MassHunter BioConfirm Software
Confirm the sequence of proteins and post-translational modifications (PTMs), and characterize variants, before you start expensive testing.
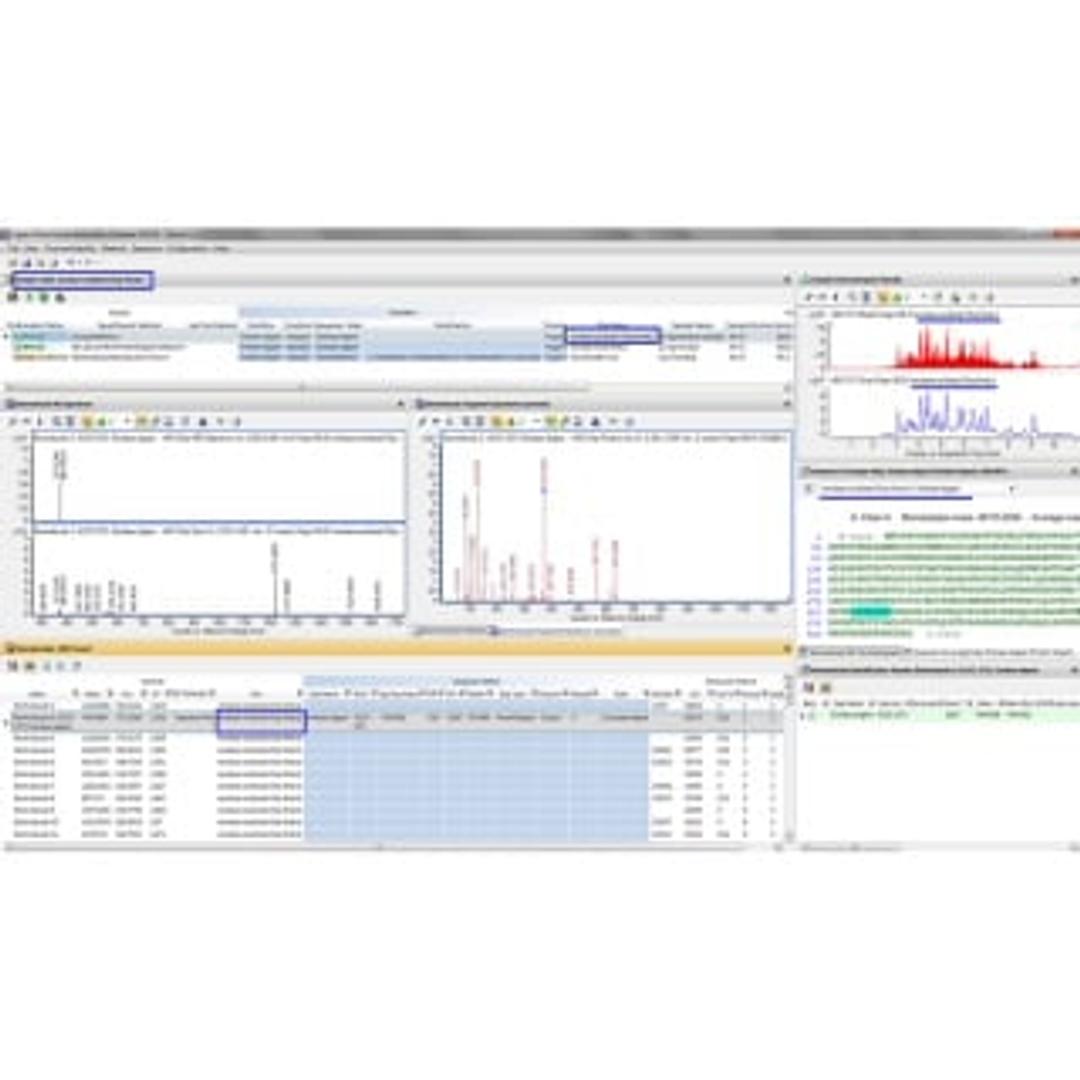
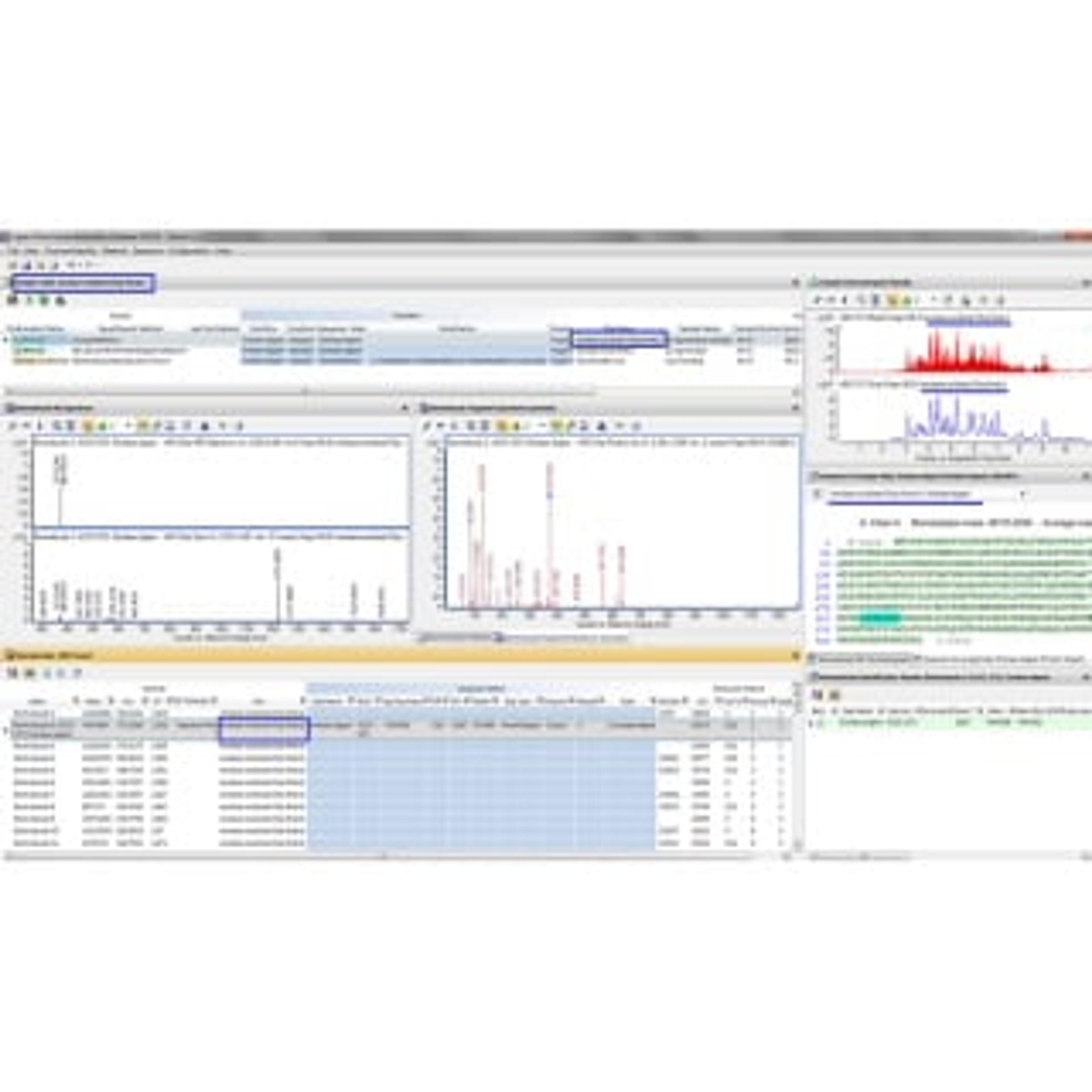
The beauty of Agilent MassHunter BioConfirm software is that it can help you confirm the sequence of proteins and post-translational modifications (PTMs), and characterize variants, before you start expensive testing. In fact, the software, with its drug-to-antibody ratio calculator, is part of a comprehensive solution for characterizing biopharmaceutical products such as monoclonal antibodies. The solution starts on sure footing: data from an Agilent 6200 Series Accurate-Mass Time-of-Flight LC/MS system or a 6500 Series Accurate-Mass Quadrupole Time-of-Flight LC/MS system. Then, because BioConfirm is fully integrated with the MassHunter Walkup environment, we’re able to deliver expert LC/MS capabilities that even non-experts can use with ease.
