Resources
25
Application eBook
2.5D cell culture models for more predictive in vitro research
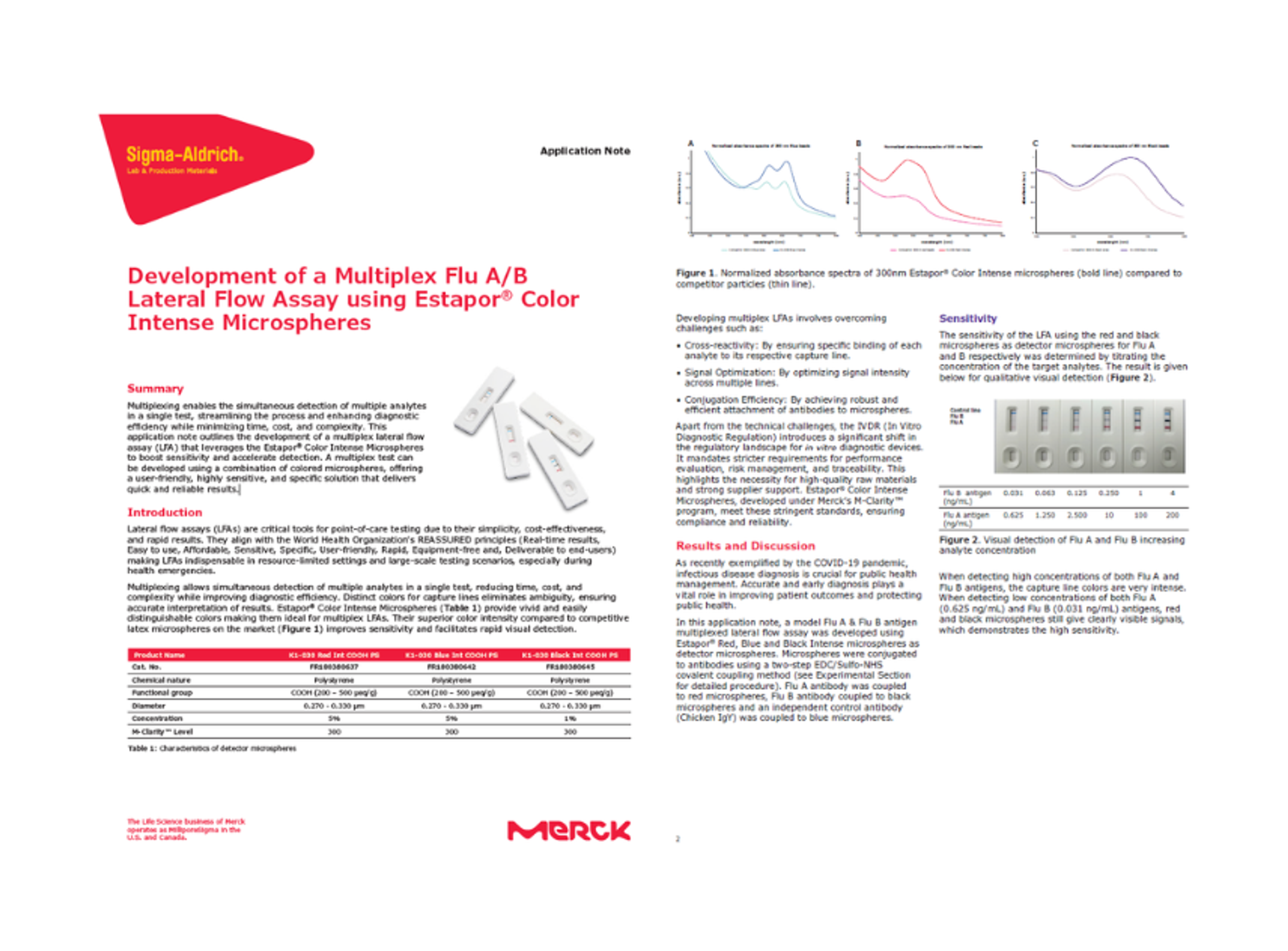
Application Note
Not all digital PCR is created equal
Application Note
The complete guide to stain-free Western blotting
