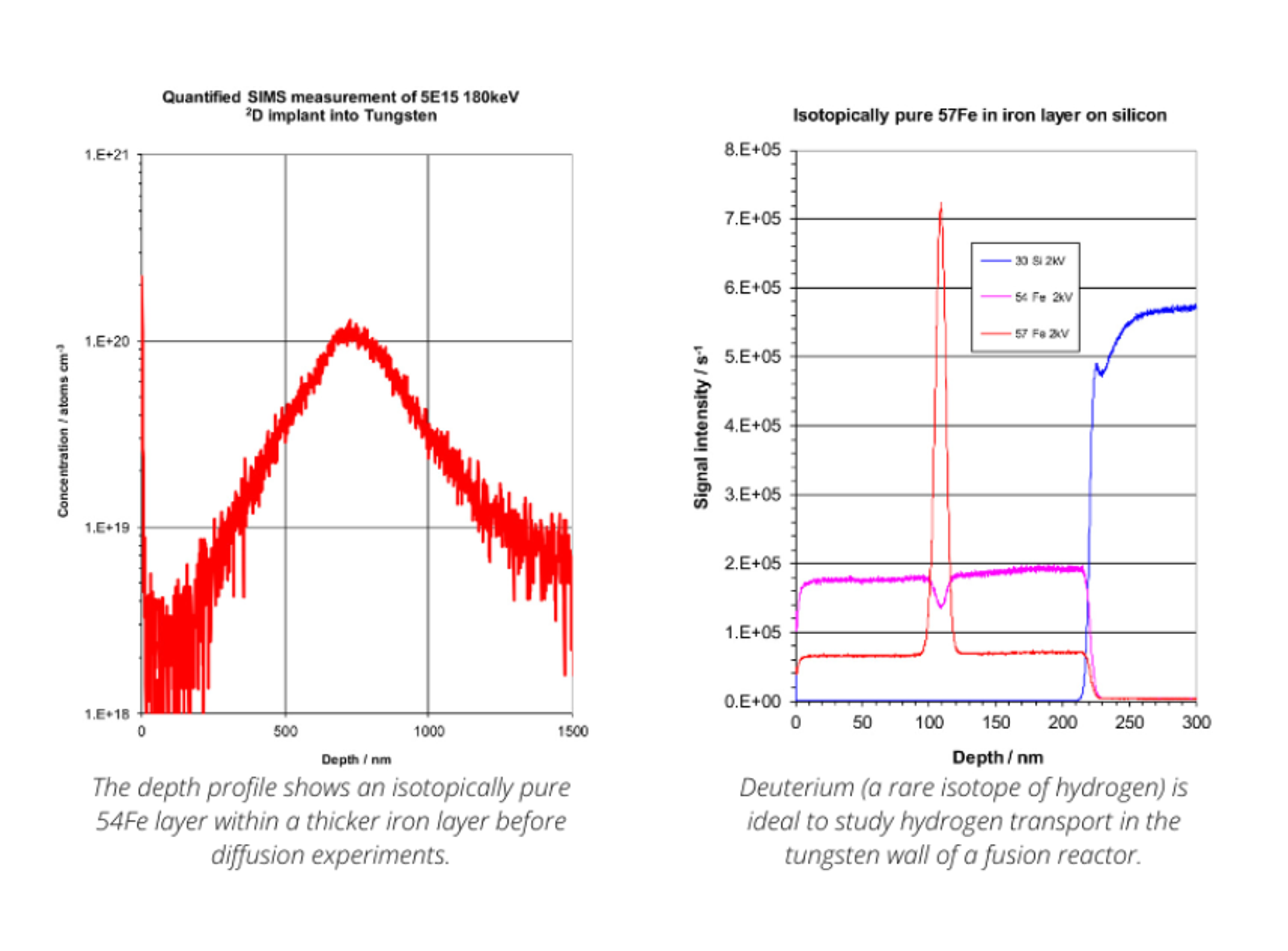
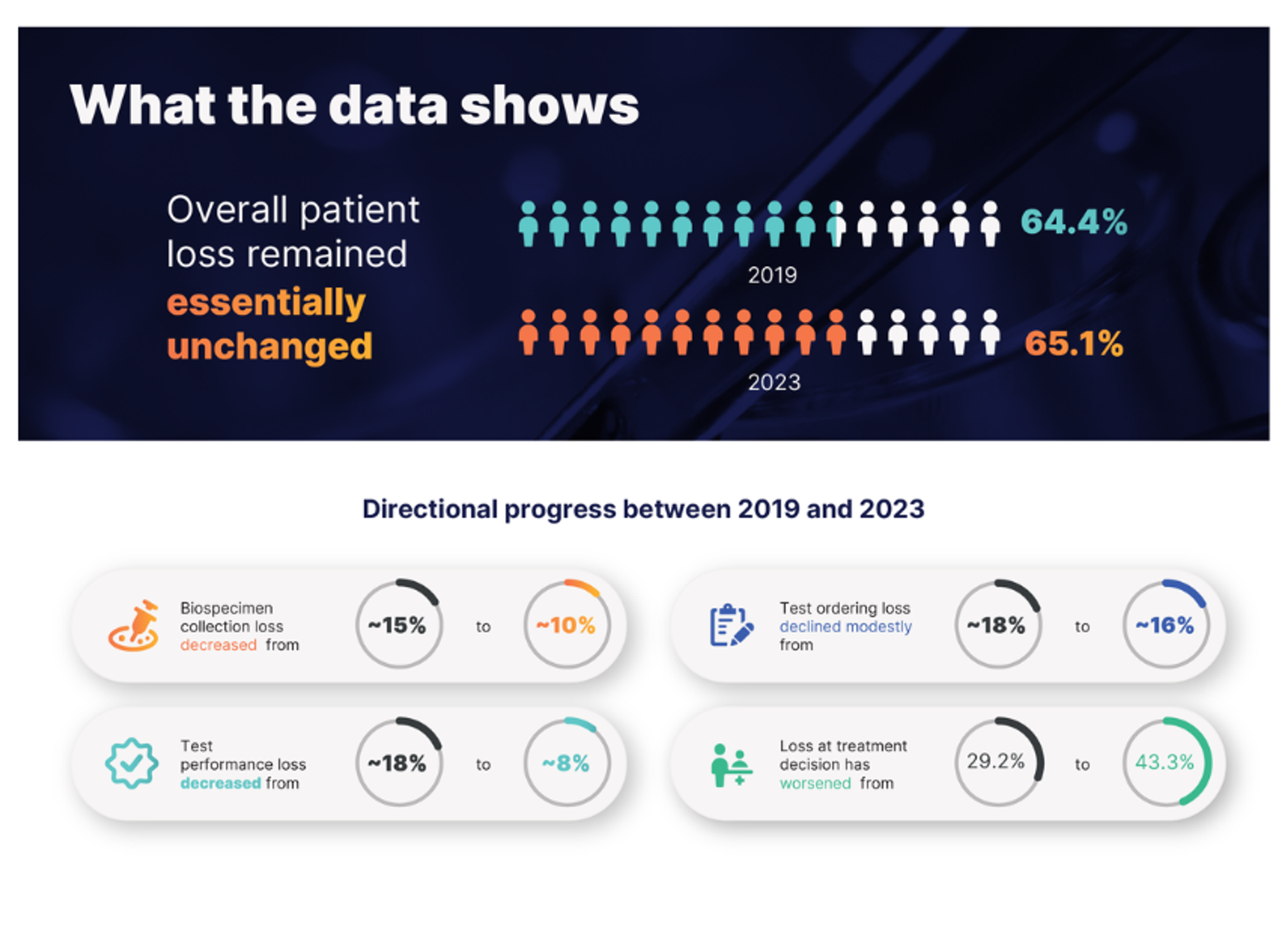
Candida auris and emerging diagnostics: A strategic look ahead in antimicrobial resistance
As Candida auris and carbapenem-resistant Enterobacterales rise, join us online to explore how emerging approaches such as next-generation sequencing and rapid susceptibility testing may support antimicrobial stewardship — plus, ask your questions in the live Q&A