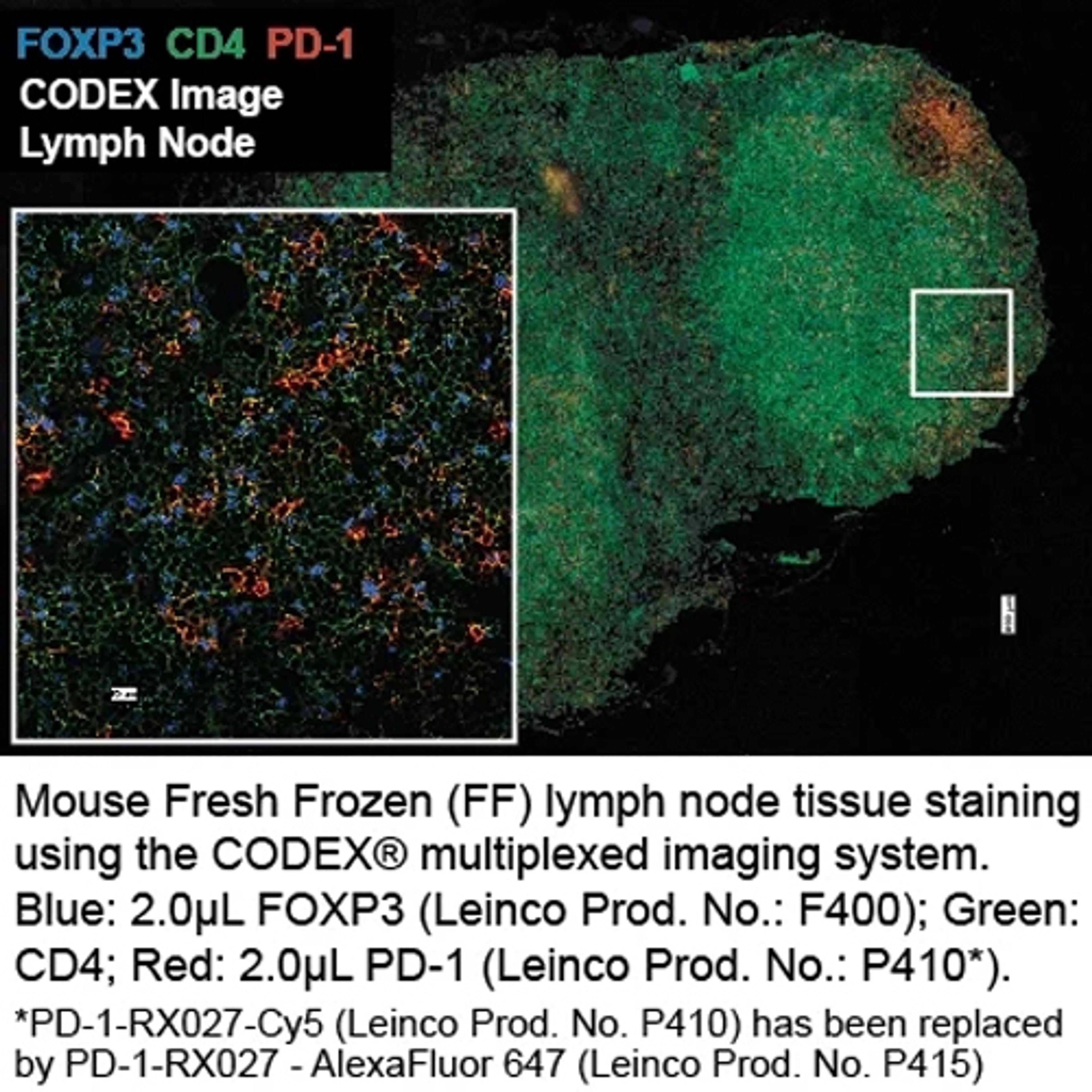
Deciphering the mechanisms of dual PD-1 and LAG-3 blockade: A tumor-killing combo or no-go?
