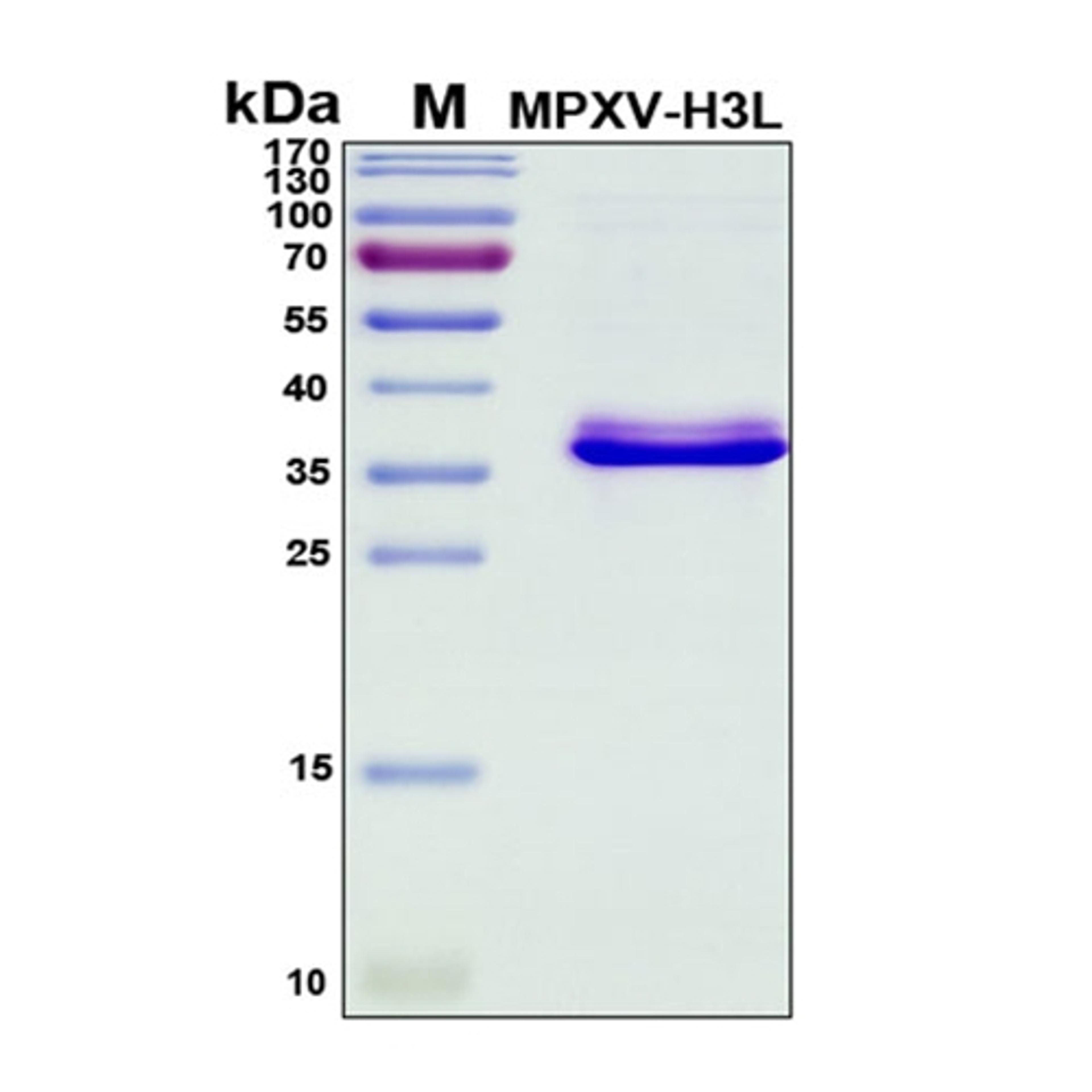
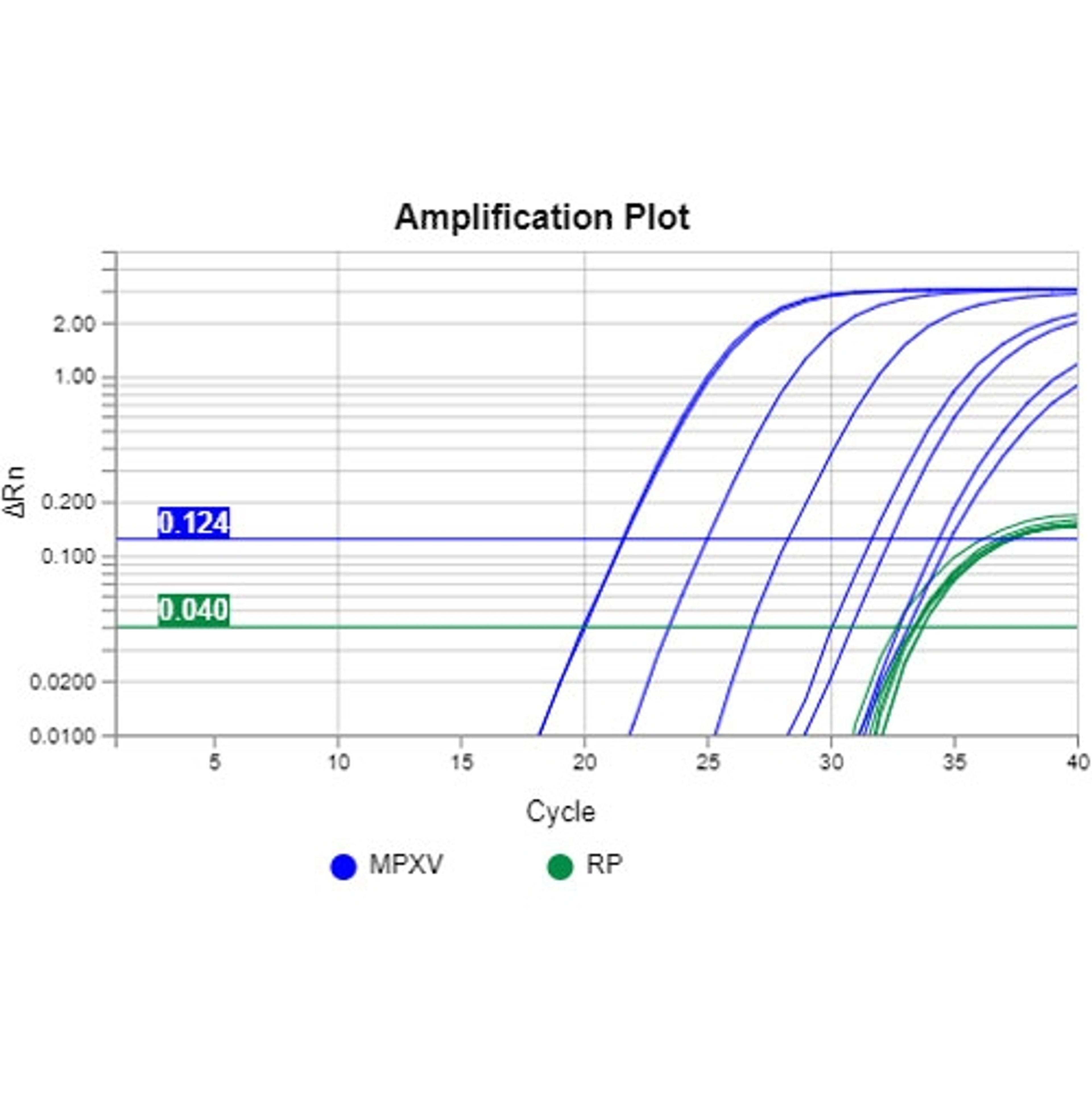
Monkeypox: Laying the groundwork for research and diagnostics in a time of outbreak
How a life sciences company is supporting scientists with understanding and controlling monkeypox transmission through cutting-edge immunoassays and PCR kits
16 Nov 2022

With the sudden emergence of reported monkeypox cases in non-endemic countries back in May 2022, health authorities and infectious disease researchers alike had to jump into action to quickly enhance their methods of surveillance. Research efforts to bolster laboratory diagnostics testing, available therapeutics, and vaccine development became a priority as new epidemiological data continued to roll in. As seen with the ongoing COVID-19 pandemic however, such a response would not be possible without a combination of efforts between scientists and organizations whose mission is to serve the research community.
Dr. Jarad Wilson is an Associate Director of Business Development and a Scientist focusing on high level experimental design at RayBiotech. He is an Emory trained immunologist where he focused on investigating antiviral responses against cleared and persistent viral infections, with a particular focus on the cellular response through interferon gamma and other antiviral molecules.
Now a director of a prominent life sciences company that delivers crucial proteomics discovery tools to scientists in industry, academia, and research worldwide, Dr. Wilson’s focus has shifted to providing cutting-edge array technologies for the discovery of disease-related protein biomarkers, the identification of new drug targets, and the advancement of personalize medicine. These three key areas of disease research have become ever more vital with the arrival of the COVID-19 pandemic, and now the multi-country outbreak of monkeypox.
