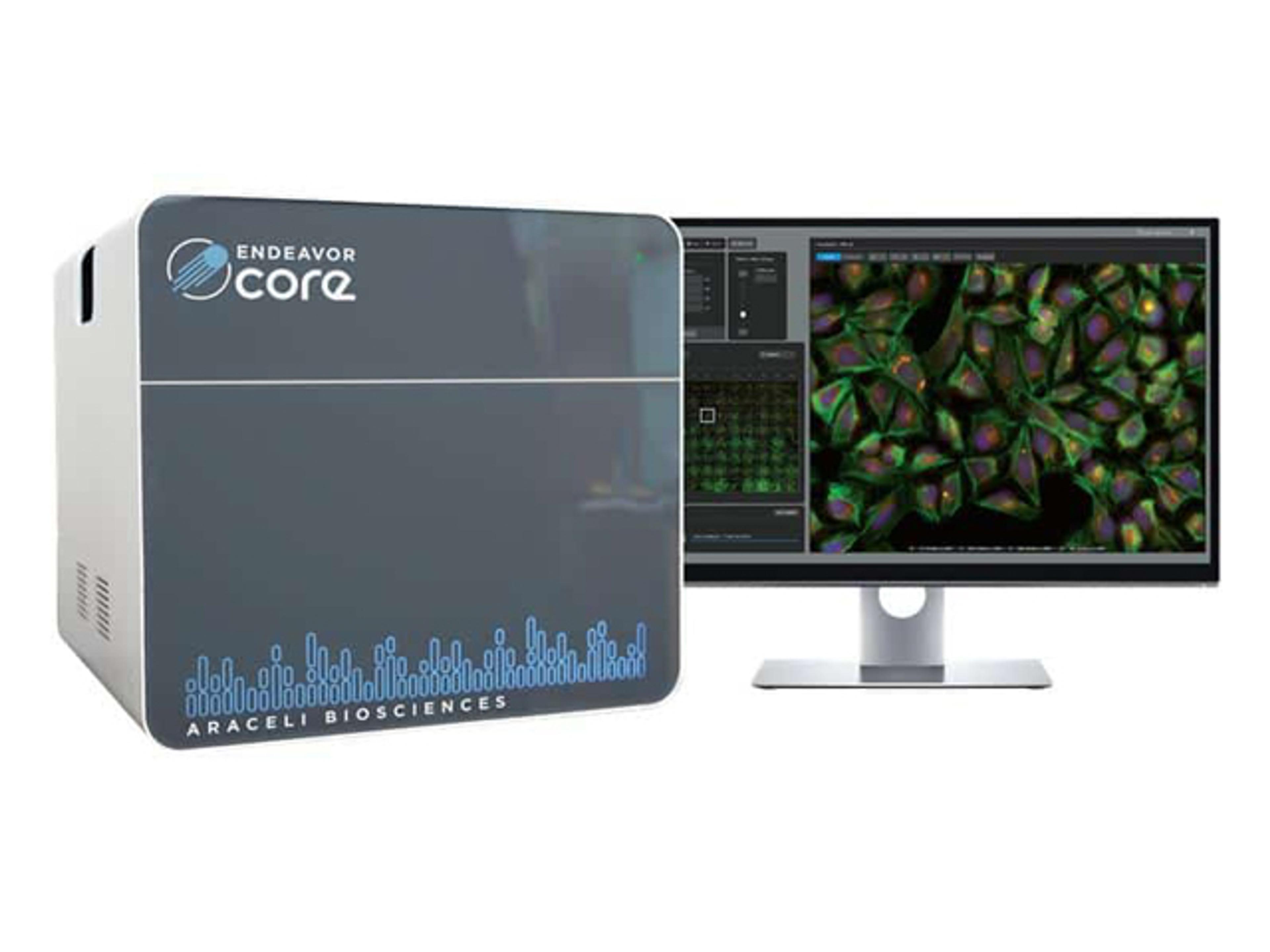
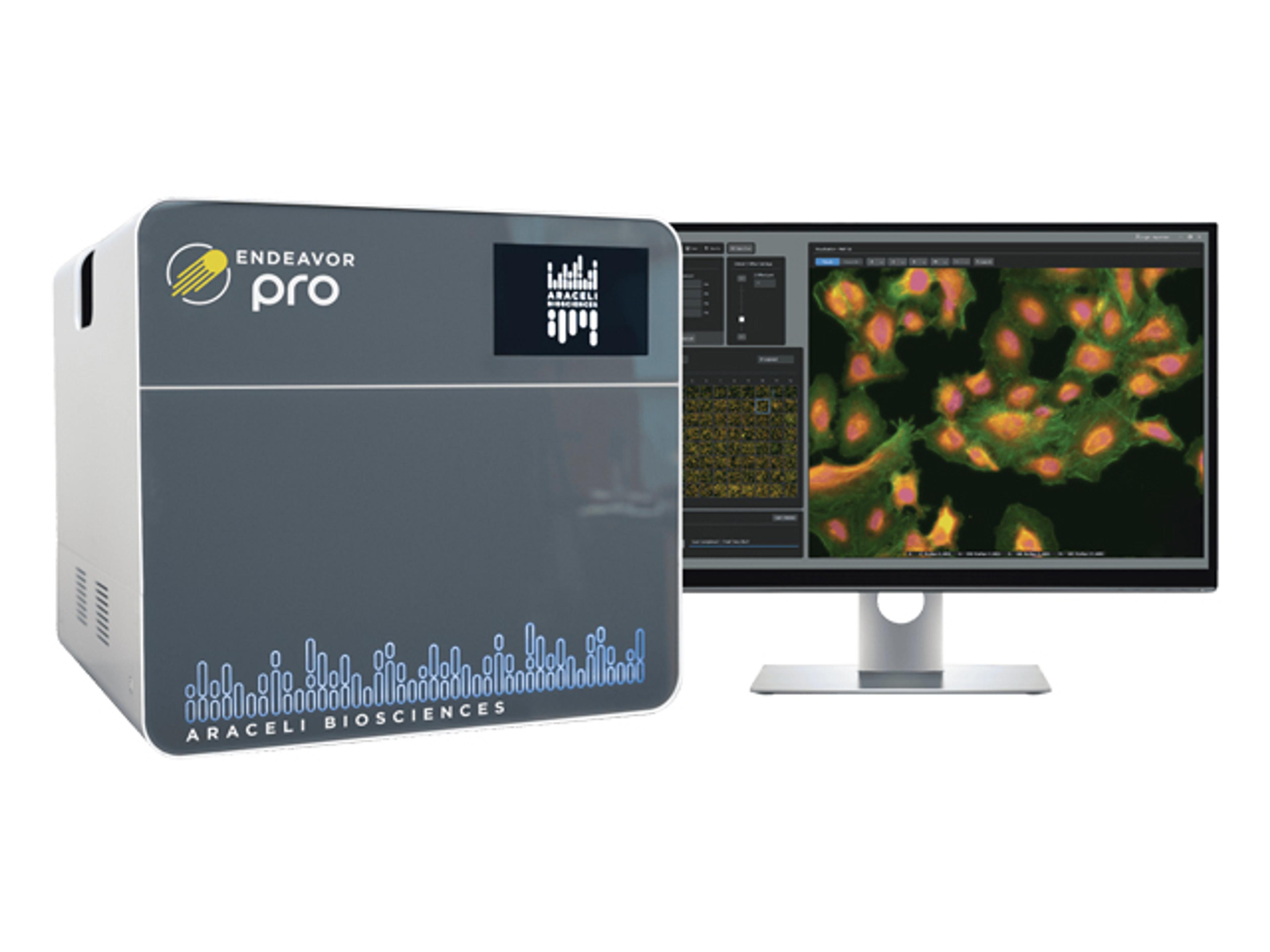
Stability Program Software Application
The Stability Program Software Application is a software application that manages the day-to-day activities of the stability department within the quality control and/or R&D divisions. Its design takes into consideration the latest guidelines from the FDA, TPP, ICH and EU, among others, pertaining to pharmaceutical, chemical, biological and biotechnological fields. This software is designed for all types of products, from pre-…

The supplier does not provide quotations for this product through SelectScience. You can search for similar products in our Product Directory.
The Stability Program Software Application is a software application that manages the day-to-day activities of the stability department within the quality control and/or R&D divisions. Its design takes into consideration the latest guidelines from the FDA, TPP, ICH and EU, among others, pertaining to pharmaceutical, chemical, biological and biotechnological fields. This software is designed for all types of products, from pre-clinical to post market, innovator or generic. The Stability module is 21 CFR part 11 compliant and has an extensive, independent audit trail.
Trend window is used to demonstrate the trend of the results for each product in the stability program through statistical calculations. Calculations of P-value and Arrhenius equation are also available.