University of Pittsburgh researcher, Dr. Lawrence Andrews, shares his latest work unraveling the synergistic role of PD-1 and LAG-3 in cancer immune evasion
6 Dec 2022There is a buzz of excitement and activity across the field of immuno-oncology following a series of swift clinical approvals for targeted immunotherapies against a variety of different cancer types. Among these, a new combinatorial immunotherapy, based upon two inhibitory immune checkpoints – programmed cell death (PD-1) and lymphocyte-activation gene 3 (LAG-3) – received US FDA approval in March 2022 for the treatment of patients with unresectable or metastatic melanoma. However, while it has been shown that dual blockade of both PD-1 and LAG-3 can synergistically limit tumor growth, the mechanism behind this synergy and its effect on different cell populations remain largely unknown.
Working to address this knowledge gap is Dr. Lawrence Andrews, a researcher in the Vignali Lab at the University of Pittsburgh. In this exclusive interview, SelectScience® learns more about Dr. Andrews’ latest study on the mechanistic role of PD-1 and LAG-3 on CD8+ tumor-infiltrating lymphocytes (TILs) and hears how in vivo functional-grade antibodies from Leinco Technologies have proved invaluable to his preclinical research efforts.
Immune checkpoint therapy

Understanding the biology of immune checkpoints has been a major goal in cancer immunotherapy research over the last decade. Immune checkpoint inhibitory receptors (IRs) play a crucial role in modulating immune responses, acting as natural brakes upon the immune system through limiting T cell activation, thereby preventing autoimmunity, autoinflammation, and tissue damage. However, while essential for homeostasis, these signaling pathways can be hijacked by tumors, leading to elevated IR expression and a corresponding reduction in anti-tumor immune responses. As a result, a key focus of recent immunotherapeutic approaches has been to target IRs to reverse immune suppression, reinvigorate T cells and promote anti-tumor immunity.
Early success has been achieved through targeting individual inhibitory receptors such as cytotoxic T lymphocyte antigen 4 (CTLA-4) and PD-1 using monoclonal antibodies. For example, ipilimumab and nivolumab, which modulate the effects of CTLA-4 and PD-1, respectively, have led to remarkable tumor remission in patients with several tumor subtypes. However, a considerable proportion of patients remain unresponsive to treatment, whilst the combination of ipilimumab and nivolumab remains controversial due to a higher incidence of toxicity. Consequently, the focus has recently shifted to targeting alternative inhibitory receptors in combination with current immunotherapies – with PD-1 and LAG-3 the latest pairing to come under intense scrutiny.
“We now know that dual blockade of both PD-1 and LAG-3 synergistically limits tumor growth compared to anti-PD-1 alone, yet the mechanisms behind the synergistic contribution of PD-1 and LAG-3 on hindering CD8+ T cell-mediated antitumor immunity have not yet been determined,” explains Andrews. “This is a population of particular interest as CD8+ TILs have the greatest expression of LAG-3 in the tumor, which hinders effective anti-tumor immunity by mediating a state of exhaustion.”
A two-pronged approach
To assess the contribution of PD-1 and LAG-3 in promoting tumoral immune escape, Andrews and his team are generating conditional knockout mice that lack PD-1 and LAG-3 surface expression on cell populations of interest. “We use a Cre-LoxP system which means we can restrict the Cre-recombinase removal of PD-1 and LAG-3 surface expression to different specific T cell populations,” he says. “Right now, we’re focusing on the role of this removal on the intrinsic mechanisms of CD8+ T cells, but we also have projects investigating other CD4+ T cell populations.
These genetic studies are complemented by experiments using anti-PD-1 and anti-LAG-3 antibodies, whereby PD-1 and LAG-3 receptors remain present, but their activity is blocked. Andrews explains the rationale behind the dual approach, “Knockout studies are great to understand the biology, but when we come to a translational perspective, we're not making genetically modified humans, patients are going to be receiving anti-PD-1 and anti-LAG-3 antibodies as an immunotherapeutic,” he says. “Therefore, we like to do parallel studies with these antibodies alongside our genetically modified mice to understand whether a specific cell population is of importance to the overall immune effects that would occur with antibody blockade.”
