SureTect Real-Time PCR System
Rapid and accurate detection of foodborne pathogens
Great device, I can't do my research in easy way without it.
Discovery Biology
It is easy to use by following the protocol for a researcher. We use it for sophisticated research work. The company always provides efficient assistance when needed. It is a product that has completely satisfied our investment. The safety measures of this device are impressive to reduce contamination. I am totally impressed with the support of this device.
Review Date: 4 Dec 2021 | Thermo Fisher Scientific
Rapid and accurate detection of foodborne pathogens
Perform food safety tests confidently with the Thermo Scientific™ SureTect™ Real-Time PCR System. Built on proven PCR technology and backed by world-class service and support, the SureTect System is designed to quickly and accurately detect foodborne pathogens in a broad range of foods and associated samples. Take advantage of simple and streamlined workflows that utilize a single same-day or overnight enrichment step, direct lysis protocol for DNA release, and universal cycling parameters.
Choose the SureTect System PCR System for:
Simplicity:
- Single versus dual enrichment protocols for faster time-to-result and streamlined workflow
- Test for multiple targets from a single enrichment for maximum productivity
- Pre-dispensed reagents, reducing handling steps and risk of operational error
- Universal PCR conditions for detecting multiple targets in the same run
- ‘Plug and play’ ready-to-use instruments out of the box
Peace of mind:
- Validated for a broad range of targets and matrices according to AFNOR and AOAC certification requirements
- Fully supported by our dedicated and experienced technical teams
Flexible platform:
- Open instrument suitable for all our food safety, authenticity, and quality PCR tests
- Intuitive, user-friendly software, avoiding subjective interpretation
- Cloud-enabled, offering the potential for data sharing, analysis and trending
- Capacity for processing several hundred samples a day
Brochures
Visibly simple food pathogen testing
In this product brochure, Thermo Fisher Scientific demonstrates how you can transform your complete food safety testing workflow with the innovative Thermo Scientific™ SureTect™ Food Pathogen Detection PCR System, enabling you to work efficiently and release products quickly with a simple, rapid pathogen detection system validated for a broad range of matrices.
SureTect Real-Time PCR range for food pathogen detection
In this brochure, Thermo Fisher Scientific showcases the SureTect Real-Time PCR System and its full range of PCR solutions for food safety, authenticity, and quality testing.
Rapid and accurate detection of foodborne pathogens with the SureTect PCR range
In this brochure, Thermo Fisher Scientific showcases its SureTect Real-Time PCR System and portfolio of SureTect PCR Assays. The SureTect range is designed to provide accurate, rapid pathogen testing for a broad range of food, ingredients and environmental samples including; dairy, meat & poultry, seafood, produce, infant formula, chocolate samples, and more.
Rapid detection of spoilage organisms in beverages
Beverage producers face increasing pressure to ensure product integrity, quality, and brand protection in the face of complex spoilage risks. From yeast and mold contamination to spoilage bacteria, identifying and addressing these issues early is critical to maintaining quality and operational efficiency.
Discover how the Thermo Scientific™ SureTect™ Beverage Spoilage Multiplex PCR Assay provides a fast, broad-range molecular solution for detecting key spoilage microorganisms that threaten beverage quality.
The PCR automation advantage for food safety professionals
In an industry where precision, speed, and compliance are non-negotiable, this guide to PCR automation for food safety offers a comprehensive roadmap for laboratories ready to modernize their microbiological testing workflows. Developed in partnership with Thermo Fisher Scientific, this essential resource explores how automated PCR technologies are transforming food safety testing, reducing human error, increasing throughput, and ensuring regulatory compliance across global markets.
Inside, you'll discover how the Thermo Scientific™ SureTect™ PCR System delivers rapid, reproducible results for high-volume labs, with proven equivalency to manual workflows. Real-world case studies from Coca-Cola Europacific Partners and OMIC USA Inc. illustrate how automation has helped leading organizations overcome labor shortages, scale operations, and maintain accuracy under pressure. Whether you're testing for Salmonella, Listeria, E. coli, or other pathogens, this guide provides actionable insights into selecting the right workflow, manual or automated, based on your lab’s size, goals, and regulatory environment.
