Understanding NMR-based breast cancer metabolomics and biomarkers discovery
Prof. Tone Frost Bathen explores how to enhance clinical research through standardization, validation and data sharing
28 Jun 2020

Despite progress in early detection and therapeutic strategies, breast cancer remains a leading cause of cancer-related death among women globally. Due to the heterogeneity and complex tumor biology, breast cancer patients with similar diagnoses might have different prognoses and responses to treatment. Metabolomics is the branch of “omics” technologies that involves high-throughput identification and quantification of small-molecule metabolites in the metabolome. Cancer cells must be able to convert nutrients to biomass while maintaining energy production, which requires reprogramming of central metabolic processes. This phenomenon is increasingly recognized as a potential target for treatment, but also as a source for biomarkers that can be used for prognosis, risk stratification, and therapy monitoring.
In this SelectScience® webinar, now available on demand, Tone Frost Bathen, Head Professor at the MR Cancer Group, Norwegian University of Science and Technology (NTNU), introduces NMR-based metabolomics research in breast cancer, covering aspects in the analytical pipeline to enable identification of prognostic and predictive biomarkers.
Read on for highlights from the live webinar Q&A session and register to watch the webinar on demand.
Watch Webinar NowQ: Are your tissues preserved in formalin?
TFB: Our tissues are not preserved in formalin. For this analysis, we use fresh frozen tissue with no preservatives at all. Formalin would disturb peaks in the NMR spectra. I know there have been tests done to remove formalin from the samples, but that also decreases the concentration of metabolites in the sample. As a result, tissue samples should be blast frozen directly after obtaining them.
Q: What sort of throughput are you able to achieve with the tissue samples?
TFB: The magic-angle spinning approach is still quite a manual approach because you have to individually test and prepare the samples. The acquisition of one sample, depending on the size and the concentration of metabolites, will take 30 minutes to an hour. This gives us a sample throughput of 8 to 12 samples a day. Magic-angle spinning analysis with the equipment we have is still challenging but I know there are now new approaches that make this simpler.
Q: As a non-descriptive technique, how does this compare with descriptive MS/MS techniques?
TFB: If we compare to mass spectrometry, the main advantage of using NMR is that you don't need to treat the sample in any specific way. You just cut it, add some buffer, and then it's ready for analysis. But of course, NMR spectroscopy is much less sensitive for metabolite detection compared to mass spectrometry. However, NMR is, by nature, a more quantitative method. It's also robust and quite reproducible. For mass spectrometry, there are now approaches where you can use the infected tissue samples for mass spectrometry imaging. You also use fresh-frozen tissue for that, but the sample will be partially destroyed by the lasers applied during mass spectrometry imaging. I would say that both methods have their pros and cons and you need to optimize depending on what you really want to detect. Both are in a way complementary, and in some of our studies we combine the two approaches.
Q: How does this NMR technique compare with immunohistochemistry (IHC) of biopsy samples?
TFB: We've used IHC after many analyses to make sure of the accuracy of what we have analyzed. Sometimes, when we receive a biopsy or a surgical sample, it may not contain tumor cells at all. Another way we use IHC is to investigate how metabolism differs between samples that are acute and receptor-positive or negative. For some studies where we've tried to establish biomarkers, the total cancerous or tumor tissue in the sample or tumor cells in the samples may vary, but it also seems like metabolite concentrations are much lower than in normal tissue. If tissue analysis is to be used to establish prognostic or predictive factors, it has to be able to visualize small changes like these in the content of tumor cells that are present in the sample being analyzed.
