Harnessing the latest imaging technology to advance angiogenesis research
Learn how one biotechnology company is using cutting-edge imaging to improve the visualization of angiogenesis in its 3D tissue models
6 Aug 2019Editorial article
Ever since the discovery that tumors undergo angiogenesis in order to grow and survive, researchers have sought to better understand the detailed mechanism of new blood vessel formation. Targeting that mechanism with new drug compounds that inhibit the development of vascularization, rather than the tumor cells themselves, has become one of the ‘Holy Grails’ of modern cancer therapeutics research. Whilst the theory is simple enough, the greatest challenge has always been technological limitation – how to develop true-to-life 3D models of human tissue and vasculature that can then be ‘interrogated’ in sufficient detail to understand exactly how angiogenesis progresses, and how it’s inhibited in the presence of new drug compounds. One biotechnology company in the Netherlands, MIMETAS, is operating at the cutting-edge of 3D human tissue models of vasculature for angiogenesis research. Its Chief Technology Officer, Dr. Bas Trietsch, has formed a collaboration with Molecular Devices to push forward angiogenesis research to new heights with their high-content imaging solutions.
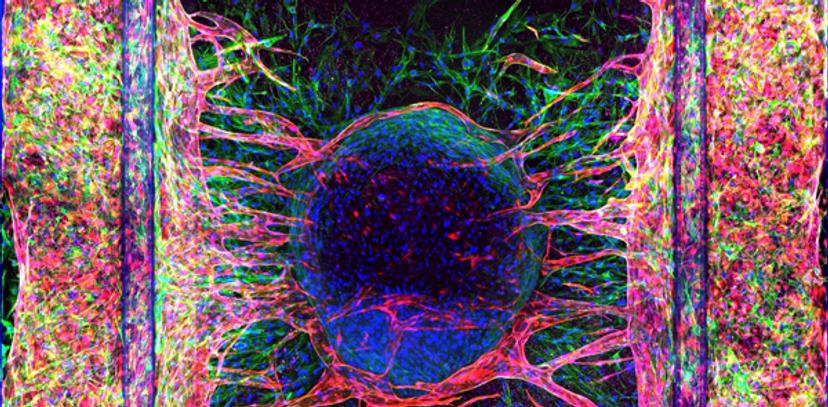
Overcoming the pitfalls of conventional tissue culture
Human vasculature is not an easy tissue to culture in vitro, at least not using 3D approaches that seek to mimic the real thing. Conventional methods using simple 2D cell culture can achieve functional tube structures, but they lack real-life context and often need to be implanted within a laboratory animal in order to mimic true physiology. As Trietsch points out, this is costly both in terms of time and money, and it does not contribute to the 3Rs agenda for the refinement, replacement and reduction of animals in research.
With Trietsch’s lead, MIMETAS therefore developed its OrganoPlate® Graft as a solution – an in vitro cell culture microplate platform that enables the vascularization of 3D tissues. This organ-on-a-chip solution provides researchers with a microvascular bed to which they can add tissue to become vascularized. The platform employs a 384-well plate with glass bottom, which allows for perfusion, easy interrogation, and high throughput, that is also compatible with the standard equipment researchers use to study 3D tissue models. This is far from the end of the story, however. The real power of this technology comes when it is allied to the cutting-edge developments in high-content imaging that have taken place over the past decade.
Advanced visualization of angiogenesis tissue models
Tracking the development of vascularization in tissues and directly observing how potential drug treatments affect the phenotype and progression of angiogenesis are crucial to the application of the OrganoPlate® Graft platform in cancer therapeutics research. MIMETAS’s partnership with Molecular Devices has enabled them to do just that, as Trietsch attests: “This means that we can do phenotypic screening; we can look at these complex structures in 3D, and in a very high throughput manner, in high detail. We can visualize the process of angiogenesis in true physiological context.”
He further adds: “Combining a complex tissue model with very advanced and powerful imaging techniques really opens up a whole new panel of assays that researchers can use to get concrete answers to their questions very, very quickly and in a robust and scalable manner.”
Going 3D for greater insight
Those powerful high-content imaging techniques are provided by Molecular Devices in the form of their ImageXpress Micro Confocal High-Content Imaging System and their ImageXpress Pico Automated Cell Imager. MIMETAS has been very successful in using these screening systems to interrogate the models it has built on its OrganoPlate®.
Trietsch is effusive in his praise: “What we've found is that these instruments combine an excellent image quality and high-throughput image acquisition within what we feel is unsurpassed flexibility and programmability. And that allows us to be creative in building the best assays to be combined with the best and most complex tissue models, for high-quality readouts.”
It is clear Trietsch considers that having these two strengths combined genuinely gives researchers options that they would not be able to get with any other combination of technologies.
He concludes: “The ImageXpress Micro Confocal system has really turned out to be the workhorse in our lab. We use it constantly to get the most detailed in-depth information from our three-dimensional tissue models at high throughput. Being able to grow 40 or 100 three-dimensional tissues in one plate is nice, but in the end, we are looking for answers to questions. That means we need to be able to very quickly, robustly and efficiently image these tissues and use the tools that the ImageXpress Systems - and their software – provide to actually turn those images into information.”
The final analysis
Trietsch’s experience is a fine example of marrying technologies to advance a research field. He closes with a final observation: “We've found that having the flexibility to use confocal imaging with a high-throughput system has been instrumental in unlocking the full potential of a system like the OrganoPlate®.”
Find out more about MIMETAS and how the ImageXpress Micro Confocal High-Content Imaging System and the ImageXpress Pico Automated Cell Imaging System from Molecular Devices could help>>

