The iKnife: Transforming the Cancer Surgery Paradigm
This intelligent surgical device is a cornerstone in the surgical removal of cancers
1 Oct 2018
With the potential to revolutionize cancer surgery and diagnostics, the iKnife has generated excitement in the field of clinical oncology. This intelligent surgical device uses electrospray ionization coupled to mass spectrometry to distinguish between cancerous and non-cancerous tissue types in real time. In an interview with Dr. James Kinross, consultant colorectal surgeon and senior lecturer at Imperial College London, where the technology was invented, SelectScience finds out more.

Please introduce yourself and your area of work
I am a senior lecturer in surgery at Imperial College London. I work as an NHS colorectal surgeon at St. Mary’s Hospital in London, and I specialize in minimally invasive surgery for colorectal cancer. I also lead a research group studying translational applications of metabolomics and the gut microbiome in surgery and cancer.
Could you explain what the iKnife technology is, and how it works
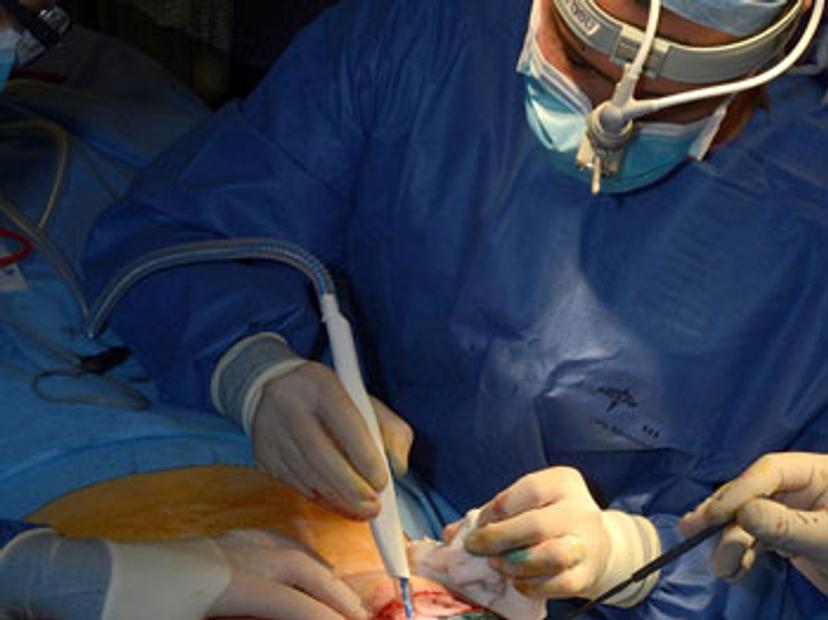
Modern surgery relies on a number of different ‘energy devices’ for tissue dissection. These are favored over blades, as they coagulate and seal tissues to prevent bleeding. A by-product of these instruments is smoke which is typically discarded. The iKnife works by aspirating this smoke into a mass spectrometer during surgery and analyzing the many thousands of chemical features that reside within it.
The technology, called rapid evaporative ionization mass spectrometry (REIMS), has been developed in partnership with Waters, after being invented by Imperial professor, Zoltan Takats. After the smoke has been aspirated, we then apply machine learning tools to the raw data to search for patterns of molecules against large databases of previously validated spectra. We can do this very quickly, in a few hundredths of a second, meaning the surgeon will know in near real time about the tissue they are dissecting. For example, if the tissue is cancerous or not. We can do this with a very high degree of accuracy.
How could this technology aid in the early diagnosis and stratification of cancer patients, to improve patient outcome?
We have found that this biological smoke gives us lots of important information about the molecular biology of specific tissue types, including the microbiology. The instrument is very flexible, and it works with all types of energy device in both open and ‘keyhole’ surgery. Going forward, this technology could enable the use of minimal amounts of energy, or very accurate energy devices (like lasers), to sample tissues in vivo for diagnostic purposes.
For example, we are developing an endoscopic application for diagnosing polyps found at colonoscopy in real time using chemical histopathology. Since the chemical information is so rich and it reflects the genetic and molecular functions of cancers, it also means that this information could be used to guide medical treatment, not just surgery. We could identify which drugs are likely to be effective in certain tumor types, and this could even work in the clinic as a bedside diagnostic instrument.
How far away are we from implementing this technology into everyday practice?
The technology has taken a long time to develop as we have to follow some pathways to regulatory approval. At the current stage we have a funded trial in breast cancer surgery, and the feedback from the surgeons suggests that the device is well accepted. However, the clinical decision-making progress won’t be altered until we have a very good idea about the diagnostic strength of the device in vivo. We have trials in a large number of other diseases: prostate, brain, ovarian, and colorectal cancers for example, and we are developing further applications in endoscopy and robotics. It has been very successful so far, and it seems to capture both the patient’s and clinician’s imagination. The technology has the potential to change the surgical paradigm.
What’s your vision for the future of oncology?
This is a tough question! Obviously, the direction of travel is in the personalization of cancer treatment, and this will involve the scaling of therapeutic technologies such as immunotherapy. Surgery is at an inflection point, and within 20 years, it will be data driven; this means surgeons will use robotics linked to deep learning of their procedures in real time to improve the precision of their care. With this is mind, technologies like the iKnife, which provide biological feedback, will be ingrained in clinical practice.
But the real gains will be in the prevention and early detection of cancer. Again, I think artificial intelligence is going to have a very significant impact on the automated detection of precursor lesions in screening programs and there will be a concerted push to create technologies that target an individual’s risk of cancer.
Do you use Waters' mass spec instruments? Write a review today to be in with a chance of winning a $400 amazon voucher

