Transcreener® ADP² Kinase Assays
The Transcreener® ADP² Kinase Assay Kit universally detects ADP produced by any kinase or ATPase using a direct mix‑and‑read format, eliminating coupling steps. Available in FP, FI, and TR‑FRET formats with far‑red tracers to minimize interference, it's HTS-ready across 96‑, 384‑, and 1536‑well plates with robust Z' ≥ 0.7 performance.
Excellent blend of simplicity, economics & good science. Excellent technical support.
The ADP2 assay is a great choice for measuring activity for any ADP-generating enzyme
The ADP2 assay is a great choice for measuring activity for any ADP-generating enzyme in both real time and endpoint formats. The universal detection system has been very useful for more challenging targets.
Review Date: 31 Jul 2022 | BellBrook Labs
Extremely versatile
Kinetic profiling
The ADP2 assay is a great choice for measuring activity for any ADP-generating enzyme in both real time and endpoint formats. The universal detection system has been very useful for more challenging targets in our lab.
Review Date: 23 Jun 2016 | BellBrook Labs
Excellent blend of simplicity, economics & good science. Excellent technical support.
Review Date: 15 Jun 2011 | BellBrook Labs
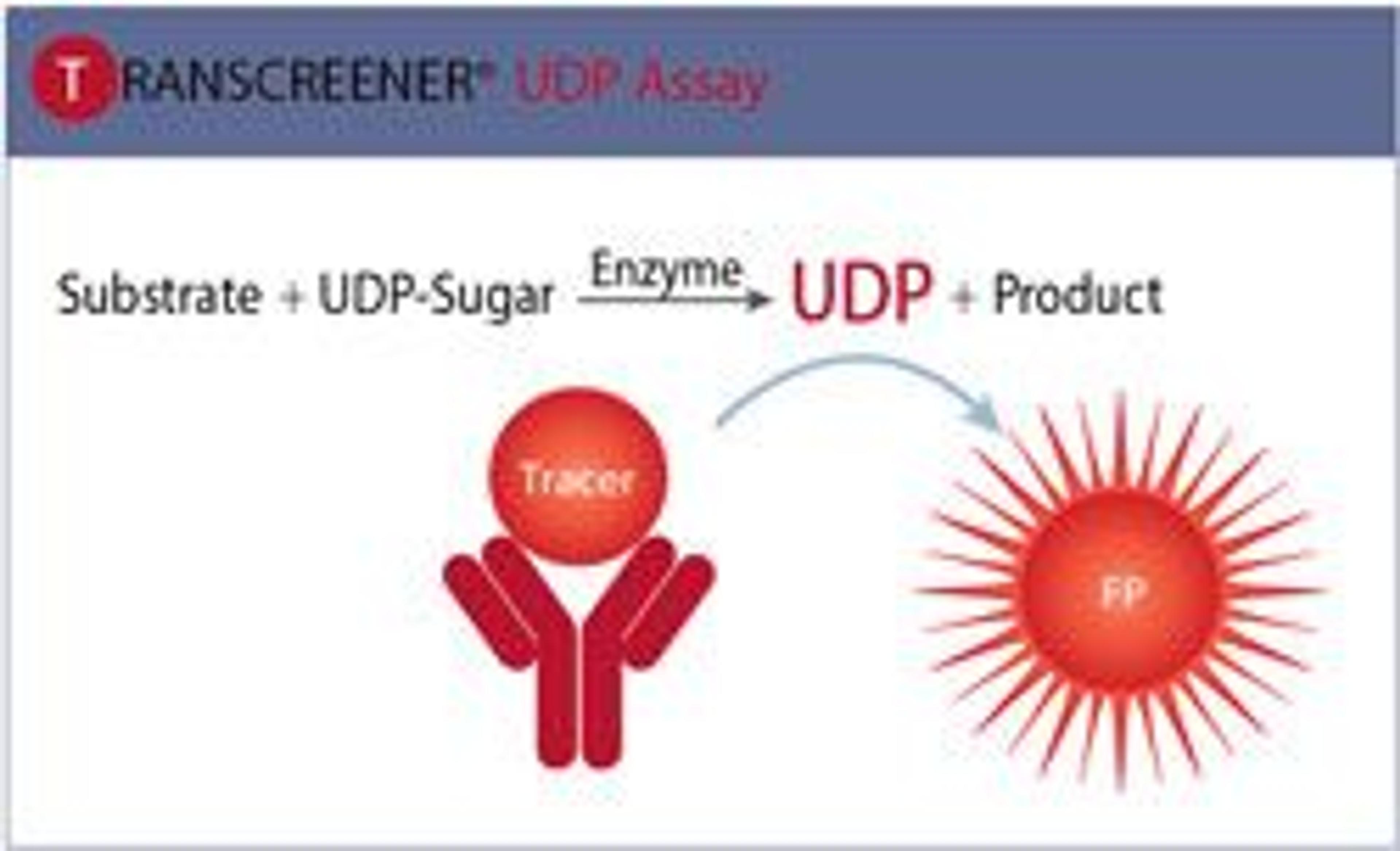
The Transcreener® ADP² Kinase Assay is a universal, mix-and-read HTS assay that directly measures ADP, the conserved product of kinase and ATPase reactions. Designed to eliminate complexity and reduce assay interference, it provides a robust, flexible solution for drug discovery and inhibitor screening.
Key Features & Benefits
- Universal Detection – Measures ADP from any kinase or ATPase, eliminating the need for multiple assay platforms.
- Direct Mix-and-Read Format – No coupling enzymes or secondary reactions, minimizing false positives and simplifying workflows.
- Multiple Readout Options – Available in Fluorescence Polarization (FP), Fluorescence Intensity (FI), and TR-FRET formats.
- Far-Red Tracers – Reduce compound interference for cleaner, more reliable data.
- HTS Compatible – Validated for 96-, 384-, and 1536-well plates, with Z’ ≥ 0.7 for reproducibility in screening campaigns.
- Automation Ready – Homogeneous format and stable reagents streamline integration into automated screening environments.
Applications
- High-throughput screening (HTS) of kinase inhibitors
- Mechanism of action and SAR studies
- Profiling ATPases and other nucleotide-dependent enzymes
