ResourceLab Informatics
Validation for SoftMax Pro 7.1.1 Data Acquisition and Analysis software
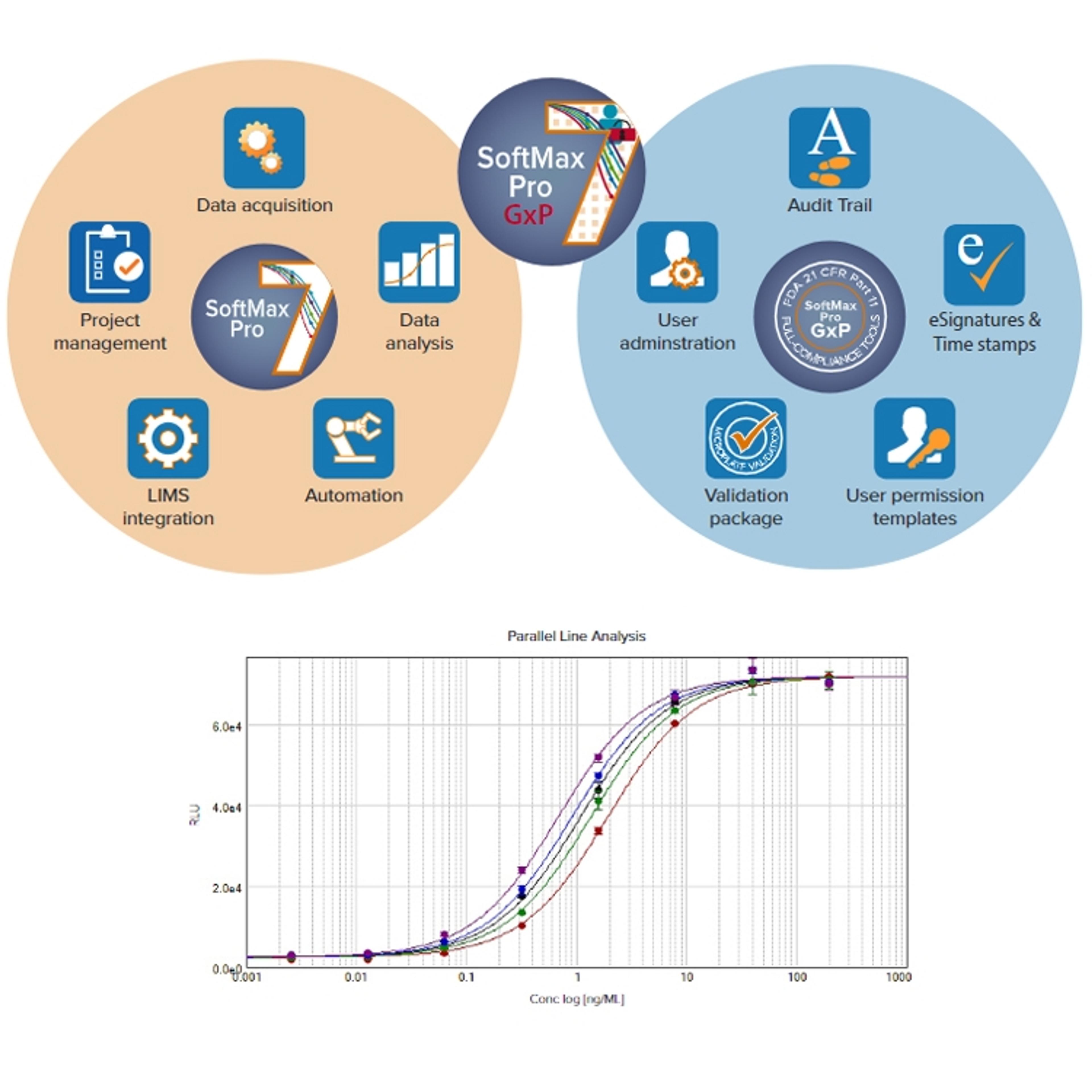
11 Mar 2020For researchers working in GLP or GMP laboratories, the SoftMax® Pro Software Validation Package provides the most comprehensive documentation and tools available to validate GxP protocols and data flow to ensure data integrity.