Download this SelectScience guide to explore:
- Learn how PCR automation can future-proof your lab against rising sample volumes and staffing challenges.
- Compare manual vs. automated workflows with detailed pros and cons.
- Explore validated methods and certifications (ISO 16140-2:2016, AOAC) for global compliance.
- Access implementation checklists, workflow audits, and expert recommendations to streamline your transition.
If your lab is ready to move beyond traditional culture-based testing and embrace a faster, more scalable molecular approach, this guide is your starting point.
Resource details:
- Document type: SelectScience guide
- Page count: 28
- Read time: 42 mins
- Edition: 1st
SureTect Assay workflow: Quick reference guide
This guide provides an overview of the Thermo Scientific™ SureTect™ Assay workflow.
SureTect Salmonella species PCR Assay workflow NF VALIDATION: ISO 16140-2:2016
The Thermo Scientific™ SureTect™ Salmonella species PCR Assay workflow has been certified by NF VALIDATION for the detection of Salmonella species from meat, dairy, seafood, vegetables, raw beef meats with and without aromatics, production environment samples and powdered infant formula samples. This application note gives a summary of the validation studies performed to gain NF VALIDATION.
SureTect Listeria monocytogenes PCR Assay workflow NF VALIDATION: ISO 16140-2 validation studies
The Thermo Scientific™ SureTect™ Listeria monocytogenes PCR Assay has been certified by NF VALIDATION for the detection of Listeria monocytogenes from meat, dairy, seafood, vegetable and production environment samples. This application note gives a summary of the validation studies performed as part of the NF VALIDATION.
SureTect Salmonella Species PCR Assay method comparison against ISO 6579-1:2017
In this application note, extended methods for the Thermo Scientific™ SureTect™ Salmonella species PCR Assay are compared against the International Organization for Standardization (ISO) 6579-1:2017 for a representative range of matrices.
SureTect E. coli O157:H7 PCR Assay method modification validation: Inclusivity and exclusivity
The Thermo Scientific™ SureTect™ E. coli O157:H7 species PCR Assay has been certified in accordance with the AOAC Research Institute Performance Tested Methods(PTM) Program to be used with the Applied Biosystems™ QuantStudio™ 5 Real-Time PCR Instrument to perform PCR and with Thermo Scientific™ RapidFinder™ Analysis Software version 1.0 or greater for data analysis. This application note details the inclusivity and exclusivity part of the validation.
SureTect E. coli O157:H7 PCR Assay method comparison study
In this application note, the Thermo Scientific™ SureTect™ E. coli O157:H7 PCR Assay method is compared against the United States department of agriculture food safety inspection service microbiology laboratory guidebook 5.09 reference method for raw beef and the ISO 16654:2001 reference method for fresh spinach.
SureTect Cronobacter species PCR Assay workflow NF VALIDATION: ISO 16140-2:2016
The Thermo Scientific™ SureTect™ Cronobacter species PCR Assay has been certified by NF VALIDATION for the detection of Cronobacter spp. from powdered infant formula and production environment samples. In this application note, a summary of the studies performed as part of the NF VALIDATION is provided.
Salmonella detection from large milk powder samples
Due to their ability to survive heating and desiccation, Salmonella and Cronobacter contamination is a concern for powdered infant formula (PIF), milk powders and their individual ingredients. In this application note, two studies are presented that evaluate the performance of the SureTect Salmonella species PCR Assay for the detection of Salmonella serovars from 375 g samples of milk powders, in comparison to ISO 6579-1:2017.
How to navigate the food regulatory landscape for Listeria spp. and Listeria mono. testing
Friday, November 21, at 13:00 GMT | 14:00 CET | 08:00 EST | 05:00 PST
Listeriosis remains a serious public health concern. In 2023, thirty EU/EEA countries reported 2,993 confirmed cases, the highest annual total since surveillance began. In the U.S., around 1,250 cases occur each year, leading to approximately 1,070 hospitalizations. Between 2016 and 2023, a multi-country cluster linked to ready-to-eat foods resulted in 64 cases across five EU/EEA countries
These persistent outbreaks continue to challenge food producers worldwide and in response to this, regulatory requirements globally have been rapidly evolving in recognition of these threats. Remaining compliant in this fast-changing landscape adds further complexity for testing laboratories.