- Hit-to-lead triage with reduced assay artifacts
Targeting innate immunity with transcreener assays
This application note by BellBrook Labs describes how the Transcreener ADP² Kinase Assay allowed rapid assay development to enable screening and dose-response measurements for five kinases involved in innate immunity.
Using the Tristar2 S LB 942 for High-Performance Fluorescence Intensity Measurements
This application note describes the validation of the Tristar2 S LB 942 Multidetection microplate reader with the Transcreener® Fluorescence intensity assay from BellBrook Labs. The TriStar2 S combines the user friendliness of a multimodal optical system, with the sensitivity and performance of a dedicated optical device. It has been developed for full modularity and equipped with the proprietary ONE-4-ALL optical system.
Using the Mithras LB 943 for High-Performance Fluorescence Intensity Measurements
This application note describes the validation of the Mithras2 LB 943 Monochromator Multimode reader with the Transcreener® Fluorescence intensity assay from BellBrook Labs. The Mithras² LB 943 is a high-end microplate multimode reader based on monochromator technology with excellent performance. Characterized by its sensitivity and robustness, especially in luminescence and BRET measurements, the reader supports all important reading technologies.
Improved Versatility of the Transcreener® ADP2 Assay: Alternative Fluorescence Detection Modes for All Major Instrument Platforms
This poster from BellBrook Labs looks at alternative fluorescence modes when measuring ADP formation for screening drugs. These methods can be used with most plate readers used today. It also includes a new EZ protocol showing a linear relationship between ATP Concentration and ADP2 Antibody concentration.
Transcreener® ADP2 TR-FRET Red Assay: Homogeneous ADP Detection
In this study the utility of ADP detection as a means to screen inhibitor potency using a far-red, time resolved Förster resonance energy transfer (TR-FRET) format is described. This assay uses the widely adopted TR-FRET technique to take advantage of low compound interference common to this method and owed to its time-gated nature as signal is measured after most background fluorescence has subsided. This assay has proven both sensitive and robust with Z´>0.7 at low substrate conversion levels enabling enzymology during initial velocity conditions and is an important addition to the HTS toolkit.
