Product Brochures
Resources
25
Selected Filters:
Product Brochures
Iterative Mapping Early Access Program
Product Brochures
Proteomic data atlases support drug discovery and target analysis
Application Note
7 ways to study protein phosphorylation
Application Note
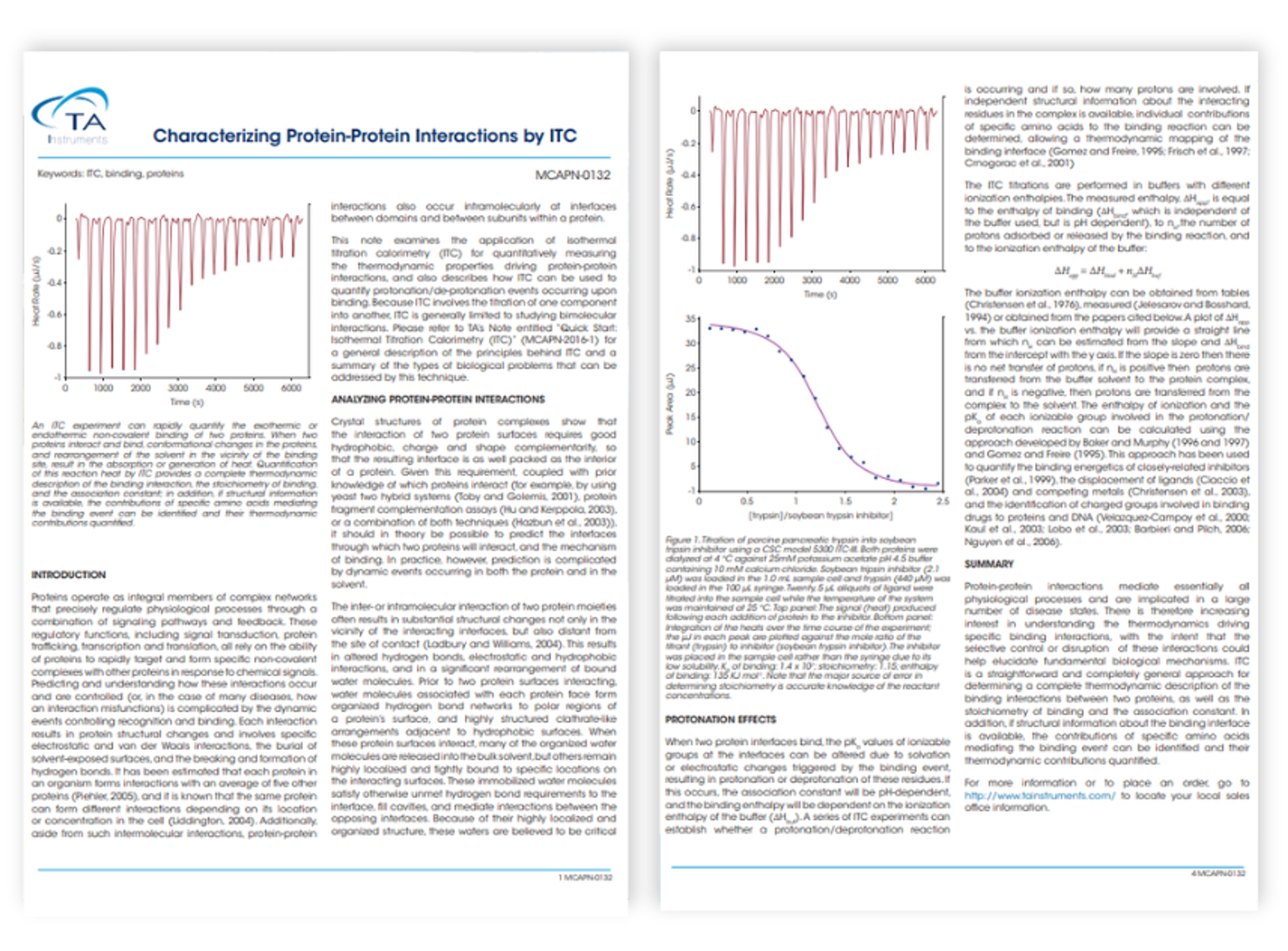
Characterizing protein-protein interactions by ITC
Application Note
Bioconjugation resource guide
Product Brochures
12 tools of click chemistry
Product Brochures
Click chemistry toolbox
Application Note
Unveiling the drivers of inflammatory bowel disease
Application Note
Determining protein turnover in fish with D7-leucine
Application Note
Top ten tips for producing 13C, 15N protein in abundance
Application Note