Ensuring reproducible science
As with any biomedical research, the use of reliable reagents, including antibodies, is critical to generating consistent and reproducible data. For this reason, Andrews’ lab uses antibodies from Leinco Technologies, a company with more than 30 years of experience supporting cancer and immunology research, and a wide portfolio of immune checkpoint monoclonal antibodies of the highest purity standards in the industry. “When injecting anti-PD-1 and anti-LAG-3 into mice, we use purified in vivo gold functional-grade antibodies from Leinco,” he explains. “The grade relates to purity and toxin levels, which is vital for in vivo studies.” Poor quality antibodies can lead to toxicity and off-target effects that have a large impact on the outcomes of in vivo experiments, contributing to a considerable amount of wasted time and research funds. To prevent this, in vivo GOLD™ antibodies are subject to rigorous quality control testing, including SDS page and analytical SEC, to ensure low endotoxin levels, no detectable aggregates, and no leachable protein A, whilst in vivo PLATINUM™ antibodies are also tested for pathogen-free status by PCR.
Andrews goes on to explain that his lab has a longstanding relationship with Leinco Technologies, highlighting their partnership during the company’s licensing of the anti-LAG-3 monoclonal antibody (C9B7W). “C9B7W was ultimately generated in our lab, and we worked closely with Leinco for it to be licensed,” he says. “This is really the only anti-LAG-3 antibody that is commercially used.”
The role of LAG-3 and PD-1
To determine the effect of PD-1 and LAG-3 removal and inactivation upon CD8+ cells, Andrews employs a variety of techniques, including scRNA-seq, TCR-seq, flow cytometry, antigen-specific adoptive transfer, and retroviral transduced knockdown of genes of interest. “We use single-cell sequencing as a discovery tool to find out which genes are most upregulated or downregulated between our conditions, and then validate these by flow cytometry,” he explains. “We’ve then looked at specific genes and have done retroviral transcription knock-down with shRNA to see if the same phenotypes can be capsulated as with the PD-1/LAG-3 knockout T cells.”
C9B7W was ultimately generated in our lab, and we worked closely with Leinco for it to be licensed.
Dr. Lawrence Andrews
Researcher in the Vignali Lab at the University of Pittsburgh
With the results of his latest study soon to be published, Andrews shares some of the key insights generated by this work so far. “In the case of PD-1 and LAG-3 deficient mice, where there's no PD-1 or LAG-3 on the CD8+ T cells, we see a transcriptionally distinct population compared to the loss of PD1 or loss of LAG3 alone,” he says. “This suggests there is synergy between the two inhibitory receptors with unique downstream inhibitory pathways, proving that dual targeting of both PD1 and LAG3 is a viable immunotherapeutic approach.”
“Once this paper gets accepted, we'll turn our focus to what other T cell populations of interest might be amenable to PD-1/LAG-3 blockade, such as Tregs or other CD4+ populations,” he continues. “It may be that blocking PD-1 and LAG-3 on Treg cells has the complete opposite effect, a situation where CD8+ T cells are more functional, yet Tregs could be more suppressive. If we know that, then we could design more focused immunotherapies to target those CD8+ T cell populations and not the Tregs.”
Future outlooks
The recent approval of relatlimab (anti-LAG-3) and nivolumab (anti-PD-1) combination therapy for patients with metastatic or unresectable melanoma has led to a surge of renewed interest in the role of LAG-3, with many questions yet to be answered. “It’s a very exciting time to look at LAG-3 and everyone’s now suddenly interested again,” asserts Andrews. “The relatlimab/nivolumab combination has been shown to be as efficacious as nivolumab and ipilimumab (anti-CTLA4) but without the severe toxicity that occurs with the latter. However, whether this combination is efficacious in other tumor types remains to be seen, and it is also still unclear which cell types are most important.”
Continuing to explore the extent of synergy and cooperative interactions between these inhibitory pathways will be critical to informing the further development of combinatorial immunotherapy regimens. “Ultimately, this preclinical knowledge may allow the development of bi-specifics or other-directed agents that can specifically target PD-1 and LAG-3 on the relevant population to provide a more efficacious treatment,” Andrews concludes.