Determination of Drug Residence Time in an HTS Format using Kinetic Analysis with the Transcreener® ADP2 Assay
This poster describes a systematic approach to determine the residence time of compounds using Abl1 and Aurora C as targets and several well characterized ATP-competitive drugs. Residence time can be determined by equilibrium binding measurements using methods such as surface plasmon resonance (SPR), which provides highly quantitative data but requires costly instrumentation and long experimental timelines. Transcreener® ADP2 Assays offer a highly sensitive and versatile method for estimating drug residence times for kinases using a multimode fluorescent plate reader.
Comparison of ADP Detection Methods Used for High Throughput Screening
ADP detection is an attractive approach for screening kinases and other ATP-utilizing enzymes because it provides a universal platform that can be used for any member of the kinase super family as well as many other ATP-dependent enzymes, regardless of the acceptor substrate. The three ADP detection approaches that have been developed into commercial HTS assay products are 1) direct immunodetection of ADP, which relies on antibodies that selectively recognize ADP in the presence of excess ATP, 2) enzyme-coupled detection, where the ADP is used to drive a cascade of detection enzymes that ultimately produces a fluorescent signal and 3) enzyme-coupled detection, where the residual ATP is first depleted and then ADP is converted to ATP and detected using luciferase. This poster compares the three methods with respect to assay principle, protocol and performance.
ADP Detection in FP, TR-FRET and FI Assay Formats: Versatile Kinase/ATPase Detection Without the Use of Coupling Enzymes
The Transcreener ADP2 Assay is a single addition, homogenous assay method that directly measures ADP formation in kinase or ATPase reactions. The first generation Transcreener ADP assay, introduced in 2006 as a far red fluorescence polarization (FP) assay, was recently improved by incorporating a new antibody (ADP2) that enables a 20-fold increase in sensitivity. Together these Transcreener ADP assays have been validated for high throughput screening with diverse targets including protein, lipid and carbohydrate kinases, carboxylases, and chaperonin ATPases. This poster compares these three assays with other nucleotide detection assay methods
A Simplified Protocol to Streamline Inhibitor Profiling with the Transcreener® ADP2 FP Assay
Transcreener® HTS is a universal, high throughput biochemical assay platform based on the detection of nucleotides, which are formed by thousands of cellular enzymes - many of which catalyze the covalent regulatory reactions that are central to cell signaling and are of great value as targets in drug discovery. The Transcreener® ADP2 FP Assay is a far-red, competitive fluorescence polarization (FP) assay based on the detection of ADP and is a simple, one-step homogenous detection assay. This Application Note describes how to streamline the process of inhibitor profiling with the Transcreener® ADP2 FP Assay eliminating the need to experimentally optimize the ADP2 antibody concentration and the need for a standard curve.
Determination of Km for ATP and Kemptide with Protein Kinase A Enzyme using Transcreener® ADP2 FP Assay
Transcreener® is a universal, high throughput biochemical assay based on detection of nucleotides, which are formed by thousands of cellular enzymes — many of which catalyze the covalent regulatory reactions that are central to cell signaling and are high value targets in drug discovery. This application note describes the use of the Transcreener® assay in the determination of Km for ATP and Kemptide with Protein Kinase A Enzyme.
Transcreener CD73 assay: Leveraging nucleotide detection to measure adenosine
Watch this webinar from Meera Kumar, Senior Scientist at BellBrook Labs, to learn more about CD73 as a target for immunotherapy, including information on how the transcreener CD73 assay was developed and how this assay can be used to help measure adenosine.
BellBrook Labs receive NIH grant to treat lupus and related autoimmune diseases
The grant will be used to accelerate the discovery of new treatments for autoimmune diseases by targeting the cGAS-STING pathway
TR-FRET readout now available with the Transcreener cGAMP cGAS Assay
With this addition, researchers can more easily screen for small molecule modulators of cGAS
Top resources and technologies to advance your drug discovery research
Exclusive interviews, new methods, free downloads and more to help advance your pharma and biopharma research & development