Regulatory bodies have responded to these threats with revisions and tightening of the current regulatory frameworks for Listeria spp. and L. monocytogenes:
- FSIS broadened its testing to include non‑Listeria monocytogenes species from January 17, 2025.
- EU Regulation 2024/2895 sets a '0 cfu/25 g' requirement for Listeria in ready‑to‑eat foods.
Register for this exclusive webinar to hear from microbiology experts, Katharine Evans, François Le Nestour and Dr. Salman Zeitouni as as they discuss how labs can stay ahead by adopting internal standards that exceed minimum regulatory requirements and by implementing robust, adaptable pathogen‑testing programs.
Key learning objectives:
- Uncover the fundamentals of Listeria and its pathogenicity
- Review major Listeria outbreaks and their human impact
- Decode recent regulatory changes from the US and EU on Listeria
- Learn how to design and validate testing protocols that go beyond minimum regulatory requirements, ensuring continuity when regulations change
- Explore Thermo Fisher Scientific's microbiology solutions for rapid detection and enumeration of Listeria and Listeria spp.
Who should attend?
- Food testing professionals
Certificate of attendance
If you attend the live webinar, you will automatically receive a certificate of attendance, including a learning outcomes summary, for continuing education purposes.
If you view the on-demand webinar, you can request a certificate of attendance by emailing editor@selectscience.net.
Reaching new heights in food safety testing using automated PCR workflows
Tuesday, September 30, at 15:00 BST | 16:00 CEST | 10:00 EDT | 07:00 PDT
Food safety testing laboratories are under growing pressure to do more with less. As testing volumes surge, labs must deliver accurate and regulatory compliant results, while navigating staffing shortages and limited resources. These challenges are further compounded by increasing sample sizes and throughput demands.
In this webinar, meet with Dr. Salman Zeitouni and Jani Holopainen to explore real-world examples of how automation is driving transformation in food safety testing laboratories. See how labs are leveraging automated solutions to enhance accuracy and efficiency, reduce human error, and build resilient testing capabilities.
Key learning objectives:
- Explore the drivers behind automation of molecular workflows in food safety testing laboratories
- Understand key considerations and common challenges in adopting lab automation
- Discover real-world examples of successful automation implementation in microbiology labs
- Gain insight into how automated molecular workflows can support lab scalability, traceability, and long-term resilience to build a future lab today.
Who should attend?
- Laboratory professionals, including food microbiologists, lab managers, and technical service directors.
- Quality, safety, and regulatory leaders, such as quality managers, operations directors, and compliance professionals.
Certificate of attendance
If you attend the live webinar, you will automatically receive a certificate of attendance, including a learning outcomes summary, for continuing education purposes.
If you view the on-demand webinar, you can request a certificate of attendance by emailing editor@selectscience.net.
Screen for pathogens in four simple steps
In this video, discover how using the SureTect Real-Time PCR System you can quickly and accurately detect foodborne pathogens in a broad range of foods and associated samples, in just four simple steps.
