How does Abcam use and develop cell-based assays to test protein bioactivity?
Abcam scientist, Dr. Jimmy Theriault, discusses how to determine if proteins are functionally active using cell-based and biochemical assays
4 Oct 2021

In this exclusive interview, we speak with Dr. Jimmy Theriault, a scientist working in the Protein Sciences group at Abcam, about his role in developing cell and biochemical-based assays to test the functionality of purified proteins. Here, Dr. Theriault shares his expert insight into the current cell-based assay landscape, including experimental design, persisting challenges, and how to determine if proteins are functionally active.
How do you demonstrate that Abcam’s proteins are functionally bioactive?
JT: We use cell-based assays to determine protein bioactivity. The assays combine the most relevant cell type available with the appropriate biological output generally associated with the known function of each protein. We then assess the potency of each protein through the generation of a dose-response curve, where an ED50 (effective dose at 50% response) is determined. If the ED50 has a similar value to what is currently accepted in the scientific literature for a specific assay, then our protein is considered functionally active.
At Abcam, we are committed to providing pure, authentic, and functional proteins. Cell-based assays are an integral part of the quality control process and validate that our proteins possess all the essential characteristics associated with their known functions. Therefore, cell-based assays are a fundamental part of the premium data package generated for our mammalian-expressed recombinant proteins, in addition to SDS-PAGE gels, HPLC chromatography, and mass spectrometry analysis data.
Why is it important to demonstrate the protein activity with a cell-based assay versus another assay format?
JT: Cells are the natural environment where a protein performs its typical function, so cell-based assays are the optimal assay format to test protein functionality. While binding methods, such as surface plasmon resonance (SPR) spectroscopy, can assess the interaction between extracellular ligands and cell surface receptors, these methods only determine the affinity between the two molecules. They do not guarantee the initiation of downstream intracellular events that can impact cellular function.
Conversely, by testing the protein activity with a cell-based assay, we can measure and quantitate the effect of ligand-induced receptor activation on cellular processes such as protein secretion, chemotaxis, and cell adhesion. In this regard, a cell-based assay enables us to recreate various key aspects observed with the protein function in an in vivo setting.
That being said, for a few proteins, biochemical assays have to be used to determine the activity of certain enzymes and their inhibitors. In these cases, the assay is performed in a cell-free environment. This assay format provides simplicity by only involving an enzyme with or without an inhibitor and a fluorometric or colorimetric substrate. The downside of utilizing a biochemical assay is that it is a more artificial system, drifting away from the intricate intracellular environment seen with the cell-based assay.
Therefore, for most of our recombinant proteins, a cell-based assay is better suited to accurately establish the bioactivity of the ligand/receptor complex. The biochemical assay is only used when assessing the enzymatic activity of our proteins is necessary.
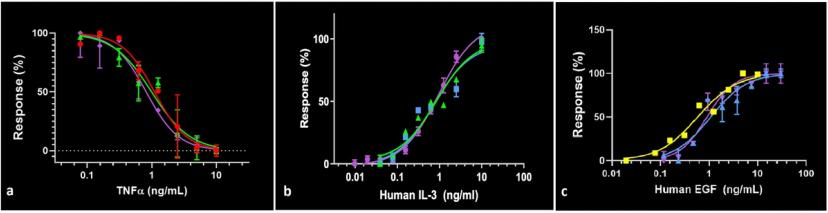
What are some of the challenges you face when selecting a cell-based assay?
JT:Since almost every premium protein produced at Abcam has a unique cellular function, a specific assay protocol or experimental procedure must be developed for each protein tested. This unicity can also apply to the same protein from different species, which, in many instances, requires two unrelated cell-based assays to be performed for each species.
The success of a cell-based assay can also be strongly affected by the quality and responsiveness of the cell type chosen. Some cell types such as primary (endothelial cells) or differentiated cells (osteoclasts) are generally quite fragile, difficult to grow, or only survive for a very short time. These factors have a pronounced detrimental effect on assay performance.
Furthermore, certain cell-based assays assessing cell migration (chemotaxis) and adhesion are notoriously known for their inconsistency due to the high variability between biological replicates and the generation of a narrow range of activity (small maximal vs. minimal response window). Unsurprisingly, the protein effect on these processes can be missed due to an insufficient signal or response induced during the cellular treatment.
Overall, there is no perfect assay. It’s usually a compromise between multiple factors that will dictate the choice of the final cell-based assay design. Hence why our cell-based assays are developed specifically for each premium protein, and ensuring assays have optimum performance.
A lot must go into ensuring a functional assay is performed correctly for each unique protein. Can you tell us more about what influences your decision to select, develop and execute a specific cell-based assay for any given protein?
JT: There are many important aspects to consider in ensuring a successful cell-based assay. A thorough literature search is the first essential step to identify and select the assay. Multiple factors, such as relevance, feasibility (level of difficulty), completion time, and cost, impact which assay is used.
After picking up an assay, a detailed protocol must be designed. An existing protocol can form the basis of the assay, but it often needs to be adapted to ensure compatibility with the chosen assay format. For example, we adapted a chemotaxis assay by labeling cells with a fluorescent viability dye before submitting them to cell migration. With this protocol modification, the assay was more robust, and the completion time was significantly reduced.
In terms of experimental design, we determine several parameters, such as cell number, assay volume, treatment duration, and microplate type to pinpoint the optimal conditions to perform an effective cell-based assay. We then identify the reagents, materials, and cells, which can often include our ELISA and alkaline phosphatase kits, to create and run the cell-based assay.
Finally, it is critical to consider the type of instrumentation that will be used for data generation. The cell-based assay outcome must be compatible with the common readouts present on a plate reader, such as absorbance, fluorescence, and luminescence, as well as the reading mode (eg endpoint versus kinetic mode).
Overall, selecting and optimizing a cell-based assay is not always a linear process and involves careful planning and flexibility to ensure the highest possible performance.
What does the future hold for proteins at Abcam?
JT: We recently moved to our new state-of-the-art facility in Waltham (Boston, MA), which has increased production capacity and the expansion of our ability to produce proteins from insect cells and bacterial-based systems.
I am proud to be part of a team generating premium-grade bioactive proteins and molecular tools for the scientific community. I strongly believe we provide an essential service that can speed up research discovery and help foster scientific innovation. It is an exciting time to be at Abcam and I cannot wait to see what the future holds for us.
Learn more about Abcam’s premium bioactive range
- Developer deep dive: Designing proteins and reagents for cell and gene therapy research >>
- Premium grade bioactive proteins to meet your cell and gene therapy application needs >>
Do you use Abcam products in your lab? Write a review for your chance to win>>
