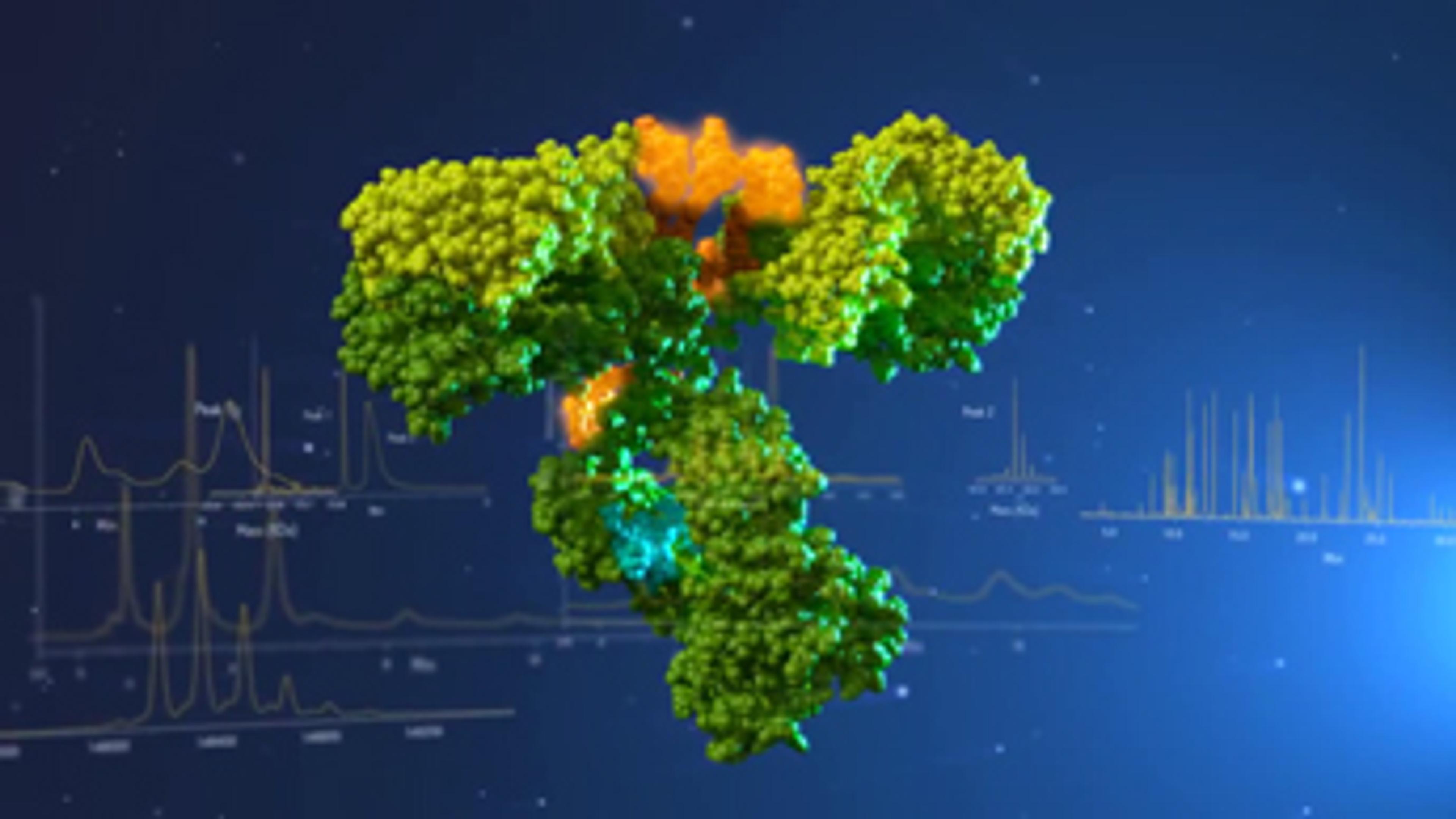
BiopharmaLynx
BiopharmaLynx™ automates the processing and interpretation of biopharmaceutical product LC/MS data, enabling you to characterize your products. Developed with biopharmaceutical thought leaders for confident characterization and optimized workflow, BiopharmaLynx™ leverages Waters' leading protein informatics expertise. Determine the identity, purity and stability of biopharmaceuticals, working with either peptide maps or intac…

The supplier does not provide quotations for this product through SelectScience. You can search for similar products in our Product Directory.
BiopharmaLynx™ automates the processing and interpretation of biopharmaceutical product LC/MS data, enabling you to characterize your products.
Developed with biopharmaceutical thought leaders for confident characterization and optimized workflow, BiopharmaLynx™ leverages Waters' leading protein informatics expertise. Determine the identity, purity and stability of biopharmaceuticals, working with either peptide maps or intact proteins.
BiopharmaLynx Features:
- Automatically process your accurate mass MS data, including peptide map results and intact mass measurements.
- Automatically analyze and assign results, define the sequence and features of known proteins and determine the identity of modified forms.
- Allow your users to edit assignments, annotate new peaks and compare experimental samples with a reference using visualization and tabular tools.
- Automatically generate biopharmaceutical-specific reports from templates.
- Identify and quantify ions in a peptide map, including intra- and inter-sample relative quantitation.
- Assign ions to expected and modified peptides from a defined protein sequence, determining sequence coverage and ID-modified peptides.
- Define deconvolution settings for known proteins and as generic for unknown proteins.
- Annotate deconvoluted mass data with protein identities and their modifications.
- Automatically calculate percentages of each protein variant.
Analysis of Antibody Drug Conjugates
As biopharmaceutical organizations explore the use of Antibody Drug Conjugates (ADC) as a class of cancer therapies, significant challenges are found in analyzing their complex, heterogeneous structures. The determination of drug-to-antibody ratios (DAR) and sites of conjugation presents unique analytical challenges that can be addressed with Waters' fit-for-purpose UPLC, QTof MS, and informatics solutions.