Addressing the knowledge gaps
Monkeypox is a species of double-stranded DNA virus that is better at correcting replication errors than an RNA virus and therefore less prone to mutations. Dr. Wilson explains that one of the biggest challenges to scientists initially was the lack of available research. “The orthopox family of viruses aren’t prone to mutation, and so scientists had to look to past research on related species such as Variola and ectromelia for insights,” he explains. “In large, that was one of the biggest challenges in terms of researching it.”
Accompanying the lack of past research was also a history of funding shortages, especially following the declaration of the eradication of smallpox in 1980. “There was not a lot of money behind it, and so there was not a lot of people researching it. When this outbreak started, there was a sudden shift in realization and an injection of funding, meaning scientists were caught having to play a lot of catch-up,” states Dr. Wilson.
For manufacturers and equipment suppliers, this sudden rush of research meant an increased demand for not just consumables, antibodies, and proteins, but also detection kits that require complex upstream materials, items that were already in short supply as a result of COVID-19.
Paving the way
Dr. Wilson explains that SARS-CoV-2 taught the industry that it needs to be proactive when adversity first strikes, rather than waiting for things to really escalate. “When we make preemptive decisions to begin developing proteins that could be relevant to forthcoming research, we’re well prepared to provide for our customers when they reach out for support,” he explains.
In the case of monkeypox, the scientists at RayBiotech had their work cut out for them as the pox family of viruses has over 200 estimated proteins encoded by its genome. “It does almost everything but replicate itself outside of a cell. It has most every other bit of host machinery that it would need to replicate, and it's great at avoiding the immune system. We had to look back at variola, mousepox, and with what little was known about the Central African clade of monkeypox at the time, to try to find the proteins that seemed to drive the most antibody response as a starting point. These are the proteins that are likely to be of interest for human vaccine development,” Dr. Wilson states. “From there, we took a step back to look at the anti-immune mechanisms that the virus employs, such as control proteins, decoy receptors for interferons and cytokines, or host regulators, to guide the rest of our portfolio.”
By taking the jump to investigate these anti-immune mechanisms, Dr. Wilson and the RayBiotech team began laying the groundwork to help scientists answer some of the questions surrounding the initial immune response to the virus. For example, customers began reaching out to ask for kits to help examine the response of patients over the age of 50 who may still have detectable antibody responses from past vaccinations.
We hope to never have to institute them, but we have a lot of the tools that we need to go in the direction that’s necessary to help the scientific community respond to the monkeypox outbreak as it evolves.
Dr. Jarad Wilson
Associate Director, Business Development, at RayBiotech
A complete offering
Leaning on the experience gained from developing a COVID-19 portfolio largely from the ground up, RayBiotech has continued well past its initial offering of proteins to develop a full host of monkeypox virus research products. “We started with a protein, now we have multiple proteins, as well as antibodies, and ELISA kits against antibodies. We're also looking into developing ELISA kits against the monkeypox proteins if there's that demand,” explains Dr. Wilson. “Alongside those, we’ve expanded into PCR kits via our 1-Step High Throughput PCR and standard Real-Time PCR kits, to detect the virus, which have performed well across a number of customers who've used them and compared us to others. We're also exploring a laboratory-developed test just in case there needs to be expansion to provide diagnostic solutions, much like we saw with COVID.”
Dr. Wilson goes on to explain this push towards a diagnostics standpoint, noting how their focus during COVID-19 of forcing the researcher to perform separate purification steps before running a PCR test led them to develop a built-in one-step buffer that is ideal for this shift. “The one-step buffer lyses the virus on contact, and it can then be run on PCR immediately, even with the lysed and diluted material.”
Evolving with the virus
The Centers for Disease Control and Prevention (CDC) recently reported three cases of a significant deletion in the tumor necrosis factor (TNF) receptor, which is the analyte detected in CDC West African MPXV and Generic MPXV real-time PCR tests. Dr. Wilson notes an additional focus for the company going forward is introducing a secondary primer set for its PCR kits. “With the TNF receptor mutation, tests produced using CDC published primers and probes that specifically target monkeypox virus, will give false negatives. With the secondary primer set that we will be building in, we have two possible hits that are unique to monkeypox, adding in an extra layer of assurance,” he explains.
Continuing to align itself with the needs of its customers, RayBiotech has identified other areas for continual innovation going forward. “We're exploring a loop mediated isothermal amplification (LAMP) assay for a more in-the-field use if that need arises. It's also something we're looking at from a non-breakout point of view, but for the second and third-world countries that may have a need in the field as well,” explains Dr. Wilson. “From immunity tracking to in-vitro diagnostics and point of care solutions, we have a lot of the tools that we need to pivot in the direction necessary to help the scientific community respond to the monkeypox outbreak as it evolves.”