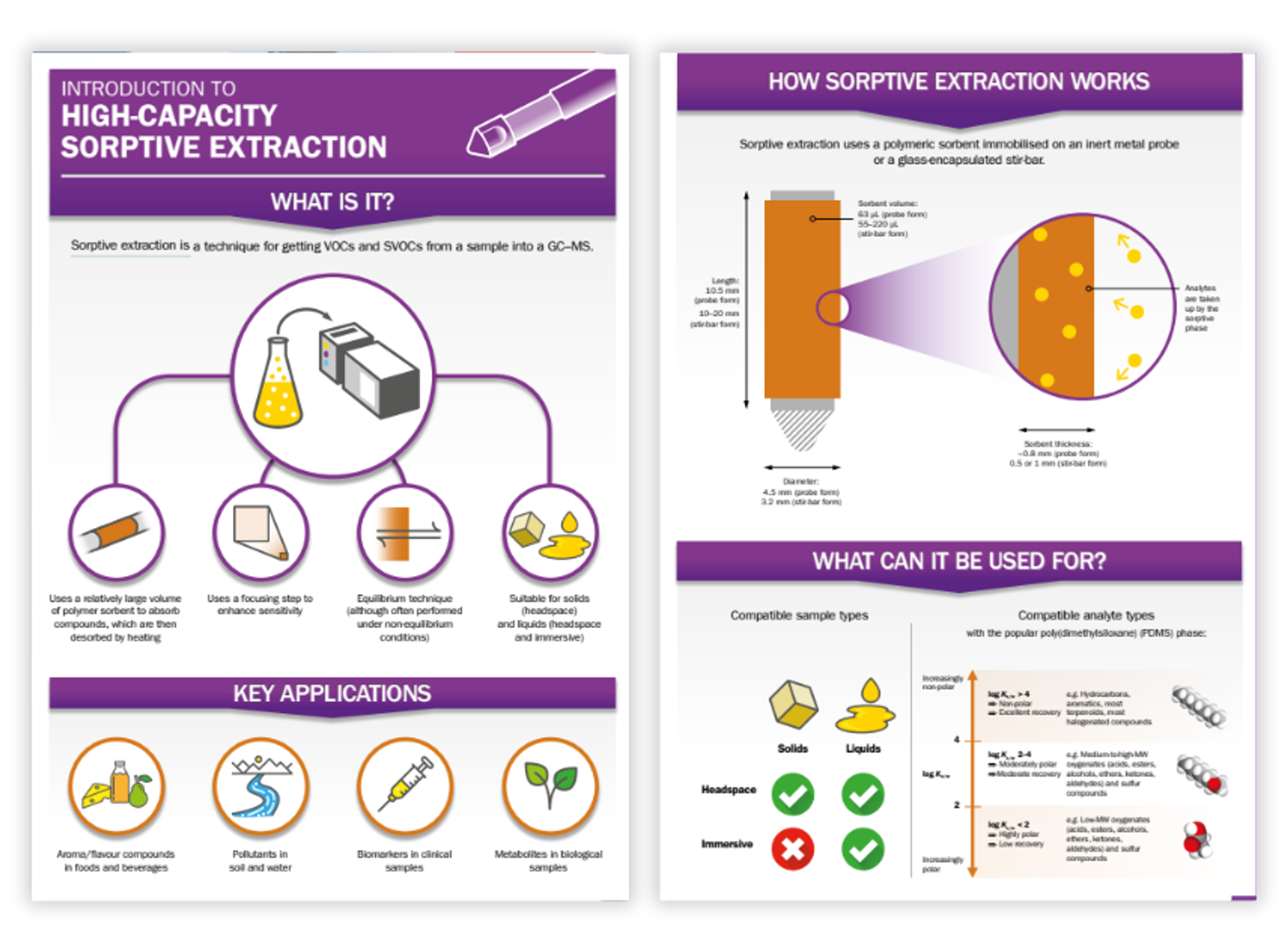
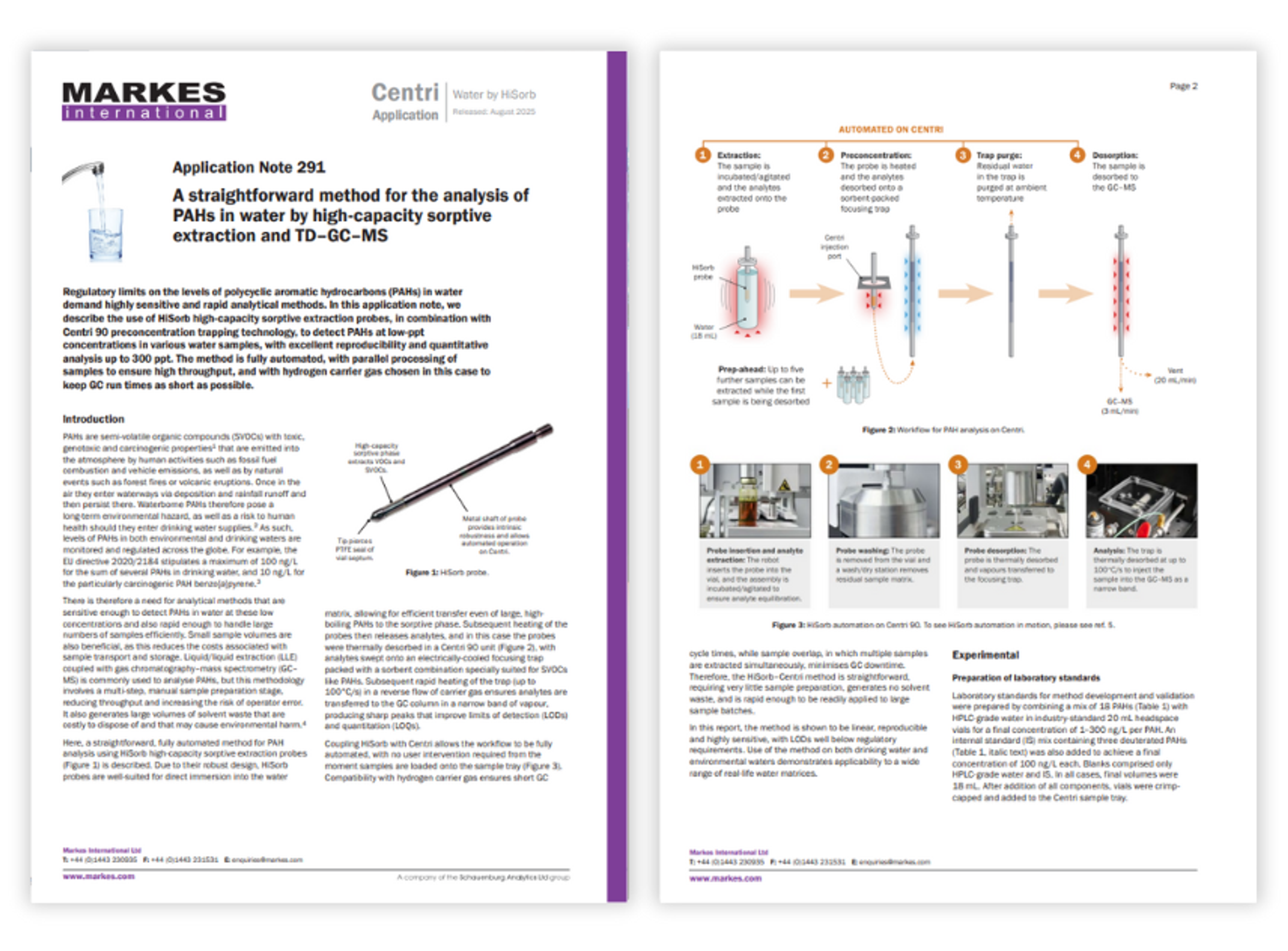
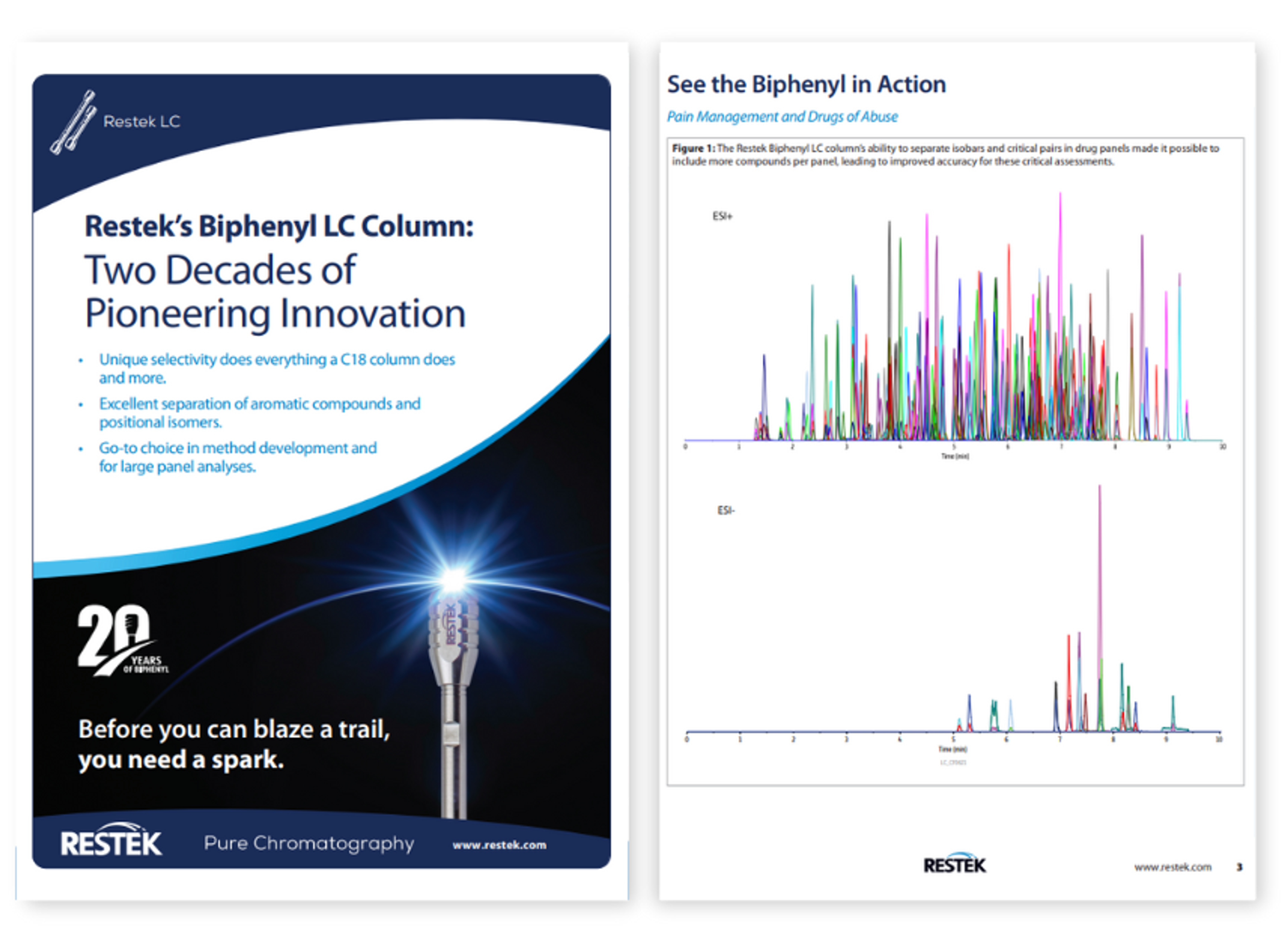
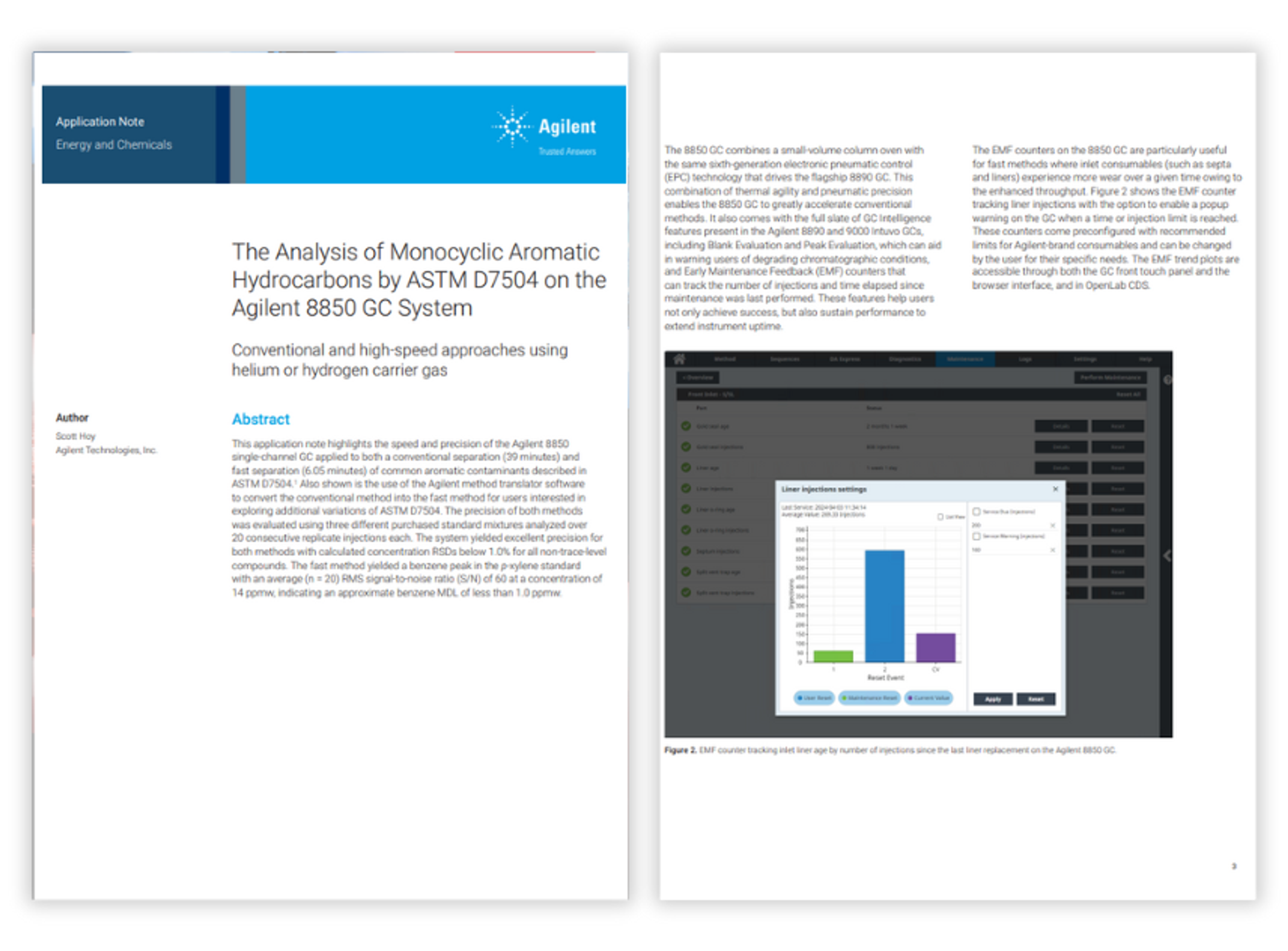
Resources
25
Selected Filters:
Product Brochures
Plate-based EV isolation and concentration with Summit
Application Note
Real-time reaction analysis
Product Brochures
PAL RSI and PAL RTC - Sample prep and injection
Product Brochures
PAL3 Autosampler: Accessories & smart consumables
Application Note
Effortless high-performance purification
Application Note
Pure Excellence ELSD
Application Note
Pure Excellence sample injection modes
Product Brochures
PAL Smart SPME Fibers: Optimized for automation
Product Brochures
PAL Smart SPME Arrow: The better SPME
Application Note
Chromapedia pocket guide
Application Note