Continuous Live Imaging Reveals Why Some Prostate Cancer Cells Resist Androgen-Deprivation Therapy
Cancer biologist, Fiorella Magani, shares her studies on the metastasis and behavior of castration-resistant prostate cancer cells in this SelectScience interview
12 Oct 2017
A common therapy for prostate cancer is to lower the levels of male sex hormones, i.e. androgens, that are considered to drive cancer growth. Known as androgen deprivation, the level of these male hormones can be curbed by drugs that stop their production in the testicles or by surgery. However, some prostate cancers metastasize and spread to other parts of the body, despite the treatment or surgery – a condition known as castration-resistant prostate cancer.
In this SelectScience® interview, we speak with Fiorella Magani, a cancer biologist at the University of Miami, who investigates why some prostate cancers don’t respond to androgen-deprivation therapy. A graduate student in Dr. Kerry L. Burnstein’s lab at the University of Miami, Magani describes the methods that she uses to examine cancer cells in real time.

Fiorella Magani Fiorella Magani, graduate student at the University of Miami, Florida, studies castration-resistant prostate cancer in Dr. Kerry L. Burnstein’s lab.
“Castration-resistant prostate cancer is an incurable stage of the disease that causes 20,000 deaths in the US every year,” says Magani. There is a pressing need to better understand the molecular basis of prostate cancer metastasis and resistance to therapy. The androgen receptor (AR) is said to have a strong implication for prostate cancer progression. “Constitutively active androgen receptor splice variants (AR-Vs) represent a well-established mechanism of therapeutic resistance and disease progression of prostate cancer,” explains Magani. “These are not inhibited by androgen-deprivation therapy.”
In numerous studies, AR-Vs, identified from human prostate cancer cell lines and patient samples, lack the ligand-binding domain, thereby being unaffected by androgen-deprivation therapy and amounting to exacerbation of the prostate cancer. One such androgen receptor splice variant found in circulating tumors of metastasized prostate cancers is AR-V7. “My research focuses on studying the interaction of the AR-V splice variants and their coactivators as a novel therapeutic approach for castration-resistant prostate cancer,” says Magani.
“I also work on identifying novel genes downstream of AR-V7 that may serve as therapeutic targets,” adds Magani. The downstream signaling triggered by AR-V7, responsible for hormone regulation and, potentially, cancer progression, is boosted when in association with a coactivator, Vav3. This signaling pathway serves as a hot target candidate for prostate cancer therapies.
Using mutational experiments, the Burnstein lab deciphered the interaction between AR-V7 and Vav3, the coactivator. By disrupting this interaction on castration-resistant prostate cancer cell lines, they were able to curb cell proliferation, a characteristic feature of cancer cells, and decrease cell migration, a trait of metastasis. Plus, they observed more apoptosis of cancer cells. These findings, published in Mol Cancer Res., 2017, suggest that inhibiting the interaction between AR-V7 and Vav3 has a therapeutic potential for castration-resistant prostate cancer.
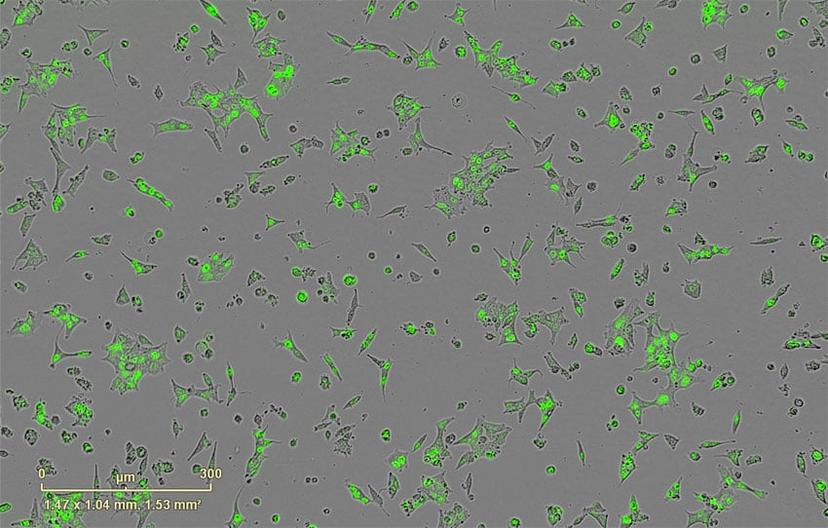
With more projects underway in the Burnstein lab, Magani now employs techniques such as RNA-seq and ChIP-seq in combination with biochemical methods on cancer cell lines, including shRNA gene depletion, luciferase assays, and live cell imaging. “Continuous live-cell imaging has allowed me to monitor, measure and create videos of single-cell apoptotic events in real time,” says Magani. “In this case, an androgen-dependent prostate cancer cell line was placed in media without androgens, which induced apoptosis.”
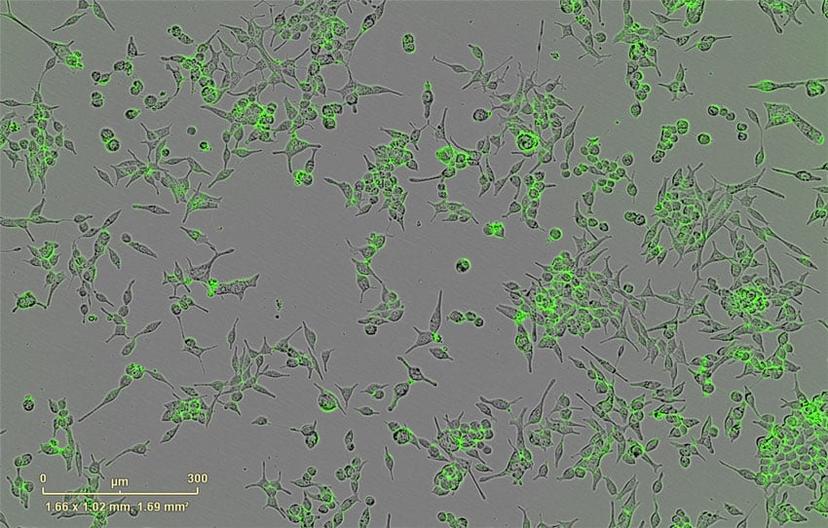
The Burnstein lab uses the IncuCyte® to image the morphology and behavioral properties of the cancer cell lines. “What I like best about the IncuCyte is that it makes my analysis faster and more reliable,” says Magani, who performs time-lapse live-cell imaging on the system. “Before acquiring this machine, I had to seed many plates for my experiments and manually count cells using a hemocytometer.”
With continued efforts to understand the role of AR-V7 in cancer metastasis, the members of the Burnstein lab will continue to closely monitor the cancer cell lines. “I have now used the IncuCyte to test the effects of single gene depletion and drug combinations in multiple cell lines, with minimal effort. The IncuCyte helps me perform multiple long-term experiments at the same time while increasing my number of replications and, thereby, reducing my standard error. It has saved us a lot of money on reagents and, most importantly, it has saved us time,” says Magani.
The ease-of-use and the versatility of the imager have spurred Magani’s interests in different directions. “Because performing experiments is so much easier with the IncuCyte, I have ventured more into exploring and testing new ideas, that, before having the Incucyte, I would have probably not tried.”
In the future, the Burnstein lab will delve deeper into the interaction between AR-V7 and Vav3 and other AR-V7 networks, eventually moving on to in vivo experiments to study castration-resistant prostate cancer.
Find out more about the IncuCyte Live Cell Analysis System by Essen Bioscience.
Did you enjoy this article? Sign up now to receive more articles like these and other exclusive content straight to your inbox.