Features:
- Quickly set up of your analysis in one dialog
- Interpret your results with confidence using relative quantitation of intact proteins, protein digests, and released glycans.
- Compare results using histogram plots and tables.
- Dig deeper with improved identification of disulfide bond peptides.
- Review multiple samples (for example, different digest enzymes) at the same time using our graphically annotated Sequence Coverage Map.
For Research Use Only. Not for use in diagnostic procedures.
An integrated workflow for the analysis of oligonucleotides and their impurities by Agilent high-resolution LC/(Q-)TOF mass spectrometry
Oligonucleotides have become fast-growing modalities in recent years. Along with the development of these candidates comes the increased need for robust analytical methods and easy-to-use data analysis workflows to characterize them. In this application note, two automated and integrated oligonucleotide data analysis approaches were developed for the characterization of a target oligonucleotide and its related impurities using high-resolution accurate mass (HRAM) MS data.
In-depth peptide mapping with iterative MS/MS acquisition on the Agilent 6545XT AdvanceBio LC/Q TOF
Therapeutic monoclonal antibodies (mAbs) represent one of the fastest growing classes of protein-based drugs in the biopharmaceutical industry. Here, Agilent Technologies presents an integrated workflow for peptide mapping of a mAb, from automated sample preparation to data analysis, featuring an Agilent AssayMAP Bravo liquid-handling robot, the Agilent 1290 Infinity II LC system, the Agilent 6545XT AdvanceBio Q-TOF, and the Agilent MassHunter BioConfirm B.09 software.
N-glycan analysis: Better together
In this brochure, Agilent Technologies introduces the Agilent and ProZyme sample preparation workflows that are designed to help push your glycoprotein analysis to new levels of performance.
An Integrated Workflow for Automated Calculation of Antibody‑Drug Conjugate (ADC) Drug‑to-Antibody Ratio (DAR)
An easy-to-use and fully integrated method for the determination of drug‑to‑antibody ratios (DARs) of antibody-drug conjugates (ADCs) has been developed using the Agilent AssayMAP Bravo, Agilent MassHunter Walkup software, and the Agilent BioConfirm DAR calculator.
Integrated Workflow for Intact and Subunits of Monoclonal Antibody Accurate Mass Measurements
Monoclonal antibody (mAb) based entities represent a rapidly growing class of biologics that require extensive characterization to obtain approval for clinical trials and subsequent market release. Accurate mass measurement is a challenging step in the analytical characterization of antibodies because of their large size and the presence of post-translational modifications such as glycosylation. These characteristics also make determining the location of modifications more complex.
