Aesica Formulation Development Service
Formulation Development SpecialistsWith a keen understanding of early stage development and clinical trial requirements, Aesica guides their customers through the processes of new medicine and material development for Phase 1 and Phase 2 clinical trials. They understand that their customers also need to maximise their budget effectiveness by minimising the amount of experimentation without sacrificing quality levels.Typical do…

The supplier does not provide quotations for this product through SelectScience. You can search for similar products in our Product Directory.
Formulation Development Specialists
With a keen understanding of early stage development and clinical trial requirements, Aesica guides their customers through the processes of new medicine and material development for Phase 1 and Phase 2 clinical trials. They understand that their customers also need to maximise their budget effectiveness by minimising the amount of experimentation without sacrificing quality levels.
Typical dosage forms we work with include tablets, capsules, liquids, suspensions, creams, ointments, inhaled and sterile pharmaceuticals. Aesica offer a suite of formulation development services…
- Pre-formulation
- Formulation and manufacturing
- Analysis and stability testing
- Manufacturing of clinical trial materials
- Regulatory support
- Technology transfer
Aesica Formulation Development Service Benefits:
- Working with a fast-growing UK company with an expanding international presence
- Working with a flexible, dynamic, innovative organisation
- Working with full service development and manufacturing capability – from process development to commercial scale manufacture
- Working with a partner who can be trusted to deliver the highest standards in all areas: consultation, development, manufacture and delivery
Practical Solutions for Poorly Soluble Drugs
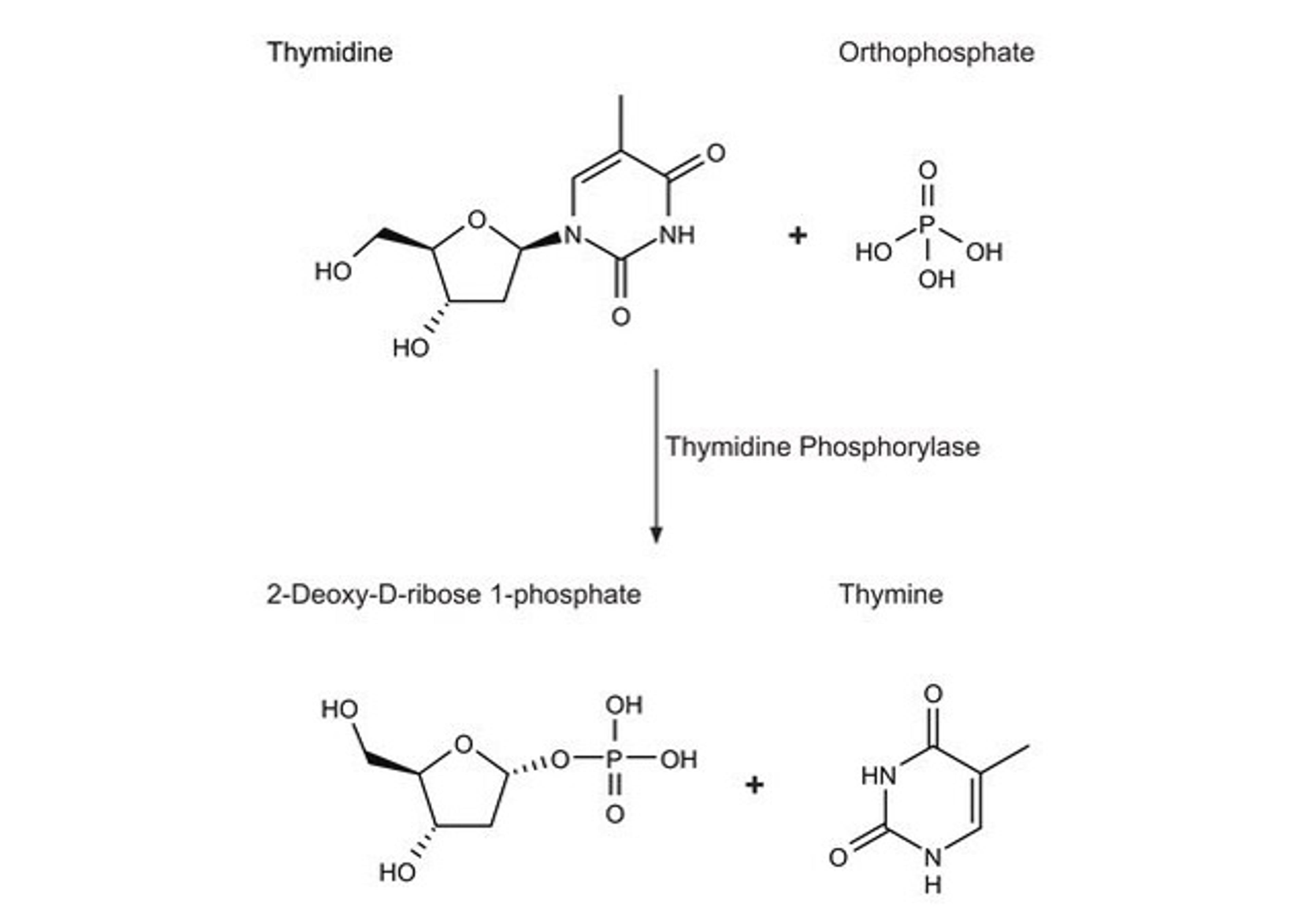
The therapeutic efficacy of a drug depends upon its bioavailability, which is directly correlated to its solubility. Many drugs, both in development and on the market, are poorly soluble in aqueous media, which can lead to poor bioavailability, and frequently results in variable dissolution rates. To achieve the desired drug concentration in systemic circulation to elicit a pharmacological response, solubility is paramount. Yet, less than 10% of new drug candidates demonstrate both high solubility and permeability, and 30–40% of the drugs that appear on the World Health Organisation (WHO) Essential Drug List were reported to be poorly water-soluble or lipophilic, based on the Biopharmaceutics Classification System (BCS). This whitepaper discusses laboratory scale formulation expertise and equipment in three distinct technologies.
Improving Drug Delivery Systems with Novel Emulsion Technologies for Sterile Product Formulation
This whitepaper discusses micro-encapsulation as one of the most interesting areas in modern pharmaceutical technology. It is a complex, interdisciplinary field requiring specialist knowledge of polymer science and familiarity with emulsion technology. It includes the process in which tiny particles or droplets are surrounded by a coating to create small microcapsules that contain a multitude of critically useful properties. Due to its complexity, microencapsulation is extensively studied inside major pharmaceutical companies, universities and research institutes. Polymeric drug delivery devices are focused on the encapsulation of large molecules including peptides, proteins and DNA/RNA for potential use as vaccines and as long acting release drug formulations. Moreover, encapsulation is used for controlling the release of water soluble drugs. Demand for the process has led to the creation of advanced emulsion solvent evaporation/extraction based micro-encapsulation technologies.