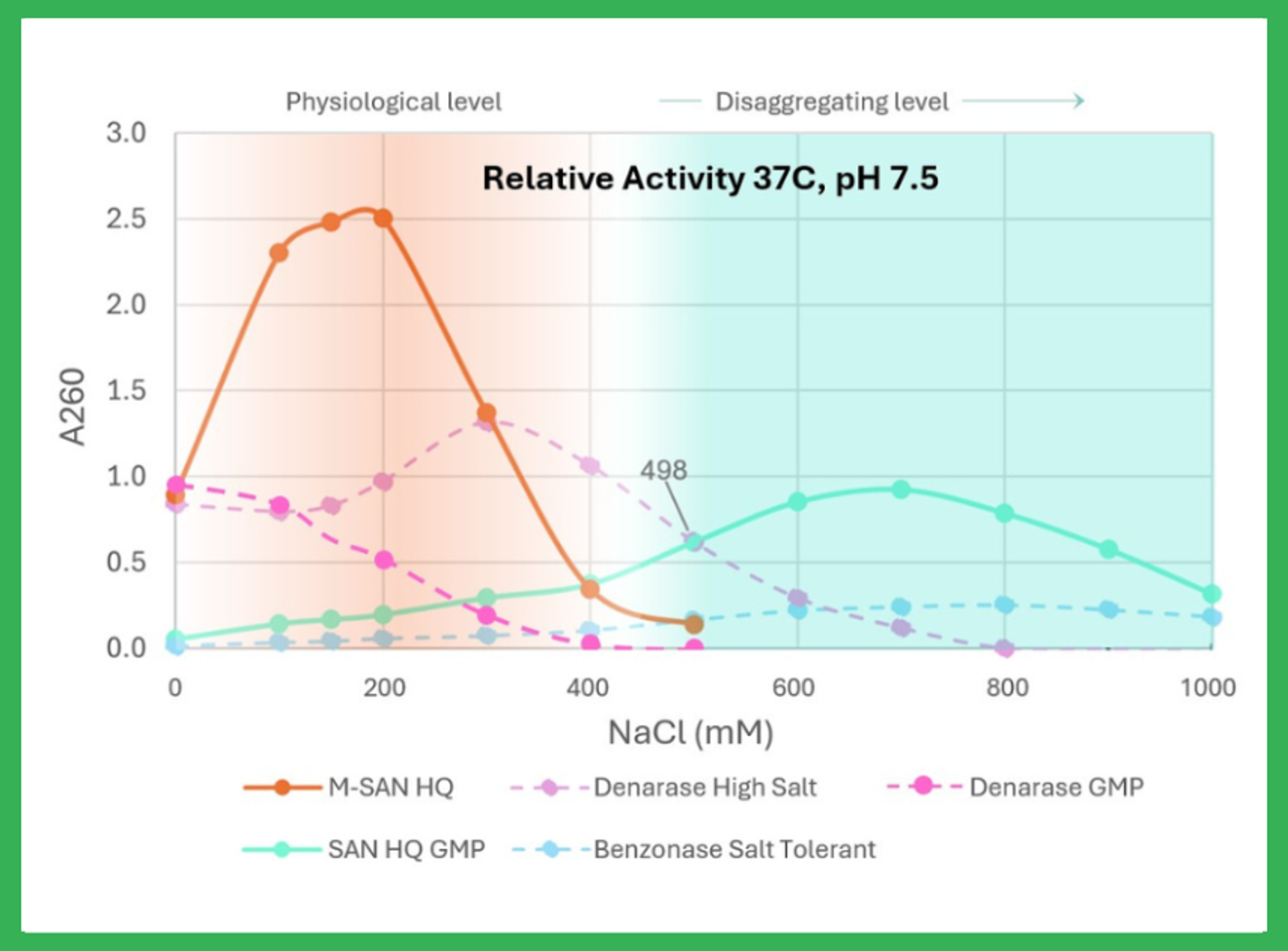
This novel, nonspecific endonuclease is active over a broad pH range and displays optimum activity at salt concentrations between 125 – 250 mM. Due to its excellent performance at physiological conditions, M-SAN HQ can be used directly in the cell medium or the harvested supernatant without buffer adjustments. This makes M-SAN HQ ideal for the manufacturing of fragile vectors such as lentiviruses and retroviruses.
M-SAN HQ can be directly used in medium without buffer adjustments The high activity of M-SAN HQ at standard cell medium conditions leads to improved DNA clearance compared to other commonly used nucleases. In the data, an over 2-fold reduction in residual DNA was achieved.
Key Benefits
- Compatibility: Ideal for working with both fragile and robust viral vectors and proteins in a variety of cell media
- Optimum activity: Optimization for cell media salinity allows both shorter DNA fragments and reduced incubation times.
- Cost-Effectiveness: Reduced need for additional reagents and steps can lower production costs
- Quality: Maintains the integrity of sensitive or labile biological molecules, ensuring a higher quality end product
- Flexibility: Can be used directly in a variety of media without the need for customization
Key Features - High purity (≥ 99%)
- No protease detected
- Endotoxin-tested
- Animal origin-free production
- Supplied with extended product documentation
- ELISA Kit available
Rethinking nuclease strategy to improve yield, product quality, and manufacturing economics in viral vector production
Viral vector manufacturing is at an inflection point. As demand accelerates, manufacturing cost and scalability are increasingly limiting patient access to life-saving therapies. Despite extensive optimization of upstream productivity and downstream unit operations, nuclease strategy remains an under-exploited lever with impact on yield, cost per dose, and product quality profile.
This white paper analyzes how optimized nuclease selection transforms viral vector manufacturing economics. Using experimentally validated data and cost modeling across AAV and lentiviral workflows, it shows how SAN HQ and M-SAN HQ enable superior chromatin clearance under process-relevant salt conditions, doubling recovery, improving downstream robustness, reducing nuclease costs by over 70%, and lowering cost of goods per dose by approximately 40%.
Smarter cleanup for lentiviral manufacturing
Discover how ArcticZymes Technologies' M-SAN HQ nuclease enhances lentiviral vector manufacturing by efficiently removing host cell DNA, including chromatin, under physiological salt conditions. This simple yet powerful solution helps improve vector yield, streamline downstream processing, and reduce regulatory risks.
Advanced nuclease applications in lentiviral vector bioprocessing
Explore how alternative nucleases, salt active nuclease (SAN) and medium-salt active nuclease (M-SAN), are evaluated in lentiviral vector (LV) manufacturing to improve gene therapy product quality. Compared to traditional nucleases, SAN and M-SAN achieved enhanced vector recovery, reduced aggregation, and significantly lower residual DNA in the final product. These findings support the use of alternative nucleases to address critical quality needs in LV manufacturing.
Efficient chromatin removal in viral vector manufacturing using salt-active nucleases
The first enzymatic solution for complete removal of chromatin at physiological salt conditions and its effect on down stream processing