From cannabis to COVID: Discover the power of RNA in situ hybridization
Dr. Jeremy Hirota takes us through the technology behind his translational research program for respiratory health
12 Aug 2020

In this expert interview, we hear from Dr. Jeremy Hirota about his work in the Hirota Lab at McMaster University and how he is working to develop an internationally recognized translational research program for respiratory health, focusing on lung health and disease.
The Hirota lab uses a research strategy involving in vitro cell culture models, in vivo pre-clinical models, and clinical studies. “The research of the lab is guided by three mutually reinforcing foci,” Hirota explains, “patient-oriented research on respiratory mucosal immunology in health and disease; small molecule drug discovery programs for commercialization opportunities; and pure basic science characterization of the biology behind innate immune receptor and related signaling pathways.”
RNA ISH for cannabis and COVID-19
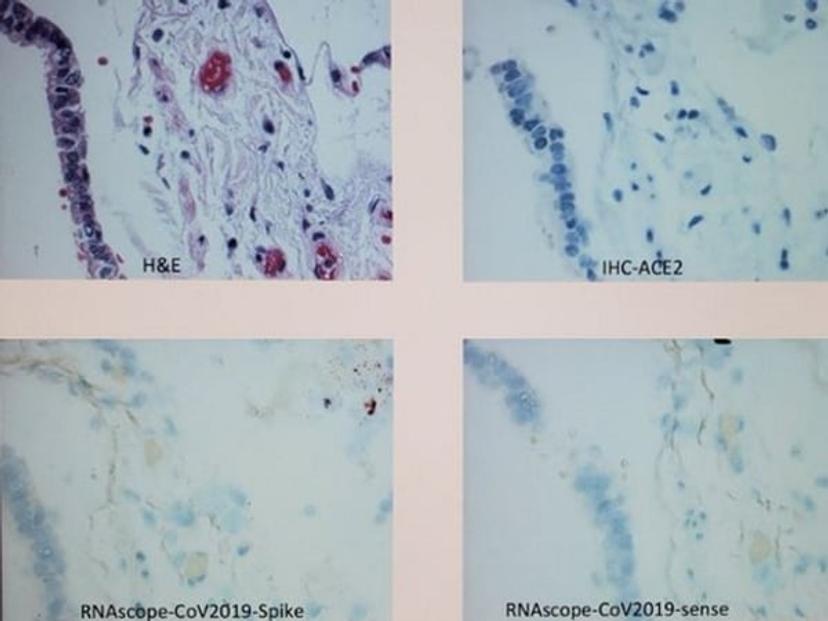
The Hirota Lab has published an impressive volume of peer-reviewed research relating to precision medicine, cell transporters, cannabis, tissue engineering, and most recently COVID-19. An invaluable tool throughout its work has been the RNAscope technology from Advanced Cell Diagnostics, which has been used for research into a variety of biomarkers. This RNA in situ hybridization multiplexing technology has enabled the profiling and identification of various cell transporters and receptors in human lung tissue. A single cell can be identified via a specific marker in one channel, whilst concurrently looking for the co-expression of other target molecules in the other available channels. This molecular approach complements traditional pathology that uses serial sections of H&E (hematoxylin and eosin) histological stains for tissue morphology assessment and cell identification, followed by staining for molecules of interest.
“We have used RNAscope to confirm the in situ presence of gene transcripts for our molecules of interest,” Hirota explains. “However, we are only just beginning to realize the potential of this technology and hope to leverage this further in the future.”
The Hirota lab has used this approach to provide evidence for the expression of cannabinoid receptors 1 and 2 in human airway epithelial cells at the gene level, and, due to the team’s experience working with airway epithelial cells and lung immunology, it has applied these learnings to help tackle SARS-CoV-2. With access to COVID-19 autopsy lung tissue samples, the lab is now assessing candidate SARS-CoV-2 receptors to identify respiratory mucosal immune responses that are predictive of negative SARS-CoV-2 infection outcomes. Regarding this current work, Hirota shares that: “It appears that the SARS-CoV-2 virus is present in areas of lung tissue that do not express the proposed receptor, ACE-2. This suggests that alternate receptors for the virus, or other mechanisms for virus entry, exist.” The lab is currently working to confirm and subsequently publish this work.
Working in the pandemic and beyond
Related publications
- Gene expression and in situ protein profiling of candidate SARS-CoV-2 receptors, available here
- Isolation, sequence, infectivity, and replication kinetics of SARS-CoV-2, available here
Hirota explains how due to the ongoing pandemic his team has needed to find new ways to work collaboratively, around the clock, and ensure that it is following the recommendations designed to limit the spread of SARS-CoV-2, whilst pushing forward COVID-19 research that aims to help patients, healthcare systems, and governing bodies. “The current environment is constraining,” says Hirota. “We are following social distancing policies in the workplace and currently have limited numbers of staff in the labs.”
Looking to the future, Hirota sees several opportunities for exciting advances in precision medicine treatments and diagnostics, including the merging of molecular technologies, well-phenotyped patient samples, and improved informatics technologies. “We hope to be at the forefront of this work,” Hirota concludes, “working with academic and industry partners to leverage respective strengths and ensure that our research makes it to market to improve health and wellbeing on a global scale.”
Find out more about RNA ISH capabilities with these expert interviews and resources:
- Plus, download your free application eBook on RNA ISH single-cell analysis