Application Note
Protein gel electroporesis handbook
Product Brochures
Countstar Spica M6 brochure
Application Note
Spatial imaging demystified
Application Note
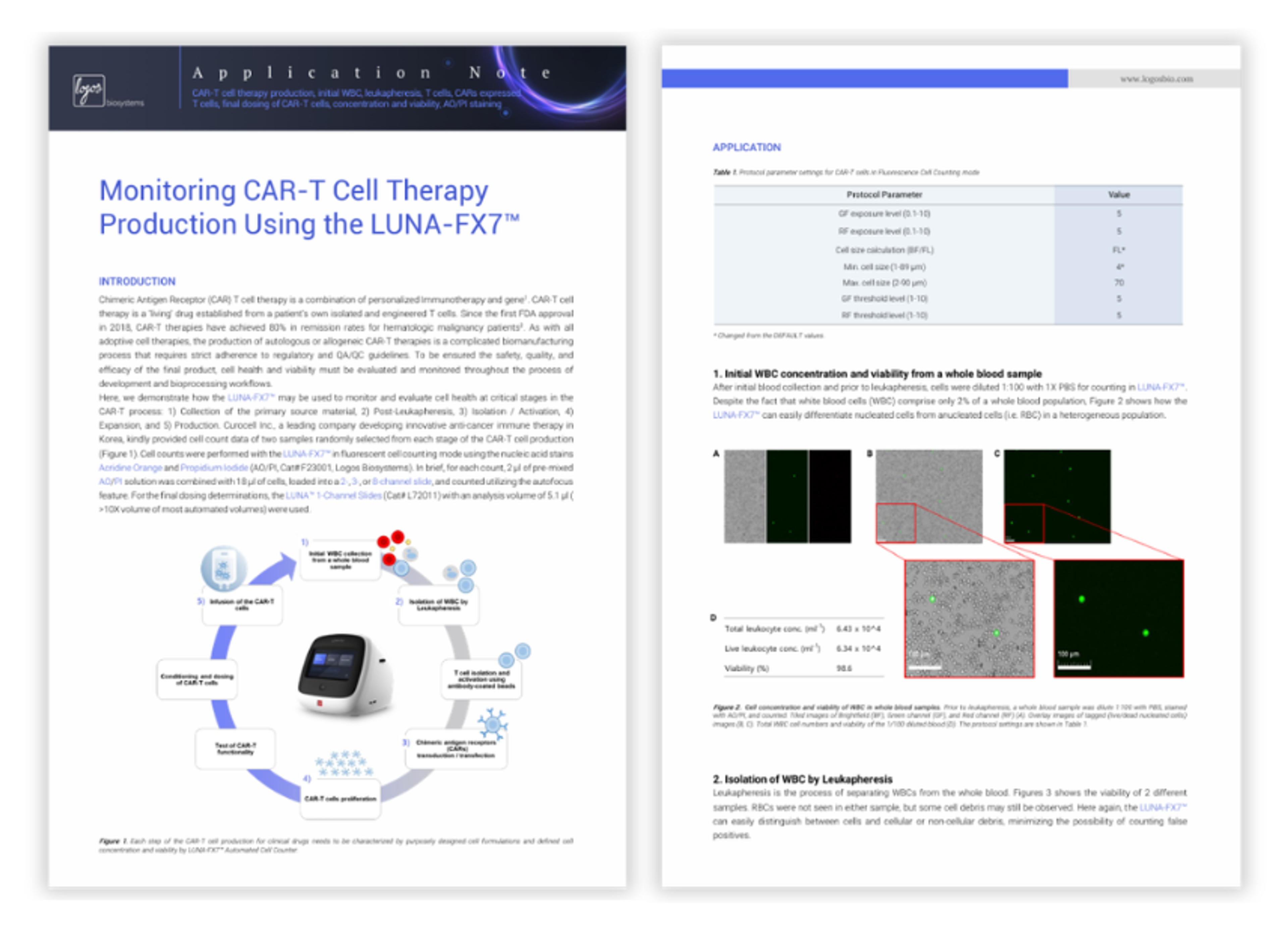
Cell therapy handbook
Application Note