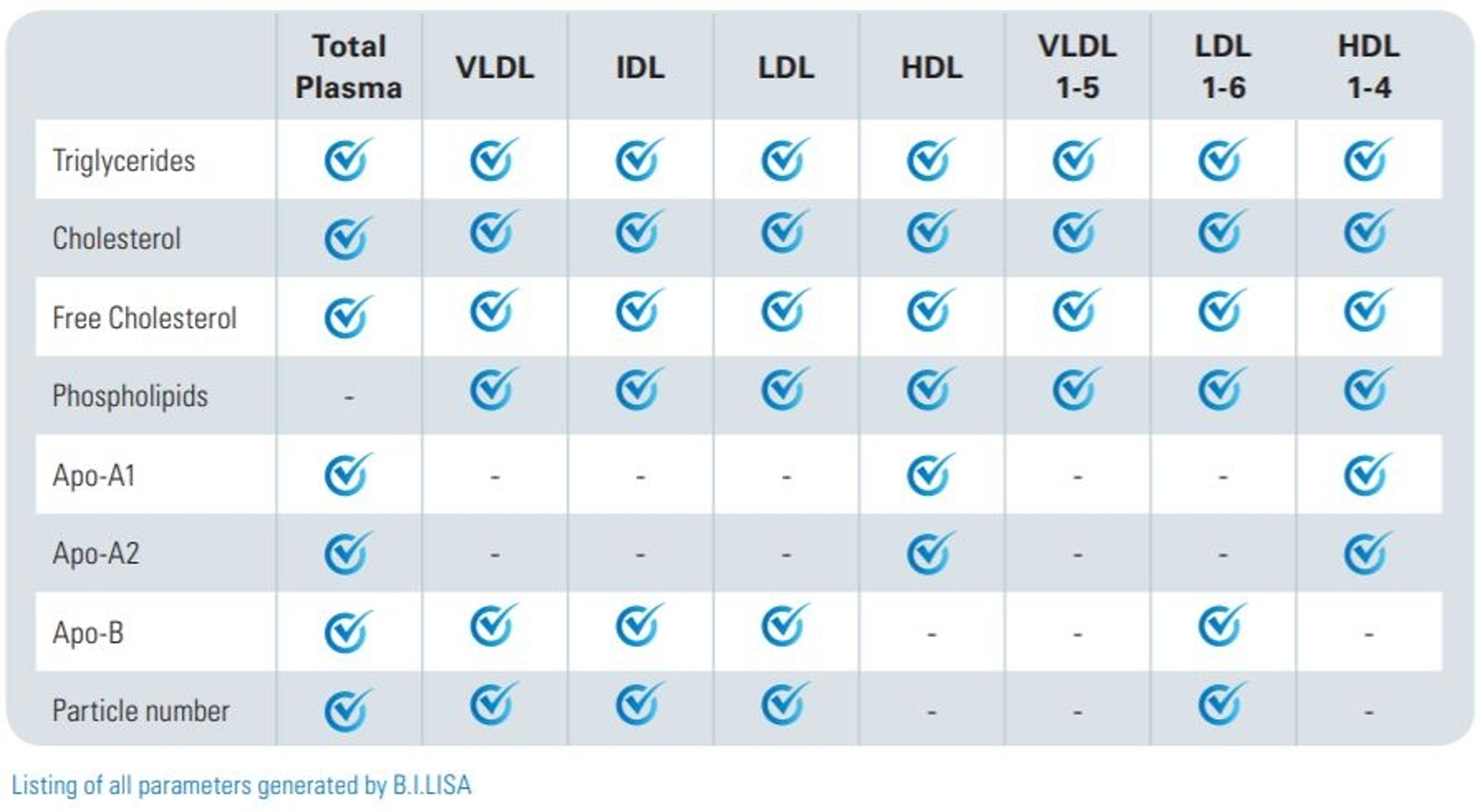
Q: How are the results from the IVDr platform delivered to users? How long is the measurement time?
TFB: With the IVDr platform, the acquisition protocols are optimized at the spectrometer. When the data is acquired, we submit it automatically to a local server from where the quantified metabolites or lipoprotein subclass variable data can immediately be retrieved. In the newest version of this protocol, we're also able for the plasma and urine quantification to have symbols that tell us also about how accurate you can expect the measurement to be.
Q: How are tissue samples prepared for magic-angle spinning MRS analysis?
TFB: When we work with tissue samples, we keep them stored in cryotanks with liquid nitrogen. When we prepare them, we are careful to keep them in a frozen condition. We have designed a small workstation that we can fill with liquid nitrogen and we have exchangeable steel plates so that we can keep the tissue frozen while working with it. Usually, we use around 10 milligrams of sample and we use a disposable insert that can be put inside a rotor. We found that some of the batches of these inserts contained contaminants that are difficult to wash out, so we have begun to move back to placing the sample in the rotor itself, which requires an increased sample size of 50 microliters. We only add a small amount of deuterium containing formate and TSP. We don't want to add too much as this can potentially extract metabolites from the tissue. The rotor is then transferred to the magnet where the sample is brought to a stable temperature of approximately five degrees Celsius.
Q: Considering the freeze-thaw cycles that samples may go through, are you still able to see the important biomarkers and metabolites in these samples?
TFB: In my group, we have not carried out any repeated freeze-thaw studies of tissue samples. But I know that there are studies that have been performed on this and that it has shown that repeated freeze-thawing will induce changes to the tissue from each freeze-thaw cycle. Most metabolites will still be visible but depending on the process that's happening to make those changes, it may also change the ratios between them.
Important note: The methods and solutions discussed during the webinar are for research use only and not for use in clinical diagnostic procedures.
SelectScience runs 3-4 webinars a month across various scientific topics, discover more of our upcoming webinars>>