Resources
25
Application Notes
Microchip CE-MS: A powerful tool for biotherapeutic characterization
Product Brochures
Effortless automation with VEYA
Application Notes
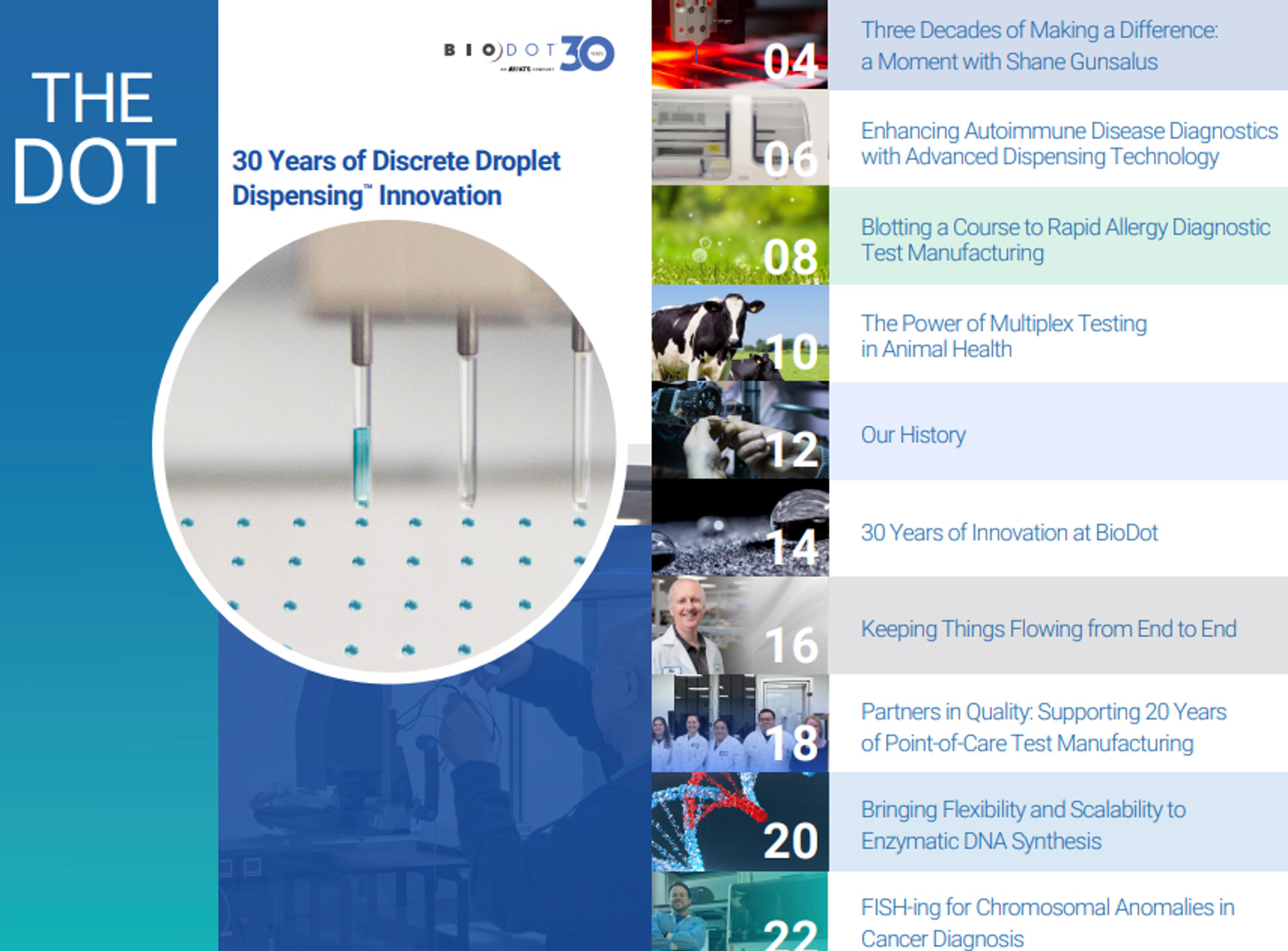
30 years of Discrete Droplet Dispensing innovation
Product Brochures
Countstar Mira HT product brochure
Product Brochures
Galileo Series 2: Your ultimate lab partner in microtomy
Product Brochures
Araceli Endeavor portfolio
Application Notes
Sample types and criteria for the diagnosis of diabetes
Application Notes
Enhance your gestational diabetes workflow with HemoCue
Application Notes
HemoCue women’s health patient survey summary
Product Brochures
Custom cGMP Oligos
Product Brochures
Contrast in oligonucleotides
Application Notes
Evaluating side-by-side placement of class II biosafety cabinets
Product Brochures
Upgrade your sample quality control workflow
Application Notes