Cancer Diagnostics: A 2016 Update
An overview of new developments in the field of cancer diagnostics
1 Feb 2016
For many patients, an early diagnosis means a prolonged survival time
Never before has so much money, time and expertise gone into cancer research. Science moves quickly and there are some really exciting new developments on the horizon for cancer diagnostics. This article looks at three main areas of interest regarding in vitro diagnostics, next generation sequencing, liquid biopsies and volatile breath analysis.
Early diagnosis
The benefits of being able to diagnose cancers at an earlier stage are significant. Put simply, the earlier a cancer is detected, the earlier it can be treated and prevented from spreading. For many patients, an early diagnosis means a prolonged survival time. Once a cancer has grown very large, or has spread from the primary site, patients typically face a poorer prognosis.
Diagnostic research
Cancer diagnostics currently rely heavily on invasive biopsies to confirm tumor type and to determine a treatment plan. Invasive biopsies are not always a practical option due to tumor location and the frailty and age of the patient. It is also now known that tumor composition and genetic make-up varies widely, even in a single tumor – so testing one biopsy site doesn’t provide you with an accurate overview of the tumor as a whole1.
There are three significant research approaches to improving in vitro cancer diagnostics.
Improving knowledge
An improved knowledge of how tumors form and grow is key to being able to identify biomarkers that are associated with the early stages of cancer. Cancer researchers have traditionally used Sanger sequencing to improve their understanding of the molecular basis of cancer; however the increasing application of next generation sequencing (NGS) to cancer diagnostics offers the potential to simultaneously screen for multiple mutations, giving a much greater understanding of the disease.
NGS is a technology in which millions or billions of DNA strands can be sequenced in parallel. NGS yields much more genetic data than Sanger sequencing, allowing an entire genome to be sequenced in as little as a day. This allows researchers to look at huge amounts of genetic data and to analyze shared mutations in genes that lead to disease.

Next generation sequencing allows millions of DNA strands to be sequenced in parallel yielding huge amounts of genetic data
Further advantages of using NGS over traditional Sanger sequencing for cancer diagnostics include the requirement for less DNA (a particular advantage when using fixed-tissue tumor samples), its speed of analysis (it is significantly faster) and it’s increasing cost-effectiveness.
Perhaps more than any other technology, NGS has the potential to unlock the true power of personalized medicine. NGS could allow clinicians to understand an individual patients genetic make-up, pinpoint the molecular aberrations that have driven their disease and determine what course of therapy is likely to be most effective.
Current market leaders in clinical oncology-related NGS include Illumina with the high-throughput DNA sequencing system, the MiSeqDx® System, QIAGEN’s new NGS GeneReader platform and the Life Technologies Ion PGM Dx System.
Liquid biopsies
The ‘holy grail’ of cancer diagnostics is the possibility of being able to diagnose cancer using a simple blood test, without the need for an invasive biopsy. It is now known that tumors release DNA into the blood stream of the patient. Liquid biopsies are blood tests that can analyze this circulating DNA, if the form of cell-free tumor DNA (cfDNA), circulating tumor cells (CTC’s) or exosomes in the patient’s blood, to give a true representation of that patients molecular profile.
An example of the effective use of ‘liquid biopsies’ was highlighted in 2013 in a paper published by Nature, featuring work undertaken by the Cancer Research UK Cambridge Institute and University of Cambridge2. In the paper the authors describe how the use of exome sequencing enabled them to analyze circulating DNA and take a snapshot of the genetic errors in patients with breast cancer.
Since then, numerous studies have been undertaken on potential liquid biopsies3, including a recent trial involving EKF’s PointManTM enrichment technology. The levels of detectable cfDNA in the blood vary considerably. One of the problems associated with liquid biopsies is their lack of ability to detect cfDNA in the blood when they are very low. PointManTM technology is able to amplify a target sequence of interest allowing researchers to detect these low level cancer mutations.
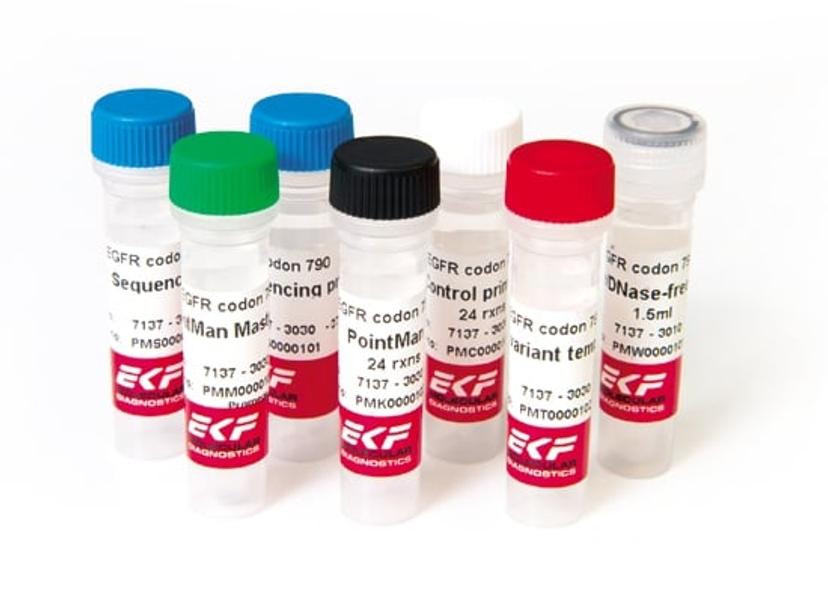
EKF's PointMan™ DNA Enrichment Kits allow researchers to amplify specific mutation sequences
Liquid biopsies have the potential to add significant value to cancer patients and to further propel us towards the goal of personalized medicine. In the near future, these tests might replace invasive biopsies to detect and monitor cancerous tumors, and could be used to identify tumors not yet visible with imaging techniques.
Volatile breath analysis
Volatile organic compounds (VOCs) are detectable in the breath in very low concentrations. Examples of VOC’s measurable in exhaled breath include alcohols, ketones and organic sulphides. Because the detectable levels are very low, analytical mass spectrometry is usually required to analyse the breath samples.
VOC breath analysis is an area of increasing interest in cancer diagnosis. It has been found that different VOC breath patterns can be correlated to various different diseases. VOC patterns have been identified in lung cancer4,5, colorectal & prostate cancer6, and breast cancer7.
Analysis of breath biomarkers is in its infancy and much more work needs to be undertaken in this field. Initially, large scale studies need to be carried out to determine normal VOC patterns before diagnostic biomarkers can be reliably identified.
Find out more about cancer research on our dedicated cancer diagnostics community page.
References:
http://www.nejm.org/doi/full/10.1056/NEJMoa1113205
http://scienceblog.cancerresearchuk.org/2013/03/13/blood-test-tracks-breast-cancer/
http://www.ncbi.nlm.nih.gov/pmc/articles/PMC4356857/
http://www.lungcancerjournal.info/article/S0169-5002(09)00208-6/abstract
http://www.sciencedirect.com/science/article/pii/S1570023210000383
http://www.nature.com/bjc/journal/v103/n4/full/6605810a.html
