In vivo Imaging to Optimize Liposome-Mediated Drug Delivery in Cancer
Drug delivery researcher, Dr. Maria Lambros, describes her goal to exclusively target pancreatic cancer cells with chemotherapy drugs, while keeping healthy cells unaffected
21 Nov 2017
Chemotherapy drugs, although effective on cancerous tissue, have undesirable effects on healthy cells. To eliminate these side-effects, researchers have been designing and optimizing drug delivery methods to specifically target tumor sites in the body. In the 1960s, reliable drug delivery vehicles for chemotherapeutics were proposed: liposomes. The field of drug delivery has since accelerated in terms of liposome design and target specificity. In this SelectScience® interview, we speak with Dr. Maria Lambros who designs drug-encapsulated liposomes to target pancreatic cancer cells.
“One of the biggest challenges is to design liposomes that accurately reach cancer cells to deliver them drugs,” says Lambros, Associate Professor of Pharmaceutical Sciences at the Western University of Health Sciences, USA. Between their lipid and aqueous components, the liposomes can be designed to hold a variety of drugs and target proteins. To ensure these reach their desired destination accurately, liposomes carry ligands on their surface which only bind specific receptors or transporters expressed at the destination. “In pancreatic cancer, certain transporters on the cancer cells are upregulated. I target these cancer cells by designing my liposomes such that the proteins on the outside of the liposomes will only bind to the particular transporter on the cancerous cells,” says Lambros. The drugs encapsulated within liposomes enter the cancer cells by receptor-mediated endocytosis. “The liposomes themselves are endocytosed by the cell delivering the drug into the cell,” adds Lambros.
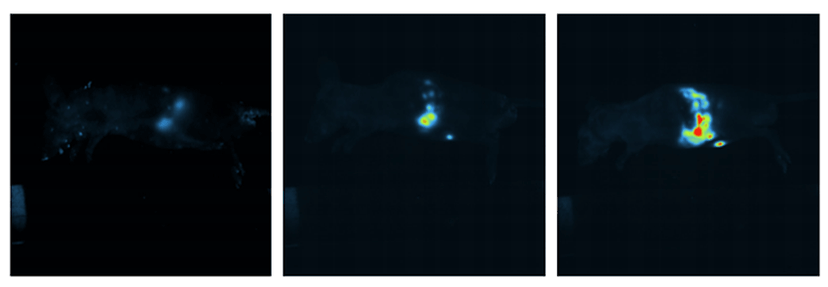
The iBox enables color-coded in vivo fluorescent imaging in mice. Image courtesy of the Lambros lab.
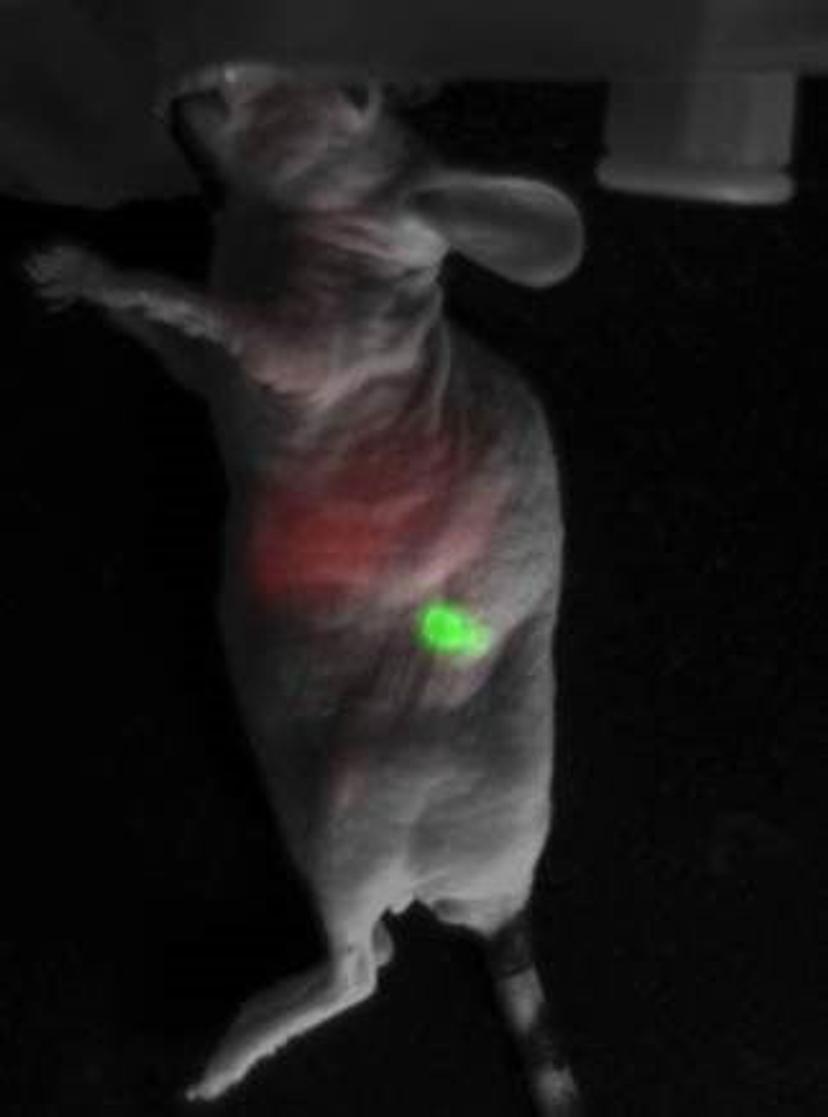
To monitor the drug delivery process, Lambros and team use fluorescently labeled liposomes in conjunction with both orthotopic and ectopic models of cancer xenograft, where the tumors are induced in the pancreas or subcutaneously, respectively, and they, too, carry a different fluorescent tag. “Our liposomes carry a red fluorescent tag and the tumor in the mouse fluoresces in green. When we inject the liposomes into the bloodstream, they will slowly localize, and bind onto the pancreatic cancer and internalized by the cancer cells,” explains Lambros. The drug delivery is deemed successful when two respective fluorescent signals coincide, thereby indicating that the liposome has successfully entered the cancer cell. “When the red and green fluorescence meet, we’ll see a yellow fluorescence.”
Lambros’ lab measures the progress of the liposomes in the body and, eventually, the success of the drug delivery via in vivo live animal imaging with the UVP iBox® Scientia™ Small Animal Imaging System. “The pancreas is located deep inside the body. Using the iBox, we can literally see through the animal’s body to monitor the drugs in a time-lapse method over several hours,” says Lambros. “Once we inject the drug, we can monitor its location within the body over time, i.e. we can follow, in a visual manner, the drug kinetics in the body. What was once invisible can now be visualized using the iBox.”
“When proposing experiments to funding institutes, in vivo imaging is always encouraged,” adds Lambros. “It is very convenient to measure fluorescence in vivo using the iBox. We can observe how the tumor grows in vivo, and examine when the drugs target the cancerous cells once injected. Other techniques such as luminescence, do not offer the same level of ease because they require the injection of a substrate.”
An expert in designing liposomes, Lambros wants to continue developing drug-encapsulated liposomal delivery systems for other forms of cancer. “In some types of cancer, the tumor cells are surrounded by vital organs and healthy cells. This poses a challenge to surgeons when removing the tumor. I’d like to design liposomes and develop drug delivery systems to target and kill the cancer cells in different organs while keeping the healthy cells unaffected.”
Find out more about the UVP iBox® Scientia™ Small Animal Imaging System here.