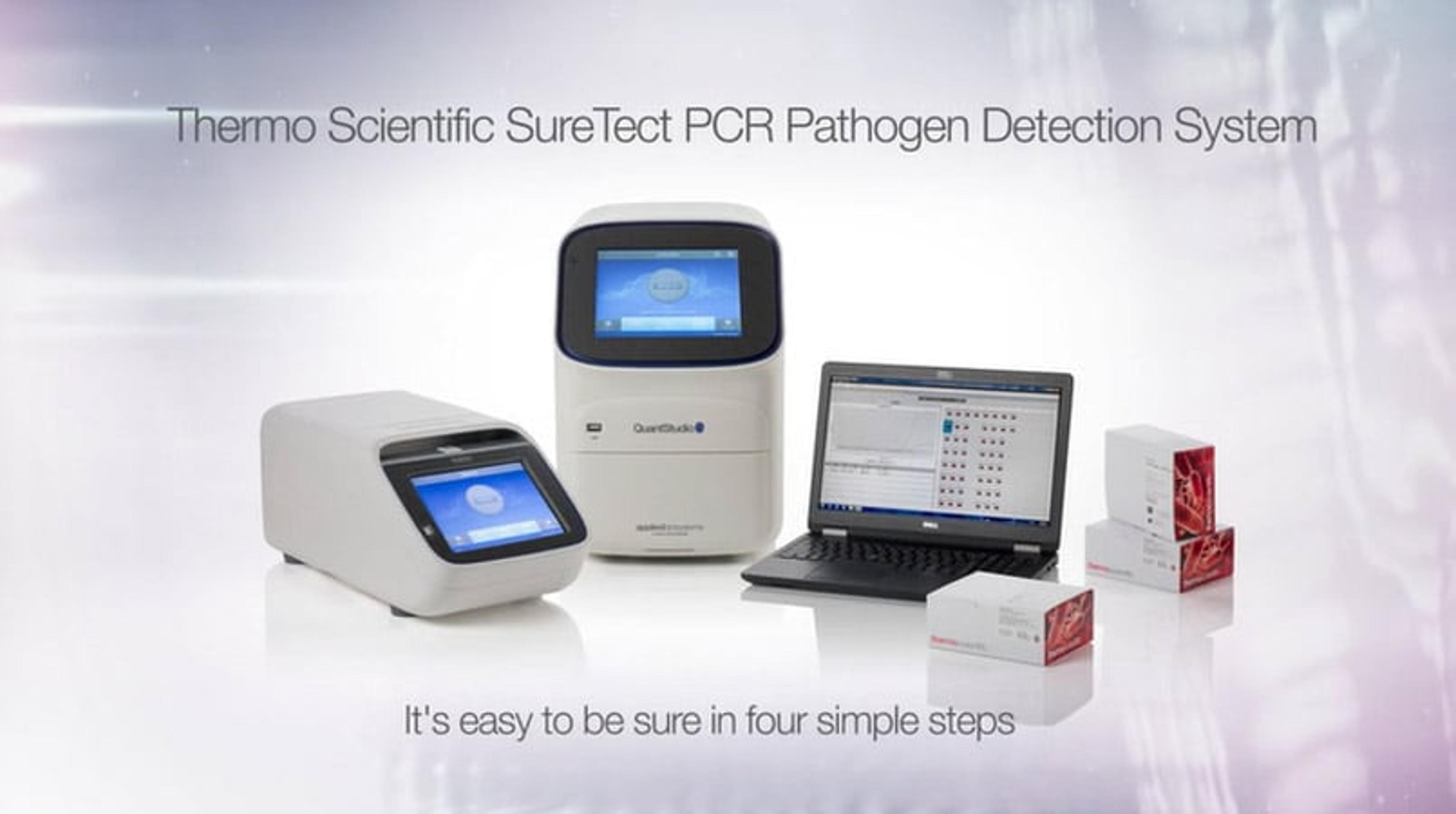
It's easy to be sure with the SureTect PCR Pathogen Detection System
In this video, learn how the SureTect workflow can provide all the information you need for food safety, authenticity, and quality testing, using a system that is rapid, simple to implement, and easy to run on a day-to-day basis.
Single‑assay PCR method transforms how labs detect beverage spoilage
Multiplex PCR assay enables beverage microbiology labs to accelerate product release testing by up to six days
Latest validation offers food laboratories additional layer of confidence when testing for Salmonella
The Thermo Scientific SureTect Salmonella Species PCR Assay receives approval by AOAC International for a broad range of foods
Thermo Scientific launches SureTect PCR Assays for expanded foodborne pathogen analysis
New assays for Campylobacter, STEC, Vibrio and Staphylococcus aureus enable more comprehensive pathogen testing for all food manufacturers
Thermo Fisher Scientific launches new test workflow for SARS-CoV-2 from food packaging and environmental monitoring
The workflow is designed to minimize the potential for contamination of the food supply and risk to handlers and consumers