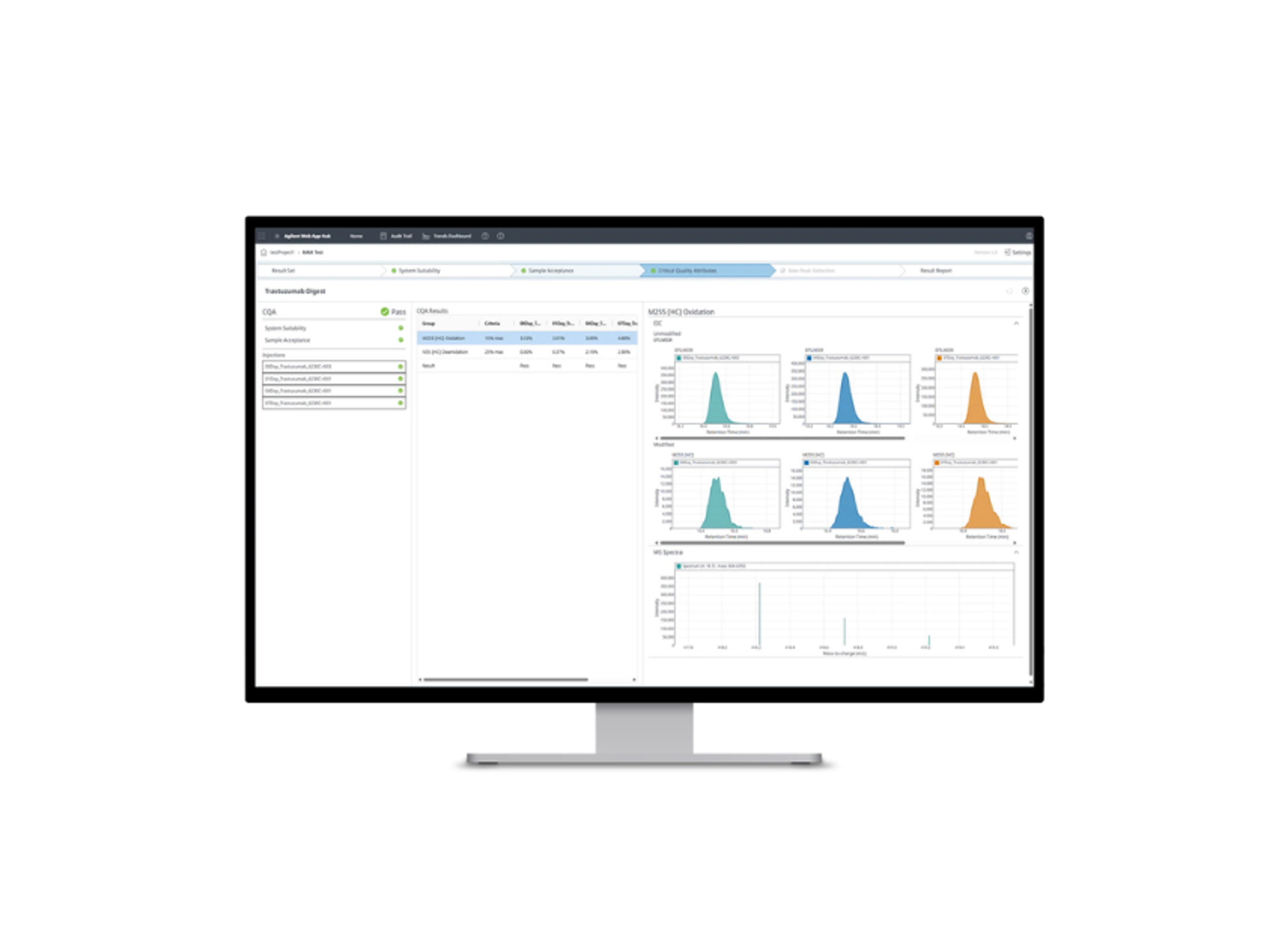
Optimize your workflow and confidently face any regulatory compliance audit
Is your lab audit ready? Ensure you have the tools to help you run a compliant lab. See how a unified software solution for LC/Q-TOF and LC/TOF with technical controls for compliant environments helps you confidently face regulatory audits.
Webinar Highlights – Enable New LC/MS Users to Perform Routine Analyses of Protein with Confidence
Find out how to set up an efficient multi-user/multi-project walk-up solution for protein biochemistry
Webinar Highlights: Obtaining Clear LC-MS Data for Complex Proteins – Your Questions Answered
Efficient analysis of intact proteins in a multi-user/multi-project walk-up environment